
Understanding Polar and Non-Polar Molecules
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard

Nancy Jackson
FREE Resource
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What two factors primarily determine whether a molecule is polar or non-polar?
The molecular weight and the type of atoms
The polarity of chemical bonds and the shape of the molecule
The temperature and pressure conditions
The number of atoms and the type of bonds
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What causes bond polarity?
Molecular symmetry
Presence of lone pairs
Difference in electronegativity
Equal sharing of electrons
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is a dipole moment represented?
As a scalar quantity
As a vector quantity
As a complex number
As a matrix
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when two vectors of equal magnitude but opposite direction are added?
They create a scalar quantity
They double in magnitude
They cancel each other out
They form a new vector at 90 degrees
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a linear molecule like CO2, why is the molecule non-polar?
Because the dipole moments cancel each other out
Because it has no polar bonds
Because it has a high molecular weight
Because it is symmetrical
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the net dipole moment of a molecule with two polar bonds at an angle less than 180°?
Dependent on the molecular weight
Equal to the sum of the bond dipoles
Non-zero, indicating a polar molecule
Zero
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
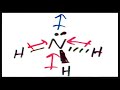
Why is water (H2O) considered a polar molecule?
Because it has no lone pairs
Because it is a diatomic molecule
Because its polar bonds are not in opposite directions
Because it has a linear shape
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
