
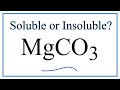
Magnesium Carbonate Solubility Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the main question addressed in the video regarding magnesium carbonate?
Whether it is a metal or non-metal
Its reaction with acids
Its solubility in water
Its melting point
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to solubility rules, which group of elements generally forms soluble carbonates?
Group 1 elements
Noble gases
Group 2 elements
Transition metals
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge on the carbonate ion as mentioned in the video?
2-
4-
1-
3-
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the solubility chart indicate about magnesium carbonate?
It is highly soluble
It reacts with water
It forms a precipitate
It is insoluble
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which ion is associated with magnesium in the solubility chart?
Mg+
Mg2+
Mg4+
Mg3+
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to magnesium carbonate when placed in water according to the video?
It dissolves completely
It changes color
It forms a gas
It sinks to the bottom
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the expected behavior of magnesium carbonate in water based on solubility rules?
It will remain mostly undissolved
It will evaporate
It will react violently
It will dissolve and dissociate
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
