

Review for Unit 3 Summative Assessment
Presentation
•
Chemistry
•
9th - 12th Grade
•
Easy
Janeen Miklowski
Used 2+ times
FREE Resource
3 Slides • 49 Questions
1
Review for Unit 3 Summative Assessment

2
What should you know? Unit 3a:
Types of matter (atom, element, mixtures, compound)
Homogeneous vs. heterogeneous mixtures and techniques of separation
States of matter: solid, liquid, gas
Physical and chemical changes and properties
Indicators of chemical reaction
Parts of a chemical equation
Express elements and compounds with symbols and formulas, respectively

3
Multiple Choice
Name the part in red:
H2 + O2 -> H2O
Coefficient
Product
Yield
Subscript
4
Multiple Choice
How many atoms of aluminum are on each side of the following equation: 4Al + 3O2 --> 2Al2 O3
2
6
1
4
5
Multiple Choice
Which side of a chemical equation is the product side?
Left (before the yields sign)
Right (after the yields sign)
6
Multiple Choice
In the chemical equation H2O2(ag) → H2O(l) + O2(g), the O2 is a
reactant
yield
product
solid
7
Multiple Choice
Chemical reactions
create and destoy atoms
only occurs outside living organisms
occurs only in living organisms
produce new substances
8
Multiple Choice
The products of a reaction have the same ________as the reactants.
molecule
atoms
subscripts
coefficients
9
Multiple Choice

Which scientific observation, shown in the chart, indicates that a chemical reaction has NOT occurred?
Number 1
Number 2
Number 3
Number 4
10
Multiple Choice
A scientist spilled a few drops of dilute hydrochloric acid (HCl) on a lab table. For safety purposes, the scientist sprinkled some baking soda ( NaHCO3 ) onto the spill. Which observation would provide the best evidence that a chemical reaction occured?
The baking soda and hydrochloric acid combined, and bubbles formed.
The baking soda absorbed the hydrochloric acid.
Some of the baking soda dissolved in the hydochloric acid.
The hydrochloric acid evaporated, leaving only the baking soda.
11
Multiple Select
Which lab experiments below describe a situation where a chemical change has occurred? Check all that apply.
After adding zinc (Zn) to hydrochloric acid (HCl), a student observes bubbles forming in the solution and a thermometer registers a 10° change in temperature.
A student uses a mortar and pestle (a grinding stone) to break up large pieces of ammonium thiocyanate ( NH4SCN ) into smaller pieces.
After combining Unknown A (a solid, white crystal) with Unknown B (a clear, odorless liquid), there is a strong smell produced and a thermometer registers a 15° change in temperature.
A student dissolves sugar ( C6H6O6 ) into water ( H2O ). The sugar crystals seem to disappear.
12
Multiple Choice
Pb is the element symbol for....
lead
gold
peanutbutter
nitrogen
13
Multiple Choice
What's the symbol for copper?
C
Co
Cu
CP
14
Multiple Choice
What is the process of separating products with densities?
centrifugation
chromatography
heating
decantation
15
Multiple Choice
To separate a mixture of soil and water what separation method is used?
Filtration
Distillation
Chromatography
Evaporation
16
Multiple Choice
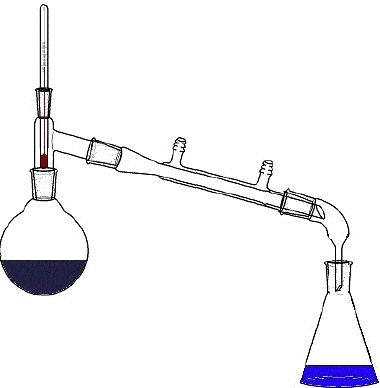
Water and alcohol are easily separated by distillation because of their
different melting points
different colors
different densities
different boiling points
17
Multiple Choice
Which one of the following would you use to separate sand from iron filings?
a bar magnet
filter paper
chromatography paper
alum
18
Multiple Choice
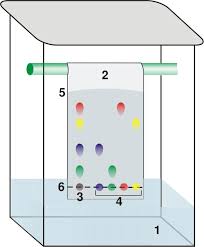
Dyes in water soluble markers may be separated by means of..
crystallization
sublimation
chromatography
sedimentation
19
Multiple Select
Which of the following is the correct set-up of filtration?
20
Multiple Choice

Which state of matter has molecules that move fastest and spread to fill the space they are in? They do not form any pattern.
solid
liquid
gas
21
Multiple Choice
If you had two different substances that looked exactly the same, which properties would you use to tell them apart?
Sound
Size
Color
Density
22
Multiple Select
Choose the 3 that are physical properties of aluminum foil
Malleable
Luster
Oxidation
High melting point
23
Multiple Choice
Electrical conductivity is an example of what kind of property?
Chemical
Large
Physical
Boiling
24
Multiple Choice
Choose the one that is a chemical property
Magnetism
Size
Color
Reactivity with water
25
Multiple Choice
Choose which one is a physical property
Flammability
Solubility
Water Reaction
Magic
26
Fill in the Blanks
Type answer...
27
Multiple Select
Mark all examples of a chemical change occurring
liquid water evaporates
a 12V current passes through water, breaking is down from
H2O (l) into H2 (g) and O2 (g) (electrolysis)Iron rusts
Coffee cools down to a drinkable temperature
28
Multiple Choice
When ice melts to water at 0 degrees C, that is an example of what type of change?
chemical
physical
intensive
property
29
Multiple Choice
I give one student 1 gallon of coffee and a second student 1,000 gallons of coffee and ask them to find the density of coffee. Will the students have the same answer?
yes because density is an intensive property
yes because density is an extensive property
no because density is an extensive property
no because density is an intensive property
30
What should you know? Unit 3b:
Explain the contributions of 4 major scientists/philosopher: Democritus, Dalton, Thomson, Rutherford
Define the points of Dalton's atomic theory and know which points were modified
Be able to identify the evolution of the atomic model (Dalton's, Thomson's, Rutherford's)
Be able to identify subatomic particles, their role in the atom, location, charge, mass contribution to the atom
Identify and interpret mass and atomic numbers
Define what an isotope is and some of their uses as radioisotopes
Calculate the average atomic mass and distinguish it from mass number

31
Multiple Choice
On the periodic table, how is atomic mass represented?
weighted average of all the element's isotopes based on their natural abundance.
as the exact mass of every atom
as the mass of the most common isotopes
As the masses of all protons added together
32
Multiple Choice
An element has two naturally occurring isotopes. One is 10.013 amu and is 19.9% abundant. The other is 11.01 amu and is 80.1% abundant. What is the average atomic mass? What element is it?
9.012, Beryllium
12.011, Carbon
6.941, Lithium
10.812, Boron
33
Multiple Choice
If a sulfur atom has 16 protons, 16 electrons, and 16 neutrons, its mass number is:
16
32
48
64
34
Multiple Choice
Isotopes are atoms of the same element that have the same number of __________ but different number of __________ . Therefore, isotopes of the same element have different masses.
protons, neutrons
protons, electrons
neutrons, protons
electrons, protons
35
Multiple Choice

How many protons would be present in this atom?
35.453
17
18
35
36
Multiple Choice

Using this element, how many electrons would be present?
108
47
61
107.87
37
Multiple Choice
Where is most of the mass in an atom?
electron cloud
nucleus
it's equally distributed among the whole atom
there is no mass in an atom
38
Multiple Choice

Which number identifies the number of protons?
12
24.305
24
24-12
39
Multiple Choice

How many neutrons does a Calcium-48 isotope have?
16
20
28
12
40
Multiple Choice
How many neutrons in C-14?
6
7
14
8
41
Multiple Choice

How many neutrons does this isotope of lithium have?
8
3
4
5
42
Multiple Choice

What do these isotopes of carbon all have in common?
same number of neutrons & same mass number
same atomic number and same number of neutrons
same atomic number and same number of electrons
same number of protons, same atomic number, and same mass number
43
Multiple Choice

When you add the number of protons and neutrons together, you are calculating...
Atomic Number
Atomic Mass
Mass Number
Pythagorean's Theorem
44
Multiple Choice

How did Rutherford describe the nucleus?
Tiny
Dense
Positively Charged
all of the above
45
Multiple Choice

What two particles are part of the nucleus of an atom?
Protons and Electrons
Electrons and Neutrons
Protons and Neutrons
Alpha and Beta
46
Multiple Choice

What was the name of the model?
Oatmeal Model
Rutherford Model
Bohr Model
Plum Pudding Model
47
Multiple Choice

Which of the following are parts of Dalton's theory?
All substances are made of atoms.
Atoms join with other atoms to make new substances.
Atoms of different elements are different
All of these are part of his theory
48
Multiple Choice

Whose model is this?
Millikan
Rutherford
Thomson
Bohr
49
Multiple Choice
How did Thomson build upon Dalton's model?
His model was the same.
He put electrons in orbitals.
He discovered the nucleus.
He discovered subatomic particles (electron).
50
Multiple Choice
The first person to propose a theory about an atom called the Atomos Theory was
Democritus
Rutherford
Dalton
Aristotle
51
Multiple Choice
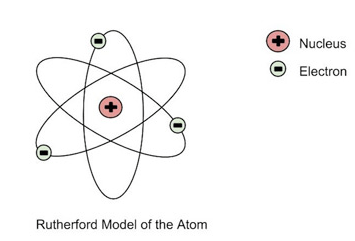
Rutherford's gold foil experiment provided evidence that...
negative and positive charges are spread evenly throughout the atom.
alpha particles have a positive charge.
gold is not a dense as previously thought.
there is a dense positively charged nucleus at the center of an atom.
52
Multiple Choice

What contribution did John Dalton make to atomic theory?
He discovered that every atom was positively charged.
He discovered that every element consisted of one type of atom that couldn't be divided.
He discovered that atoms had nuclei.
He discovered that atoms could be divided into smaller parts.
Review for Unit 3 Summative Assessment

Show answer
Auto Play
Slide 1 / 52
SLIDE
Similar Resources on Wayground

48 questions
Moles, Mass & Equations
Presentation
•
9th - 12th Grade

45 questions
3.2 Gas Laws
Presentation
•
9th - 12th Grade

47 questions
Naming Ionic Compounds
Presentation
•
9th - 12th Grade

44 questions
Unit 2 Lesson 4 Atomic Theory and PEN
Presentation
•
10th - 12th Grade

51 questions
Unit 2: Lesson 7 Half Life Decay
Presentation
•
10th - 12th Grade

45 questions
Measurements in Science and Sci Notation
Presentation
•
10th - 12th Grade

47 questions
Unit 7 Molar Relationships Presentation Part 1
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade