
Bamford-Stevens Reaction and Carbenes
Interactive Video
•
Chemistry
•
11th - 12th Grade
•
Practice Problem
•
Hard
Amelia Wright
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the general formula for a carbene?
R3C
R2CH2
RCH
R2C
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main challenge in synthesizing cyclopropane?
Excessive stability
High ring strain
Complex reaction mechanism
Lack of suitable reactants
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the context of carbenes, what does 'in situ' mean?
Prepared in a gaseous state
Prepared in a solid state
Prepared in the reaction mixture
Prepared in a separate reaction
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do singlet carbenes typically react?
By losing electrons
As concerted reactions
By forming ionic bonds
Through stepwise radical additions
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
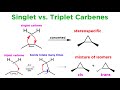
What is a key difference between singlet and triplet carbenes?
Singlet carbenes are diradicals
Triplet carbenes have a lone pair
Singlet carbenes react in a concerted manner
Triplet carbenes are more stable
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bonds do carbenes typically insert into?
Pi bonds
Sigma bonds
Ionic bonds
Metallic bonds
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a common application of carbene insertion?
Intramolecular cyclization
Breaking down polymers
Forming ionic compounds
Creating double bonds
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
