

Y10T2W7L2 Revision Separating Techniques
Presentation
•
Chemistry
•
10th Grade
•
Medium
Christina Celine Sutherland
Used 7+ times
FREE Resource
22 Slides • 25 Questions
1

2

3
Open Ended

4

5

6

7

8

9

10

11

12

13

14

15
Open Ended

16

17

18

19

20

21

22
Open Ended

23

24

25

26
Multiple Choice

Ethanol has a higher boiling point than water.
Ethanol has a lower melting point than water.
Ethanol has a higher melting point than water.
Ethanol has a lower boiling point than water.
27
Multiple Choice

This makes it impossible to calculate Rf values.
This makes it impossible to use a locating agent.
This results in a safety hazard caused by solvent fumes.
This causes the spot nearest the bottom of the paper to catch up with the spot above it.
28
Multiple Choice
29
Multiple Choice
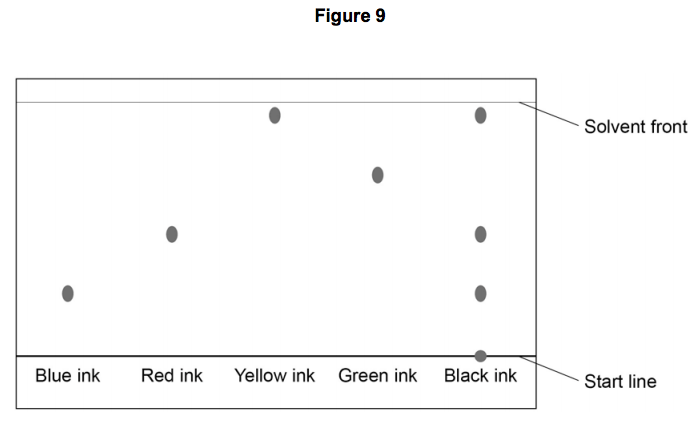
Which ink is most soluble?
Blue
Red
Yellow
Green
30
Multiple Choice

A
B
C
D
31
Multiple Choice
Select the correct equation to calculate Rf values.
(Distance travelled by solvent) ÷ (Distance travelled by sample)
(Distance travelled by sample) ÷ (Distance travelled by solvent)
32
Multiple Choice

Label number 1 in the above experiment.
Solvent front
Solvent
Baseline
33
Multiple Select
Rf values can be obtained with the following information.
Distance moved by the solvent.
Width of the baseline.
Length of the chromatography paper.
Distance that the substance has moved.
34
Multiple Choice
What is the method suitable to obtain pure water from seawater.
Simple distillation
Fractional distillation
Filtration
Evaporation to dryness
35
Multiple Choice

A
B
C
D
36
Multiple Choice
What method is used to obtain copper(II) sulfate crystals from its solution?
Crystallisation
Evaporation to dryness
Paper chromatography
Fractional distillation
37
Multiple Choice
A mixture contains two substances. What property must be the two substances have for them to be separated by paper chromatography?
Both substances must be soluble in the chromatography solvent,
Both substances must have low boiling point.
Both substances must be coloured.
Both substances must be solid at room condition.
38
Multiple Choice

39
Multiple Choice
Which property indicates that a solid is pure?
It burns completely in oxygen, leaving no residue.
It has a fixed melting point.
It has a fixed volume.
It is soluble in water.
40
Multiple Choice
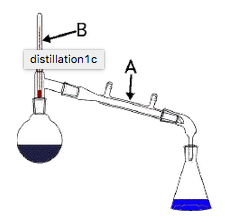
41
Multiple Choice
42
Multiple Choice
Distillation is used to separate ______.
an insoluble solid from its solution.
a soluble solid from its solution.
43
Multiple Choice
A pure substance shows -------spot(s) on chromatogram
0
1
2
3
44
Multiple Choice
45
Multiple Choice
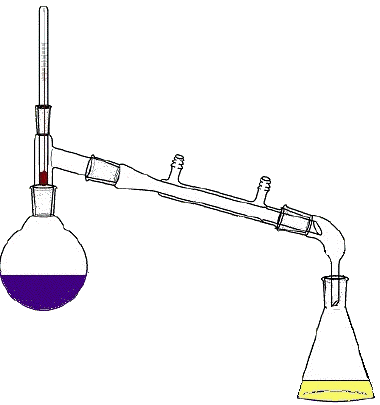
46
Multiple Choice
Distillation is made up of ___
boiling, evaporation and then condensation
condensation, boiling and evaporation
47
Multiple Choice
A leibeg condenser is used in...
Filtration
Chromatography
Evaporation
Distillation

Show answer
Auto Play
Slide 1 / 47
SLIDE
Similar Resources on Wayground

44 questions
Chemistry Chapter 5 Section 2
Presentation
•
11th Grade

42 questions
Matter and Energy Review
Presentation
•
9th - 10th Grade

39 questions
organic compounds
Presentation
•
11th Grade

40 questions
Week 3_ Part 2__Effects of Intermolecular Forces of Attraction
Presentation
•
11th Grade

42 questions
Cells Cell Theory
Presentation
•
9th Grade

41 questions
chemical equillibrium
Presentation
•
9th Grade

42 questions
Chemical Bonding
Presentation
•
10th Grade

44 questions
X-CET-Biology-Lecture 1
Presentation
•
10th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade