

Elements, Compounds, & Mixtures
Presentation
•
Chemistry
•
8th Grade
•
Practice Problem
•
Medium
Standards-aligned
Michael Stange
Used 142+ times
FREE Resource
14 Slides • 18 Questions
1
Classifying Matter:
Elements, Compounds, & Mixtures

2
Matter is classified into 4 main categories:
Atoms
Elements
Compounds
Mixtures
3
Multiple Choice
This contains a variety elements and compounds that ARE NOT chemically combined.
Water
Mixture
Elements
Compounds
4
Multiple Choice
A substance made of only one type of atom is called —
an element
a mixture
a compound
a salt
5
Multiple Choice
1 A substance made up of two or more chemically combined elements is —
an element
mixture
compound
solution
6
Multiple Choice

7
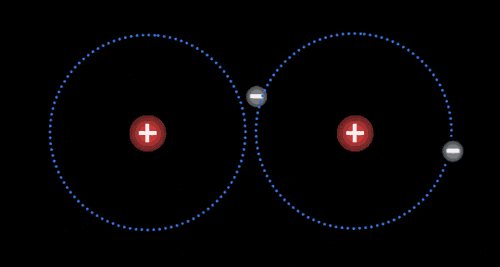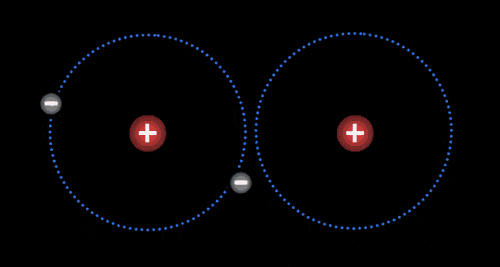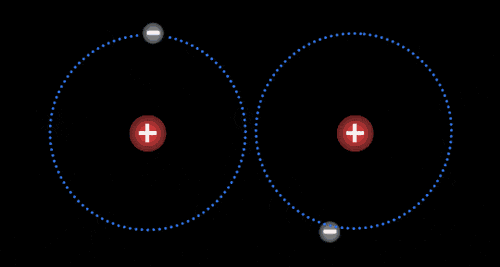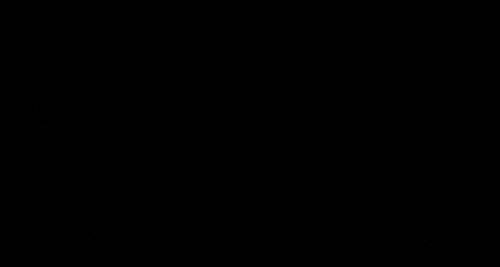
Atoms
The basic building block of all matter.
Made up of protons (+), neutrons (0), and electrons (-).
Different atoms have different numbers of these energy particles

8
Elements
Pure substance
Made up of identical atoms
Cannot be separated into a simpler substance by physical or chemical means

9
10
Periodic Table
Developed by Dmitri Mendeleev.
A way of organizing all of the known elements.
Elements are organized by the number of protons.

11

12
Multiple Choice
Elements can be broken down into simpler substances.
false
true
sometimes
13
Multiple Choice
A molecule can be made of the same kind of atoms.
true
false
Say What??!!!
Dude!! Sweet!!
14
Multiple Choice
Water is a(n) ___________.
element
mixture
compound
15
Compounds
Pure substance.
Made up of two or more different elements joined by chemical bonds.
Made of elements in a specific ratio that is always the same
Has a chemical formula

16
Chemical Formula
Specific ratio of elements in a compound
Makes it easier to describe elements and molecules
C + O2 = CO2 = Carbon Dioxide
N + H3 = NH3 = Nitrogen Trihydride (Ammonia)

17
Chemical Reactions
Process where different compounds react to form new compounds
Happens when Individual atoms from both compounds break their chemical bonds,Separate from their compounds, and
Form new chemical bonds with other recently-separated atoms.

18
Multiple Choice
NaHCO3 is an example of —
an abbreviation
a compound
a mixture
an element
19
Multiple Choice
Contains only one kind of atom.
Element
Compund
Mixture
20
Multiple Choice
21
Multiple Choice
A substance that cannot be broken down into any other substance by ordinary chemical means is called —
molecule
mixture
element
compound
22
Multiple Choice
1 A substance made up of two or more chemically combined elements is —
an element
mixture
compound
solution
23
Mixtures
A combination of two or more pure substances that are not chemically bonded.
Substances held together by physical forces, not chemical.
No chemical change takes place.
Each item retains its properties in the mixture.
Can be separated physically.

24
Types of Mixtures
There are two types of mixtures: homogeneous and heterogeneous.

25
Homogeneous
The components of a homogeneous mixture are uniformly distributed so the composition is the same throughout the mixture. (ex: steel, milk, kool-aid, blood).

26
Heterogeneous
The components of a heterogeneous mixture are not uniformly distributed and are separated into their own regions in the mixture (ex: pizza, cereal in milk, Oil & Vinegar, mixed nuts, bowl of different candies).

27
Multiple Choice
A mixture that appears to be evenly mixed throughout:
a homogeneous mixture
a heterogeneous mixture
28
Multiple Choice
This is made up of one or more of the SAME kind of atom
Element
Mixture
Compound
Matter
29
Multiple Select
How many of the following are elements?
Oxygen
H2O
Salt
Air
Carbon
30
Multiple Choice
Oil floating refusing to mix with water is an example of?
Homogeneous Mixture
Heterogeneous Mixture
Chemical Reaction
Compound
31
Multiple Choice
gold, Au, is an example of a(n)
element
compound
mixture
32
Multiple Choice
made up of?
Classifying Matter:
Elements, Compounds, & Mixtures

Show answer
Auto Play
Slide 1 / 32
SLIDE
Similar Resources on Wayground

24 questions
Notes Systems of Equations Graphing
Presentation
•
8th Grade

27 questions
Ionic and covalent bonding
Presentation
•
8th Grade

28 questions
Periodic Table Review
Presentation
•
8th Grade

25 questions
Explicit vs. Implicit Evidence
Presentation
•
8th Grade

26 questions
Scatterplots Lesson and Quiz
Presentation
•
8th Grade

24 questions
Review: Watersheds, Estuaries, and Dead Zones
Presentation
•
8th Grade

24 questions
10/17 - Weather Maps and Symbols
Presentation
•
8th Grade

24 questions
Interpreting Linear Graphs
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade