

Y10T1W10L3 Group I: The Alkali Metals
Presentation
•
Chemistry, Science
•
10th Grade
•
Practice Problem
•
Medium
Christina Celine Sutherland
Used 19+ times
FREE Resource
18 Slides • 14 Questions
1
Y10T1W10L2 Group I: The Alkali Metals

2
3
Open Ended
Starter: Why are elements grouped together?
Extension: Think about their atomic structure and the bonding what would result from their reactions.
4
5

6
Multiple Choice
How many outer shell electrons do the alkali metals have?
1
2
4
7
7
Multiple Choice
What type of ions are formed by the alkali metals?
+4
-1
-2
+1
8
9

10
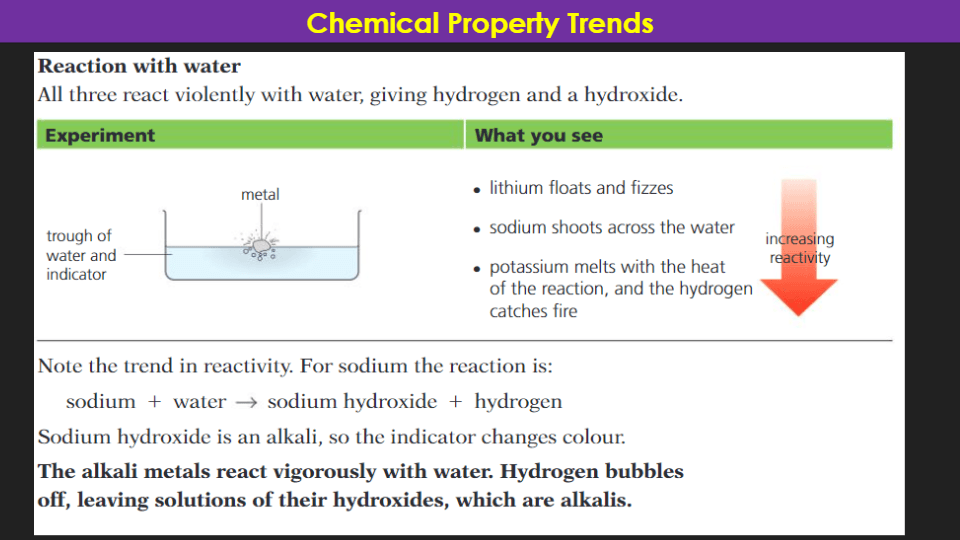
11
Multiple Choice
What is the correct order of reactivity of alkali metals, from least to most reactive?
Na, Li, K
K, Na, Li
Li, Na, K
Li, K, Na
12

13
Open Ended
Task One: Alkali Metals
1.Why are the metals stored in sealed glass jars with oil?
2.Why do the metals have darker appearances on their outside and are lighter inside when cut?
3.How does the softness of each metal change as you move down the group?
4.Describe the trend in reactivity as you move down the group.
14

15
16
Multiple Choice
What are the products of the reaction between an alkali metal and water?
A metal oxide and oxygen
A metal oxide and hydrogen
A metal hydroxide and oxygen
A metal hydroxide and hydrogen
17
Multiple Choice
What are the products of the reaction between an alkali metal and air?
A metal oxide and oxygen
A metal oxide and hydrogen
A metal oxide
A metal hydroxide and hydrogen
18

19

20

21
Open Ended
Task 2: Alkali Metal Reactions
1. Write balanced chemical equations (not word equations) for:
a)Lithium reacting with oxygen
b)Potassium reacting with water
c)Sodium reacting with chlorine
2. Describe the test for hydrogen gas?
22

23
24
Multiple Choice
True or False
Alkali metals are so reactive, they must be stored in oil
True
False
25
Multiple Choice
True or False
Alkali metals need to be heated to react with air
True
False
26
Multiple Choice
True or False
Sodium lights with a purple flame when it reacts with water
True
False
27
Multiple Choice
True or False
All alkali metals float in water
True
False
28
Multiple Choice
True or False
Hydrogen is produced when an alkali metal reacts with water
True
False
29
Multiple Choice
True or False
Universal indicator turns blue when added to water in which an alkali metal has reacted
True
False
30

https://www.physicsandmathstutor.com/chemistry-revision/igcse-cie/periodic-table/
31

32

Y10T1W10L2 Group I: The Alkali Metals

Show answer
Auto Play
Slide 1 / 32
SLIDE
Similar Resources on Wayground

25 questions
Thermochemistry pt 1
Presentation
•
10th Grade

21 questions
CARBON AND ITS COMPOUNDS
Presentation
•
9th - 10th Grade

23 questions
Scientific Method
Presentation
•
9th - 10th Grade

21 questions
Scientific Method
Presentation
•
10th Grade

22 questions
Nomenclatura
Presentation
•
9th - 10th Grade

24 questions
Periodic Table Introduction
Presentation
•
9th Grade

25 questions
LECȚIE: Decalogul sau Cele zece porunci
Presentation
•
10th Grade

28 questions
Section 2 Lesson 5-Empirical Formulas
Presentation
•
10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade

34 questions
Nuclear Chemistry Review
Quiz
•
9th - 12th Grade