

Electron Configuration
Presentation
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Medium
Standards-aligned
Audrey Nebzydoski
Used 386+ times
FREE Resource
13 Slides • 7 Questions
1
Electron Configuration
Mrs. Nebzydoski's Lesson

2
Electron Configuration tells you the location that an electron "lives" at in an atom
There are four types of orbital shapes
S, p, d, & f
3
Multiple Choice
There are _____ types of atomic orbitals.
1
2
3
4
4
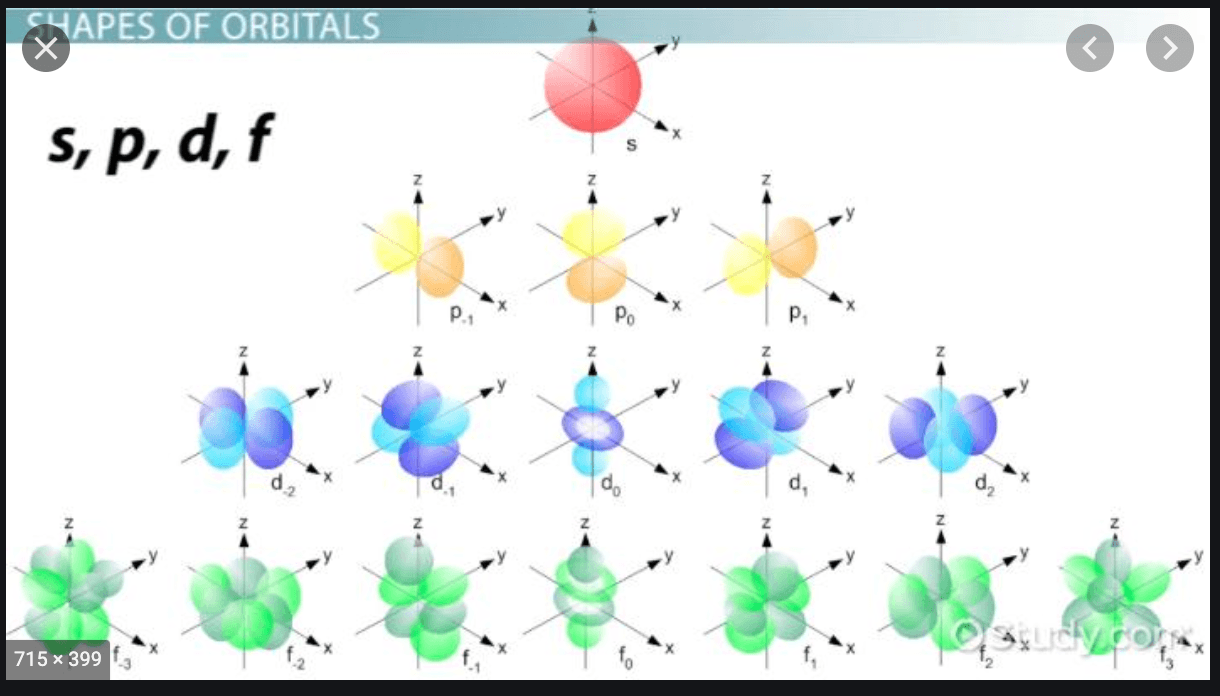
5
The orbital shapes can be described as the path that the electron takes around the nucleus.
There is:
1 s orbital
3 p orbitals
5 d orbitals
7 f orbitals
6
Orbital shapes
As you can see, the s orbital is a circular path around the nucleus and that is why there is only one option. The p orbital is a figure-8 path that can be on the x-axis, the y-axis, or the z-axis in a 3-D atom. This is why there are 3 different p orbitals.

7
Multiple Select
Which of the following are TRUE of atomic orbitals?
There is 1 s orbital
There are 3 p orbitals
There are 5 d orbitals
There are 7 f orbitals
8
Each orbital can contain up to 2 electrons
s orbitals can hold a total of 2 electrons (1 orbital)
p orbitals can hold a total of 6 electrons (3 orbitals)
d orbitals can hold a total of 10 electrons (5 orbitals)
f orbitals can hold a total of 14 electrons (7 orbitals)
9
Multiple Choice
How many electrons can fit in one orbital?
1
2
3
4
10
Periodic Table and Orbitals
The periodic table gets its shape from the type of atomic orbitals that are filled in the atoms of the collumns.

11
The blocks of the periodic table
As you can see here the "s" block is shown on the left in pink, the "p" block is on the right in orange, the "d" block is in the middle shown in blue, and the "f" block is shown below in yellow.

12
Multiple Choice
Look at a periodic table. Count how many columns on the periodic table are in the "p" block. How many columns did you find?
2
6
10
14
13
Columns on the periodic table and the orbitals...
The s block contains 2 columns
The p block contains 6 columns
The d block contains 10 columns
The f block contains 14 columns

14
Think about it....
The s block contains 2 columns and the "s" orbitals can contain 2 electrons. The p block contains 6 columns and the "p" orbitals can contain up to 6 electrons. The d block contains 10 columns and the the "d" orbitals can contain up to 10 electrons! (This is NOT a coincidence!)

15
Multiple Choice
If the f block contains 14 columns, then it would make sense that an f orbital can hold up to _____ electrons.
2
5
7
14
16
Orbital filling
Electrons fill these orbitals in a way that allows them to take the easiest root possible. The LOWEST energy levels are filled first.

17
Orbital filling cheat sheet
You copied this orbital filling diagram cheat sheet in your notes. It is important that you always use it to fill electrons into orbitals when completing electron configurations.

18
Multiple Choice
Using your orbital filling cheat sheet, tell me which orbital fills after the 2 p orbital is full.
3p
3s
2s
1p
19
Multiple Choice
Using your orbital filling cheat sheet, which orbital fills after the 4s orbital?
4p
5s
3d
4f
20
Up next....Electron Configuration!
The end.
Electron Configuration
Mrs. Nebzydoski's Lesson

Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

19 questions
Review Periodic Table
Presentation
•
10th - 12th Grade

16 questions
Stoichiometry - Limiting Reagents & Percent Yield
Presentation
•
9th - 12th Grade

11 questions
Multistep Stoichiometry
Presentation
•
10th - 12th Grade

15 questions
Dividing Complex numbers
Presentation
•
10th - 12th Grade

14 questions
Camera Parts
Presentation
•
10th - 12th Grade

14 questions
Angle Relationships
Presentation
•
10th - 12th Grade

18 questions
Physical Vs. Chemical Changes/Properties
Presentation
•
10th - 12th Grade

16 questions
Counting Atoms
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade