

Matter and Energy Review
Presentation
•
Science
•
9th - 10th Grade
•
Medium
+5
Standards-aligned
Katie Borton
Used 11+ times
FREE Resource
10 Slides • 32 Questions
1

2
Matter Notes
Slide 8 has some vocab for to write down
Slide 9 something you need to know about periodic tables, atoms and matter.
There is a periodic table on slide 10.
Good Luck!
3

4

5

6

7

8
Atom: anything that has mass and takes up space
Element: a substance consisting of the same atoms that cannot be broken up any further.
Compound: a substance consisting of two or more different atoms
Electron: negatively charged subparticle located outside of the atom's nucleus
Proton: positively charges subparticle located inside of the atom's nucleus
Neutron: subparticle without a change located inside the atom's nucleus
Matter Stuff to know...
9

10

This Periodic table only has
Atomic Numbers listed.
***Reminder that Atomic Number the number of protons present in the atom!***
11
Multiple Choice
The center of an atom where its neutrons and protons are is called the _____________.
nucleus
electron cloud
center
12
Multiple Choice
An atom with atomic number 6 would have how many protons?
6
12
3
13
Multiple Choice
14
Multiple Choice

15
Multiple Choice

16
Multiple Choice
What do we call a substance consisting of a single type of atom?
molecule
element
compound
substance
17
Multiple Choice
18
Fill in the Blanks
Type answer...
19
Multiple Choice

Which state of matter has both definite shape and definite volume?
Gas
Liquid
Solid
20
Multiple Choice

Which state of matter has no definite shape and no definite volume?
Gas
Liquid
Solid
21
Multiple Choice

Which state of matter has no definite shape but does have definite volume?
Gas
Solid
Liquid
22
Multiple Choice
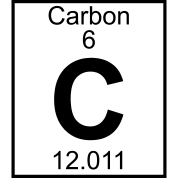
23
Multiple Choice
The amount of ______ in a substance will determine its state
atom
energy
compounds
mixture
24
Multiple Choice
Did you watch and listen to the video? What is the difference between water and hydrogen peroxide?
less energy
an extra hydrogen
more energy
an extra oxygen
25
Multiple Choice
True or False: The law of conservation of energy and the law of conservation of mass must exist in unison.
True
False
26
Multiple Choice
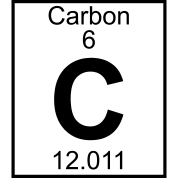
27
Multiple Choice
28
Multiple Choice
the basic structural unit of matter
atom
matter
element
nucleus
29
Multiple Choice
a collection of atoms of one type that cannot be broken down into simpler substances by ordinary chemical or physical means
element
subatomic particle
electron
matter
30
Multiple Choice
the positively charged subatomic particle contained in the nucleus of an atom
proton
neutron
electron
matter
31
Multiple Choice
a subatomic particle, contained in the nucleus of an atom, having the same mass as a proton but no electrical charge
proton
neutron
electron
matter
32
Multiple Choice
a negatively charged subatomic particle that orbits the nucleus of an atom
proton
neutron
electron
matter
33
Multiple Choice
a unique number for each element that equals the number of protons in an atom of that element
element name
element symbol
atomic number
atomic mass
34
Multiple Choice
the mass of an atom, calculated by adding the number of protons and neutrons in the nucleus of the atom
element name
element symbol
atomic number
atomic mass
35
Multiple Choice

36
Multiple Choice

37
Multiple Choice

38
Multiple Choice
Everything in the universe is compose of
matter
energy
both
what?
39
Fill in the Blanks
Type answer...
40
Multiple Choice
True or False: There are more than 3 states of matter
True
False
41
Fill in the Blanks
Type answer...
42
Multiple Choice
_____ combine to determine what substance is formed
atoms
mixtures
energy
compounds

Show answer
Auto Play
Slide 1 / 42
SLIDE
Similar Resources on Wayground

37 questions
CP9a Electric circuits
Presentation
•
9th - 10th Grade

37 questions
Part I: Unit 7 PPT Review Lesson
Presentation
•
9th - 10th Grade

36 questions
Proving Triangles Congruent (Intro to Proofs)
Presentation
•
9th - 10th Grade

37 questions
Human Impact Biology Lesson
Presentation
•
9th Grade

36 questions
8.2 Fluids
Presentation
•
9th - 10th Grade

38 questions
Basic Economic Concepts
Presentation
•
9th - 10th Grade

38 questions
Periodic Table
Presentation
•
9th Grade

39 questions
Classification terms 2023
Presentation
•
9th - 10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade