

Calculating Density
Presentation
•
Science
•
6th - 8th Grade
•
Practice Problem
•
Medium
Standards-aligned
Helina Bedasa
Used 164+ times
FREE Resource
12 Slides • 22 Questions
1
Density Practice
6th Grade Science

2
Watch this video for an explanation of calculating Density.
3
Multiple Select
Check all the parts of the formula that you need to calculate density
Mass
Force
Acceleration
Volume
4
To Calculate Density You NEED:
Mass
Volume
5
Multiple Choice
What is the formula for Density?
Density = Volume ÷Mass
Denisty=Volume ×Mass
Density=Mass÷Volume
6
This is the formula for Density
7
Multiple Choice
What is the Density of water?
100 g/ml
1 g/ml
2 g/ml
1.5 g/ml
8
1 g/cm3 or 1 g/ml
This is the Density of water
9
Float or Sink?
You will have 30 seconds to decide if the object will float or sink in water. (REMEMBER THE DENSITY OF WATER IS 1)
10
Multiple Choice
The density of ice is 0.93 g/ml. Will ice float or sink in water?
Float
Sink
11
Multiple Choice
The density of the metal tin is 5.75 g/cm3 , will tin float or sink in water?
Float
Sink
12
Multiple Choice
The density of gasoline is 0.74 g/cm3, will gasoline float or sink in water?
Float
Sink
13
Multiple Select
Check all items that will sink in water.
Wood has a density of 0.85 g/cm3
Aluminum has a density of 2.7 g/cm3
Lithium has a density of 0.53 g/cm3
Gold has a density of 19.3 g/cm3
14
Open Ended

The density of different objects are shown in the table. If the density of water is 1g/ml, which of the objects will float in water and which will sink?
15
This is the formula for Density
16
Tips for Calculating Density
First find the mass and the volume
Divide the Mass by the volume
17
Multiple Choice
A bar of copper has a mass of 216g and a volume of 24 cm3. What is the density of copper? (You can use a calculator)
9g/cm3
5184g/cm3
.11g/cm3
322mL
18
Multiple Choice
Frank has an eraser. It has a mass of 4 g and a volume of 2cm3. What is the density?
2 g/cm3
4 g/cm3
8 g/cm3
2 g/in3
19
Multiple Choice
Jack has a rock. The rock has a mass of 14g and a volume of 2cm3. What is the density of the rock?
7 mL
7 g/cm3
28 g/cm3
1/7 g/cm3
20
Calculating for Volume
There will be questions that ask you to find the volume before you can calculate Density.
21
How to calculate Volume:
If you are given an image like the one shown here. You have to subtract the number the water rises to minus the number the water started at. It rises to 35 and started at 30 in the image so the volume is 35-30 which is 5.

22
Multiple Choice

A scientist is trying to calculate the density of green jade. He first must calculate the volume. The image shows the amount of water that is displaced by the green jade. Use this information to calculate volume.
11.5 mL
7.25 mL
10.5 mL
5.0 mL
23
Multiple Choice

What is the volume of the yellow block?
10 mL
15 mL
20 mL
5 mL
24
Multiple Choice
It is the amount of space occupied by the object
volume
density
mass
liquids
25
Multiple Choice

Calculate the Volume: (Remember the formula to calculate volume l x w x h)
84 cm3
420 cm3
60 cm3
144 cm3
26
Calculating for Mass
There will be questions that give you a triple beam balance picture to find the mass before calculating for Density.
27
Multiple Choice

What does this triple beam balance read?
285.2 g
282.5 g
280.2 g
28
Fill in the Blanks

Type answer...
29
Fill in the Blanks

Type answer...
30
Putting it all together to calcuate Density
First use the image to find Volume
Then use the image to find Mass
Finally Divide the Mass by the Volume
31
Multiple Choice

An irregularly shaped rock was lowered into a graduated cylinder holding a volume of water equal to 50 ml. The height of the water rose to 75 ml. If the mass of the stone was 250 g, what was its density?
15 g/mL
5 g/mL
10 g/mL
20 g/mL
32
Multiple Choice
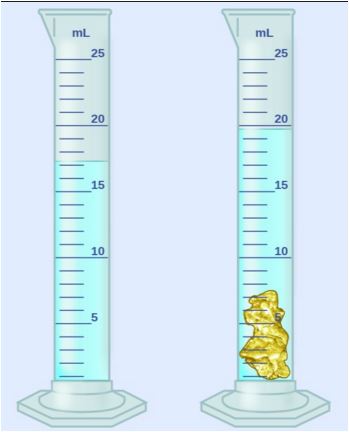
An irregularly shaped piece of gold was lowered into a graduated cylinder holding a volume of water equal to 17 ml. The height of the water rose to 20 ml. If the mass of the gold was 27 g, what was its density?
9 g/mL
10.5 g/mL
6.5 g/mL
8 g/mL
33
Multiple Choice
If a block of wood has a density of 0.6 g/cm3 and a mass of 120 g, what is its volume?
200 cm3
0.005 cm3
250 mL
72 mL
34
Multiple Choice

There are 24 cm3 Find the total mass.
Density Practice
6th Grade Science

Show answer
Auto Play
Slide 1 / 34
SLIDE
Similar Resources on Wayground

24 questions
Climate Regions Review
Presentation
•
6th - 8th Grade

30 questions
States of Matter
Presentation
•
7th - 8th Grade

27 questions
Levels of Organization
Presentation
•
5th - 7th Grade

28 questions
Contact and Non-contact Forces
Presentation
•
6th - 8th Grade

26 questions
Water Pollution
Presentation
•
6th - 8th Grade

24 questions
Watersheds
Presentation
•
6th - 8th Grade

26 questions
Intro to Waves
Presentation
•
5th - 8th Grade

26 questions
Sexual vs Asexual Reproduction
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

45 questions
8th Grade Science - EOG Review
Quiz
•
8th Grade

20 questions
Human Body Systems
Quiz
•
6th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade