

8.5(D) & 8.5(E) - Checkpoint 3 Review
Presentation
•
Chemistry
•
8th Grade
•
Practice Problem
•
Medium
+3
Standards-aligned
Leonel ResendizLlanas
Used 66+ times
FREE Resource
23 Slides • 67 Questions
1
8.5(D) & 8.5(E) - Checkpoint 3 Review
8th Grade Science

2
8.5(D)
Recognize that chemical formulas are used to identify substances and determine the number of atoms of each element in chemical formulas containing subscripts.
3
How are elements represented?
- Elements are represented by a chemical symbol.
- This can be a single capital letter OR a capital letter with a lowercase letter.
- ONLY ELEMENTS are on the Periodic Table of Elements - NO COMPOUNDS.

4
Multiple Choice

What is the symbol for Iron?
Ir
In
Fe
FE
5
Multiple Choice

What is the name of the element with the symbol N?
Sodium
Nitrogonous
Nonium
Nitrogen
6
Multiple Choice

What is the chemical symbol for Hydrogen?
Hy
H
Hr
Hn
7
What are Compounds?
They are represented by a chemical formula.
The chemical formula has TWO or MORE elements.
Every capital letter is a new element present.

8
Multiple Choice
9
Multiple Choice

Can compounds be found in the periodic table?
Yes
No
10
Multiple Choice

Which of the following is not a compound?
HCl
Ag
NaCl
CO2
11
Multiple Choice
Na2CO3
12
Subscripts
The small number written directly behind the symbol of an element within a chemical formula.
It represents the number of atoms of that element.
If you DO NOT see a subscript it means there is ONLY ONE atom.

13
Subscripts
If there are parenthesis you must multiply the subscript by all the subscripts (INSIDE) the parenthesis.
Remember if you DO NOT see a subscript just multiply by one.

14
Multiple Choice
Fe(NO3)2
15
Multiple Choice
Fe(NO3)2
16
Multiple Choice
Fe(NO3)2
17
Multiple Choice
18
Coefficients
Number on the left side (or in front) of an element or formula.
The coefficient tells you how many molecules or compounds you have.
You must distribute the coefficient by multiplying it with each subscript.

19
Coefficients
You must distribute the coefficient by multiplying it with each subscript.
REMEBER,if there are parenthesis you must multiply the subscript by all the subscripts (INSIDE) the parenthesis.
THEN multiply all your atoms by the coefficient.

20
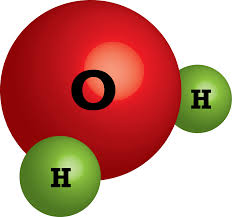
Multiple Choice

Which chemical formula below matches the image shown here?
2H2O
H4O2
4H2O
2HO
21
Multiple Choice
2C6H12O6
In this chemical formula, which number represents the coefficient (the number of molecules)?
2
6
12
1
22
Multiple Choice
Which image below could represent this formula?
2NaCl
23
Multiple Choice
3H2O
Which image below matches this chemical formula?
24

Let's Count Atoms!
25
Multiple Choice

Are there any compounds on the periodic table?
yes there are many
no because only elements are on the periodic table
I think they are?
there is no way to tell
26
Multiple Choice

How many atoms are present if there is no subscript following the element symbol?
0
2
only 1
1 million
27
Multiple Choice
How many elements are present in this compound? (Careful!)
Mg2As3COHNi2
2
4
6
10
28
Multiple Choice

How many Aluminum atoms are in Al2O3?
3
2
5
1
29
Multiple Choice
A subscript is the small number below the element symbol that tells the number of _______ of that element.
atoms
valence electrons
protons
elements
30
Multiple Choice
How many Hydrogen atoms in 3(NH₄)₂CrO₄?
4
2
8
24
31
Multiple Choice
How many Oxygen atoms are in Al₂(SO₄)₃?
4
12
7
24
32
Multiple Choice
How many atoms of Hydrogen are in this compound?
1
2
3
4
33
Multiple Choice
What is C2H8O ?
Element
Letters
Numbers
Compound
34
Multiple Choice

How many Nitrogen atoms are in (NH₄)₂CrO₄?
1
2
8
16
35
Multiple Choice
How many Sodium atoms are in 6NaCl?
1
12
6
36
Multiple Choice
How many atoms are there TOTAL in H2SO4 ?
6
5
7
3
37
Multiple Choice
38
Multiple Choice
How many Magnesium atoms are in 10MgCl2?
10
5
20
39
Multiple Choice
How many Oxygen atoms are in H2O?
1
2
0
4
40
Multiple Choice
How many Hydrogen atoms are in 4H2O?
6
8
2
4
41
8.5(E)
Investigate how evidence of chemical reactions indicates that new substances with different properties are formed and how that relates to the law of conservation of mass.
42

43
Physical Change
ONLY changes the form or the appearance of a substance.
Does not change or form a new substance.
Phase Changes

44

45
Multiple Choice
46
Multiple Choice
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
What kind of change occurs if matter changes in amount, size, or shape?
substance change
chemical change
physical change
elemental change
50
Chemical Changes
Substance is transformed into one or more different substances, such as when iron becomes rust.
IRREVERSIBLE - Cannot change it back to its original form.
A new product is produced.

51

52
Multiple Choice

53
Multiple Choice

54
Multiple Choice

55
Multiple Choice
56
Multiple Choice
57
Multiple Choice
58
Multiple Choice

59
Chemical Reactions
a process in which atoms of one or more substances (reactants) are rearranged to form one or more new substances (products).
Chemical changes are the result of chemical reactions.

60
Indicators of a Chemical Reaction:
COLOR CHANGE
GAS PRODUCTION
LIGHT PRODUCTION
TEMPERTURE CHANGE
SMELL/ODOR
PRECIPITATE

61
Open Ended
In your own words, tell me what is a precipitate and how is it formed?
62

63
Multiple Choice

Identify what type of change happening in the picture below...
Chemical Reaction
Physical Change
Change in State
Change in appearance only
64
Multiple Choice
The formation of a solid when two liquids combine is called?
Solidification
Freezing
Precipitate
Solubility
65
Multiple Choice
A Chemical Change is also known as what?
Physical Change
Physical Property
Chemical Reaction
Chemical Property
66
Multiple Choice
A green powder is heated & a gas is given off while it turns to a black solid. This describes
Physical Change
Chemical Change
Change in State
Change in Temperature
67
Multiple Choice
During Chemical Reactions, the new substances created are called?
Precipitate
Corrosion
Reactants
Products
68
Multiple Choice
During Chemical Reactions, the substances that are used are called?
Precipitate
Corrosion
Reactants
Products
69
The Law of Conservation of Mass
Mass or matter cannot be created nor destroyed.
Reactants EQUAL Products

70
Reactants
What you START with.
LEFT side.
EQUAL to the products.

71
Products
What you END with.
RIGHT side.
EQUAL to the reactants.

72
Multiple Choice
73
Multiple Choice
74

Mass is NEITHER created nor destroyed.
75
Multiple Choice
76
Multiple Choice
77
Multiple Choice
A chemical reaction is balanced when
both sides have the same elements
Both sides have the same number of atoms
Same subscripts
Same coefficients
78
Multiple Choice
79
Multiple Choice
80
Multiple Choice
81
Multiple Choice
82
Multiple Choice

83
Multiple Choice
84
Multiple Choice
85
Multiple Choice
86
Multiple Choice

87
Multiple Choice

88
Multiple Choice

How many Carbons are present on the reactant side?
1
2
3
4
89
Multiple Choice
A chemical change is different than a physical change because in a chemical change
chemicals are used
molecules do not physically touch
a new substance is formed and in a physical no new substance is formed
the change can be seen but in a physical change it cannot
90

Study and complete your exit ticket!
8.5(D) & 8.5(E) - Checkpoint 3 Review
8th Grade Science

Show answer
Auto Play
Slide 1 / 90
SLIDE
Similar Resources on Wayground

87 questions
Topic 12 Lesson 3 & 4 Notes
Presentation
•
8th Grade

86 questions
Thursday August 15Lesson
Presentation
•
9th Grade

83 questions
Copy of Unit 3 - Texas Revolution Master Slidedeck
Presentation
•
7th Grade

84 questions
F4 Ready For Anything L20
Presentation
•
9th Grade
![SS7 - Fall Common Assessment 2 [Units 1 & 2] / PART 2](https://cf.quizizz.com/image/image-loader.svg)
83 questions
SS7 - Fall Common Assessment 2 [Units 1 & 2] / PART 2
Presentation
•
7th Grade

87 questions
Ecology STAAR Review
Presentation
•
8th Grade

88 questions
RL2 Part 2 Retell Stories GA Milestones Review
Presentation
•
KG

83 questions
F3(S) The Wonders of Nature L25
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade