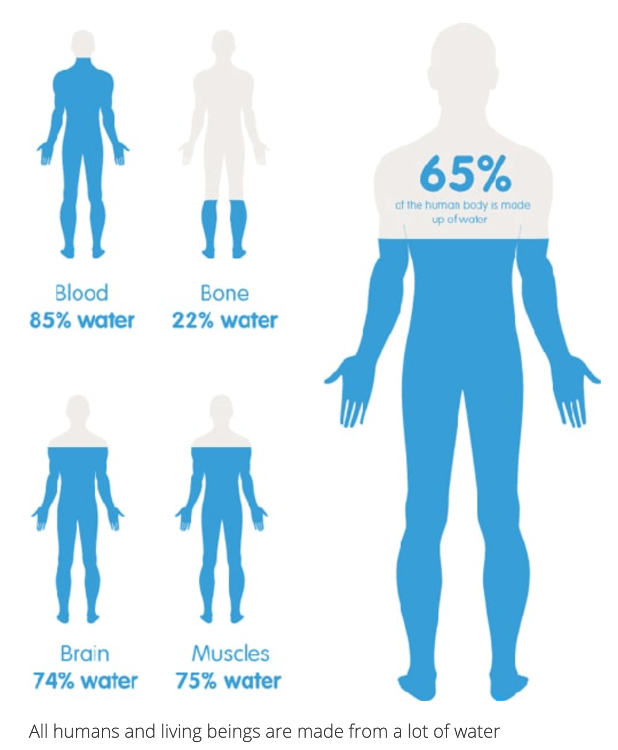
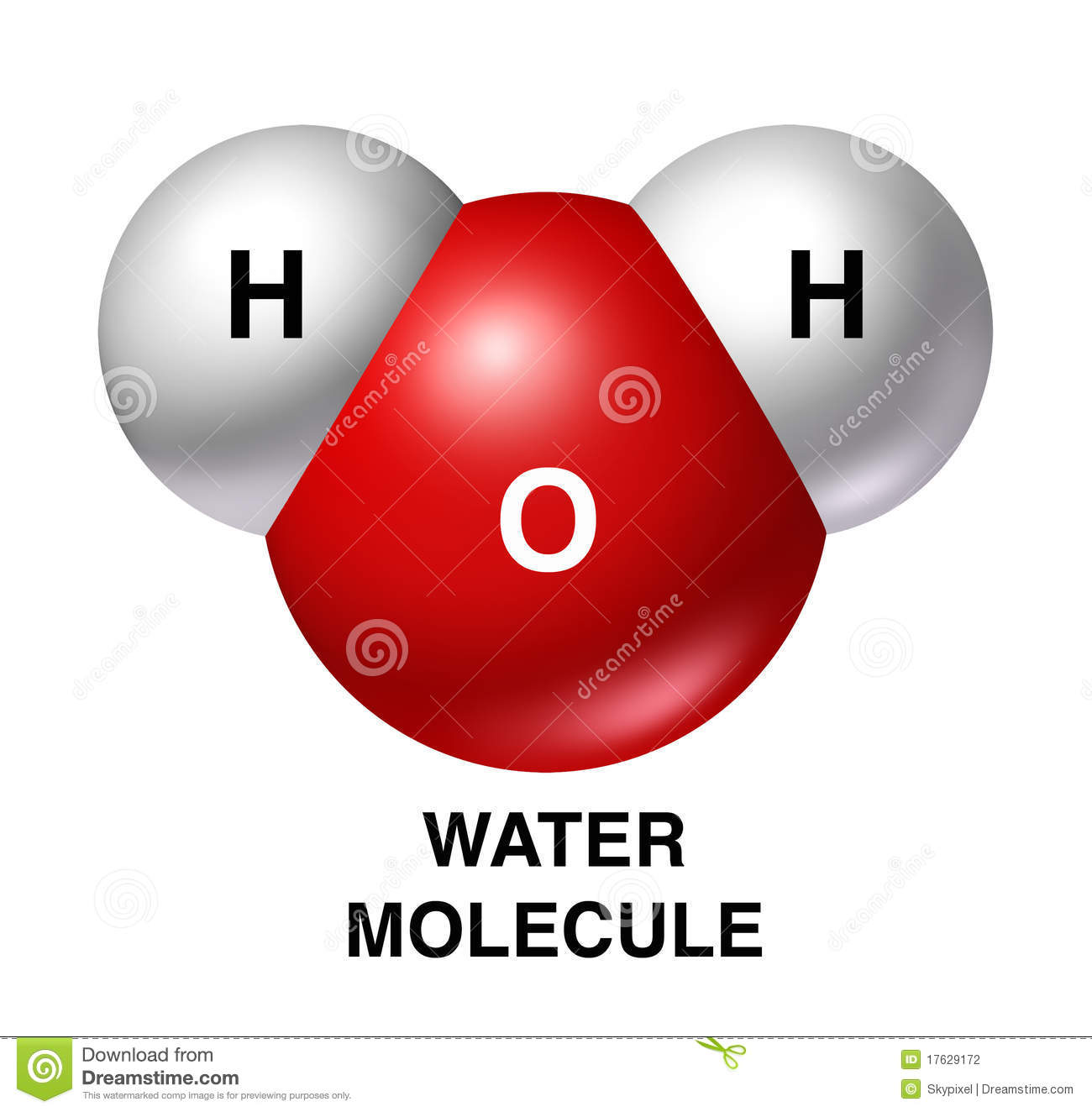
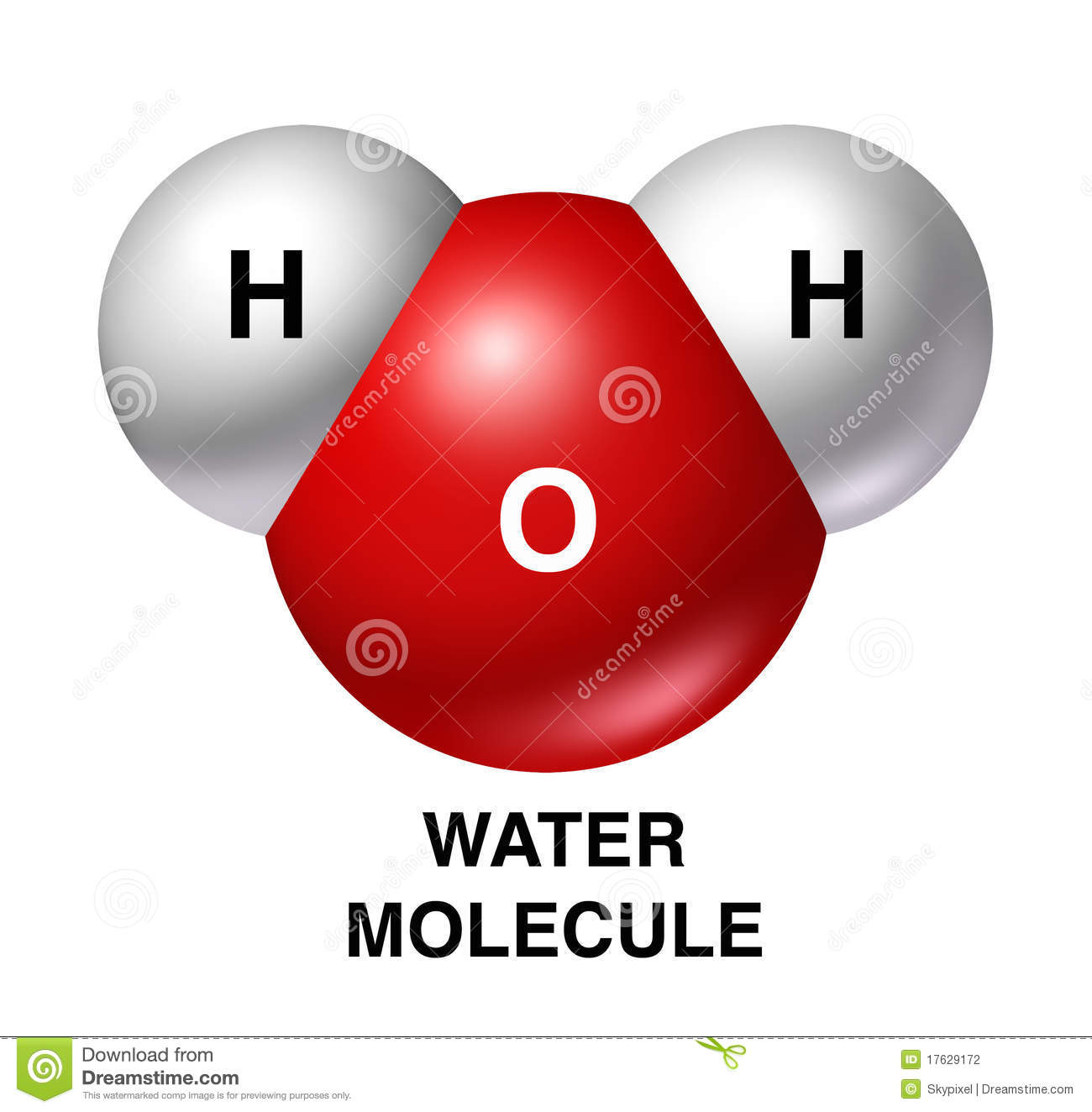
2 Lesson: BIO.A.2.1. Water Properties
Presentation
•
Biology
•
9th - 12th Grade
•
Practice Problem
•
Medium
+1
Standards-aligned
Rose Marsh
Used 19+ times
FREE Resource
13 Slides • 14 Questions
1
2
3
4
5
6
7
8
9
10
11
12
13
14
Multiple Choice
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
Show answer
Auto Play
Slide 1 / 27
SLIDE
Similar Resources on Wayground

22 questions
2.1 The Structure of DNA Lesson
Lesson
•
9th - 12th Grade

20 questions
Motivation and Emotion
Lesson
•
9th - 12th Grade

20 questions
Properties of Water
Lesson
•
9th - 12th Grade

21 questions
Healthy 4 Life
Lesson
•
9th - 12th Grade

21 questions
Introduction to DNA
Lesson
•
9th - 12th Grade

21 questions
4 Lesson: BIO.A.4.1 Cell Membrane and Diffusion
Lesson
•
9th - 12th Grade

24 questions
Lesson 11 Cell Differentiation
Lesson
•
9th - 12th Grade

22 questions
DNA Replication!
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

10 questions
Probability Practice
Quiz
•
4th Grade

15 questions
Probability on Number LIne
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

6 questions
Appropriate Chromebook Usage
Lesson
•
7th Grade

10 questions
Greek Bases tele and phon
Quiz
•
6th - 8th Grade
Discover more resources for Biology

22 questions
Human Body Systems Overview
Quiz
•
9th Grade

25 questions
photosynthesis and cellular respiration
Quiz
•
9th Grade

18 questions
Enzymes and Their Functions Quiz
Quiz
•
9th Grade

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade

30 questions
Exploring the World of Plants
Quiz
•
9th Grade

65 questions
Review for TEST 1
Quiz
•
9th - 10th Grade

10 questions
Exploring Food Webs and Energy Pyramids
Interactive video
•
6th - 10th Grade

20 questions
Meiosis and Sexual Reproduction
Quiz
•
9th Grade