

Density lesson
Presentation
•
Physics, Science
•
6th Grade
•
Medium
Kasthuri Subramaniam
Used 19+ times
FREE Resource
7 Slides • 45 Questions
1
Density lesson

2

3

4

5

6

7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
What is the volume of a cube with the following dimensions length = 2cm, width = 2cm, height = 2cm?
8 cm
8 cm3
6 cm
6 cm3
11
Multiple Choice
Find the mass of a container that has a volume of 380 cm3 and a density of 0.97 g/cm3
368.6 grams
0.0026 grams
391.8 grams
None of the above
12
Multiple Choice
When does an object float?
If it has a density of Less than 1 g/ml
if it has a density of more than 1 g/ml
It has a density of exactly 1 g/ml
None of the above
13
Multiple Choice
What is the density of water?
10 g/ml
1 g/ml
0.1 g/ml
100 g/ml
14
Multiple Choice
A box has a mass of 50 grams and a volume of 30 cm3. What is the density of the rock?
0.6 g/cm3
80 g/cm3
1500 g/cm3
1.67 g/cm3
15
Multiple Choice
How do you find the density of an object
Mass times Volume
Mass divided by Volume
Volume times Mass
Volume divided by Mass
16
Multiple Choice

Which box has the higher density?
Box A
Cannot be determined
Box B
They are the same
17

18
Multiple Choice
19
Multiple Choice

What is the mass on this triple beam?
506g
576g
570g
20
Multiple Choice

What is the value on the balance?
240.0 g
142.0 g
100.4 g
21
Multiple Choice

What is the value on the balance?
901.5 g
900 g
190.4 g
22
Multiple Choice

Find the mass.
550.4 g
559.4 g
550.6 g
23
Multiple Choice

What is the volume of the ring? (new volume - old volume)
64 mL
68 mL
8 mL
4 mL
24
Multiple Choice
25
Multiple Choice
What is the curve at a liquid's surface called?
meniscus
graduated cylinder
displacement
depth
26
Multiple Choice
What instrument is used to measure volume?
ruler
triple beam balance
graduated cylinder
scale
27
Multiple Choice
What is the formula for measuring the volume of a regular solid?
length x width
length x width x height
water displacement method
width x height
28
Multiple Choice
Steven finds the volume of a graduated cylinder with a penny in it. The volume was 43 mL. If the volume before the penny was added was 38 mL. Then what is the volume displacement?
4 mL
4
5
5 mL
29
Multiple Choice

50 mL of water was added to the graduated cylinder. After we drop in the object, the new water level goes up to 60 mL. What is the volume of the blocks? (Hint: new volume- old volume)
8 mL
10 mL
60 mL
5 mL
30
Multiple Choice
displacement
a procedure used to measure volume of a solid object by placing it in a known amount of liquid
measures how much matter is present in a substance
a metric unit of measurement used to measure small volumes of liquid
31
Multiple Choice

32
Multiple Choice

33
Multiple Choice
34
Multiple Choice
Which of the following is an example of an object with a low density?
Pillow
Bowling Ball
Brick
35
Multiple Choice
In a density column, is the substance with the highest density on the bottom or the top?
Bottom
Top
36
Multiple Choice
37
Multiple Choice
38
Multiple Choice

Will a peeled lemon float or sink?
Float
Sink
39
Multiple Choice

Will a lemon float or sink?
Float
Sink
40
Multiple Choice

Will a paper clip float or sink?
Float
Sink
41
Multiple Choice
Two cubes of the same size were measured. Cube A was 52 grams and cube B was 130 grams. Which cube has the greatest density?
A
B
42
Multiple Choice
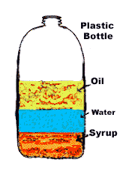
When two materials that have different densities, like oil and water, encounter one another, they will?
exchange densities.
mix uniformly.
repel one another.
separate according to their densities.
43
Multiple Choice
You are given a mystery object in science class. Your teacher asks you to determine if your object is more or less dense than water. You place the object in the water and it sinks. What statement would best describe this situation?
The mystery substance is denser than water.
Water is denser than the mystery substance.
They both have the same mass.
The mystery substance must be hollow.
44
Multiple Choice

What causes an object to sink in a water?
if the object's density is less than the water's density.
if the object's density is the same as water's density.
if the object's density is greater than water's density.
if the object's density is zero
45
Multiple Choice

Which box has a higher density?
A
B
They are the same
46
Multiple Choice

Use the picture to select the correct statement.
Oil is denser than water
Pasta is less dense then crayon
The paper clip is denser than every thing else
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice

51
Multiple Choice
If a 96.5g piece of aluminum has a density of 2.7 g/cm3, what is its volume?
Hint: density = mass/volume
35.7 cm3
260.55 cm3
0.03 cm3
99.2 cm3
52
Multiple Choice
If the density of a diamond is 3.5 g/cm3 , what would be the mass of a diamond whose volume is .5 cm3?
Hint: density = mass/volume
1.75 g
7 g
0.14 g
4 g
Density lesson

Show answer
Auto Play
Slide 1 / 52
SLIDE
Similar Resources on Wayground

45 questions
Protons, Neutrons, and Electrons - How they differ. Outline
Presentation
•
6th Grade

45 questions
3/20/23
Presentation
•
6th Grade

45 questions
Mountain Building
Presentation
•
6th Grade

42 questions
Module F Unit 2 Lesson 1 - Earth's Oceans and the Ocean Floor
Presentation
•
6th Grade

46 questions
Heat Transfer Review 23-24
Presentation
•
6th Grade

47 questions
ACAP Review 6th grade math
Presentation
•
6th Grade

44 questions
Microbiome Review
Presentation
•
6th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade