

Structure of Matter
Presentation
•
Science
•
8th Grade
•
Medium
Standards-aligned

C Krejci
Used 21+ times
FREE Resource
16 Slides • 19 Questions
1
Structure of Matter
YOU WILL WANT TO HAVE YOUR NOTEBOOK HANDY...
GET IT NOW!
I tell you this for a reason, even if you have no notes in it.

2
YOU WILL WANT TO HAVE YOUR NOTEBOOK HANDY...
GET IT NOW!
I tell you this for a reason, even if you have no notes in it.
3
2H2 + O2 = 2H20
(reactant) (product)
Notice that the
reactant is on the LEFT side and the product is on the RIGHT side of the chemical reaction
Write this down!
4
The Law of Conservation of Matter/Mass
That the number of atoms on the reactant side must equal the number of atoms on the product side
5
The Law of Conservation of Matter/Mass
That the number of atoms on the reactant side must equal the number of atoms on the product side
NO ATOMS ARE CREATED OR DESTROYED during a reaction
6
The Law of Conservation of Matter/Mass
That the number of atoms on the reactant side must equal the number of atoms on the product side
NO ATOMS ARE CREATED OR DESTROYED during a reaction
You will have the same amount of each type of atom on each side of the reaction
7
The Law of Conservation of Matter/Mass - Write this down!
That the number of atoms on the reactant side must equal the number of atoms on the product side
NO ATOMS ARE CREATED OR DESTROYED during a reaction
You will have the same amount of each type of atom on each side of the reaction
The mass on each side of the reaction must total the same on each side of the reaction
8
2H2O means
there are 2 molecules of water
Write this down!
9
2H2O means
there are 2 molecules of water...
made of 4 atoms of Hydrogen
write this down!
10
2H2O means
there are 2 molecules of water...
made of 4 atoms of Hydrogen
and 2 atoms of Oxygen
Write this down!
11
2H2O means
there are 2 molecules of water...
made up of 4 atoms of Hydrogen,
and 2 atoms of Oxygen?
...so basically the big 2 means multiply everything in that molecule
Write this down!
12
Elements * Write this down!
on the periodic table
are a single type of ATOM
13
Elements * Write this down!
on the periodic table
are a single type of ATOM
Each element is made of an atom of a different size
14
Elements * Write this down!
on the periodic table
are a single type of ATOM
Each element is made of an atom of a different size
Example: Carbon (C) is made of an ATOM with 6 protons & 6 neutrons & 6 electrons
Example: Potassium (K) is made of an ATOM with 19 protons & 19 neutrons & 19 electrons
15
Elements * Write this down!
on the periodic table
are a single type of ATOM
Each element is made of an atom of a different size
Example: Carbon (C) is made of an ATOM with 6 protons & 6 neutrons & 6 electrons
Example: Potassium (K) is made of an ATOM with 19 protons & 19 neutrons & 19 electrons
NO2 would not be an element, it has more than one atom
16
Now get ready, we are doing a quiz on everything we have learned so far...
This counts as a QUIZ GRADE.
17
Multiple Choice
When a chemical reaction is balanced, what do you know about the atoms on each side?
The number of atoms on each side are equal.
The number of atoms on each side are way different.
The number of atoms might change.
18
Multiple Choice
In the reaction
2H2 + O2 = 2H2O
The molecules involved are...
Reactant: H = 2 O = 1
Product: H20 = 2
Yes, this is correct. The number of molecules on each side can vary, it's all about the number of atoms.
No, a reaction cannot have 3 molecules as a reactant and 2 as a product.
Yes, this is correct. A reaction cannot have 3 molecules as a reactant and 2 as a product.
No, a reaction should have the the same molecules on each side.
19
Multiple Choice
In the reaction
2H2 + O2 = 2H2O
The atoms involved are...
Reactant: H = 4 & O = 2
Product: H = 4 & O = 2
true
false
20
Multiple Choice
Two or more atoms chemically bonded together are called a
atom
periodic table
molecule
mixture
21
Multiple Choice
2H2 + O2 = 2H20
What does the underlined 2 mean?
There are 2 molecules of water in the product.
There are 2 atoms of water in the reactant.
There are 2 molecules of water in the reactant.
There are 2 atoms of water in the product.
22
Multiple Choice
The mass of the products of a reaction will always equal what?
The same sum of the mass of the reactants
You never can tell
A bit more than the mass of the reactants
23
Multiple Choice
2H2 + O2 = 2H20
What does the blue 2 mean?
There are 2 oxygen atoms bonded together to make an oxygen molecule in the reactant.
There are 4 oxygen molecules in the reactant.
Water is what we started with and what we ended up with in the reaction.
24
Multiple Choice
What does the law of conservation of matter say?
Matter is neither created nor destroyed in a chemical reaction.
Matter is always destroyed in any ordinary chemical reaction.
Chemical reactions always result in a heavier substance.
25
Multiple Choice
2H2 + O2 = 2H20
How many molecules of hydrogen are in the reactant?
2
1
0
4
26
Multiple Choice
2H2 + O2 = 2H20
How many atoms of Hydrogen are in the reactant?
4
2
8
1
27
Multiple Choice
What happens during a chemical reaction?
The atoms of the original substances rearrange themselves into a new configuration to form new substances.
Molecules are lost somewhere.
Molecules turn into protons and neutrons.
28
Multiple Choice
How are elements different from atoms?
Elements are made out of atoms
Atoms are made out of elements
Elements are made out of molecules
Molecules are made out of atoms.
29
Multiple Choice
How many carbon atoms are there in C3H12O5?
1
3
12
5
30
Multiple Choice
How many different types of atoms are in NO2?
1
2
3
4
31
Multiple Choice
Is K an element or a molecule?
element
molecule
32
Multiple Choice
Is NO2, Sodium Dioxide, an element or a molecule? (hint* Where do we find Elements?)
element
molecule
33
Multiple Choice
Elements are made of
many types of atoms
the compound table of molecules
one type of atom, just of various sizes
molecules
34
Multiple Choice
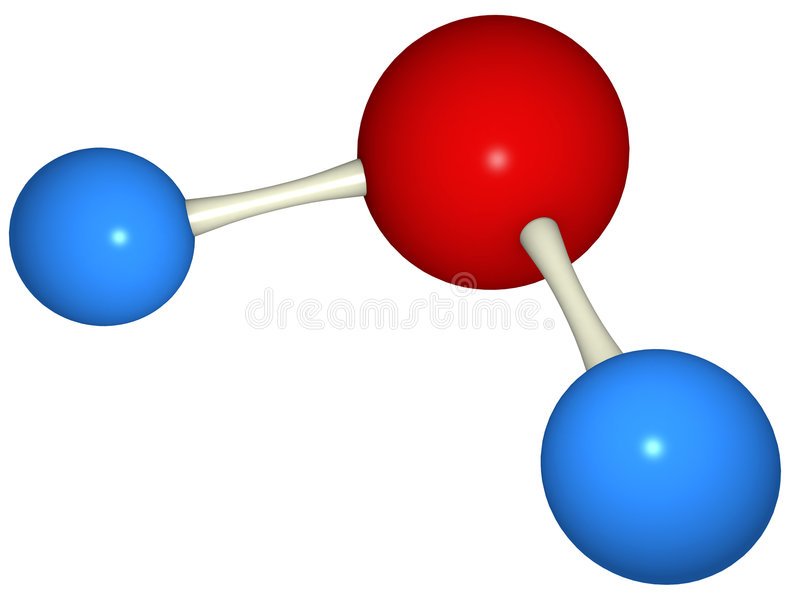
How many hydrogen atoms are there in a water molecule? H20
0
1
2
3
35
Multiple Choice
What would be the correct order of complexity? From most basic to most complex
Atoms->Elements->Molecules
Elements->Atoms->Molecules
Compounds->Molecules->Atoms
Molecules->Electrons->Atoms
Structure of Matter
YOU WILL WANT TO HAVE YOUR NOTEBOOK HANDY...
GET IT NOW!
I tell you this for a reason, even if you have no notes in it.

Show answer
Auto Play
Slide 1 / 35
SLIDE
Similar Resources on Wayground

26 questions
Energy In = Energy Out
Presentation
•
8th Grade

25 questions
Atoms & Periodic Table
Presentation
•
8th Grade

25 questions
Chemical Bonds
Presentation
•
8th Grade

33 questions
Wave Interaction Notes
Presentation
•
8th Grade

33 questions
Chemical Properties
Presentation
•
8th Grade

28 questions
Atomic Structure
Presentation
•
8th Grade

27 questions
Phases and Eclipses
Presentation
•
8th Grade

30 questions
Measuring Matter
Presentation
•
8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

18 questions
Carbon Cycle
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

10 questions
Evidence of Evolution
Presentation
•
6th - 8th Grade

20 questions
8th Grade Science NC EOG Review
Quiz
•
8th Grade