

2.9 Covalent Bonding
Presentation
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Medium
Standards-aligned
Matthew Martino
Used 163+ times
FREE Resource
18 Slides • 7 Questions
1
2.9 Covalent Bonding
Sharing electrons to achieve stability.
It's how nonmetals bond with other nonmetal atoms.
Molecules are formed. (discrete, electrically-neutral particles of matter... they be small y'all)

2

3
Covalent Bonds involve nonmetals only.
...Who are the Nonmetals again?
Elements to the right of the staircase line AND Hydrogen , H

4
Prior Knowledge
Electron configuration
Valence electrons
location of nonmetals on P.T.
Octet Rule
rapid fire set of questions up next. Be ready to flip to periodic table

5
Multiple Choice
What is the electron configuration of chlorine? (symbol, Cl , Group 17)
2-7
2-8-7
2-8-8
6
Multiple Choice
How many valence electrons does an atom of chlorine have?
2
7
8
7
Multiple Choice
If forming an ion, What will an atom of chlorine do to achieve 8 valence electrons?
gain 1
lose 7
8
If a metal atom is around, 1 electron will transfer to the chlorine atom... making it a 1- chloride ion (that is ionic bonding)
But what if there are no metal atoms around?
What if there are only other nonmetal atoms nearby?
9
Covalent Bonding
Sharing valence electrons , not transferring them.

10
Key information for Covalent Bonding
" Co " = share ; "valent" = valence shell (valence electrons)
A Covalent Bond is: A chemical bond formed when two atoms share valence electrons. (Typically, two nonmetal atoms)
Sharing takes place until each atom has access to 8 valence electrons (Octet Rule ; exceptions of Hydrogen... needs only 2)
Example: In a molecule of Cl2 , each chlorine atom donates an electron into a covalent bond in the molecule , which results in each Cl atom having access to 8 total valence electrons (each started with 7).
11
Hydrogen is a nonmetal and is an exception to the Octet Rule
It only needs to form one covalent bond and have access to 2 valence electrons.
Can bond to itself, making H2
Or to others, such as carbon (CH4)

12
Electrons are shared to form one, two, or three bonding pairs
Single Covalent Bond: One shared pair (Ex: in Cl2)
Double Covalent Bond: Two shared pairs (Ex: in O2)
Triple Covalent Bond: Three shared pairs (Ex: in N2)
Bonding pairs of electrons represented as dots or as dashes in Lewis Dot Diagrams (examples on next slide)
13

14

15

16
New Term: Valency
The number of covalent bonds that an atom needs to reach stability
(Typically, to reach access to 8 valence electrons.)
How-to:
Simply count the number of unpaired electrons in the atom's Lewis Dot Diagram
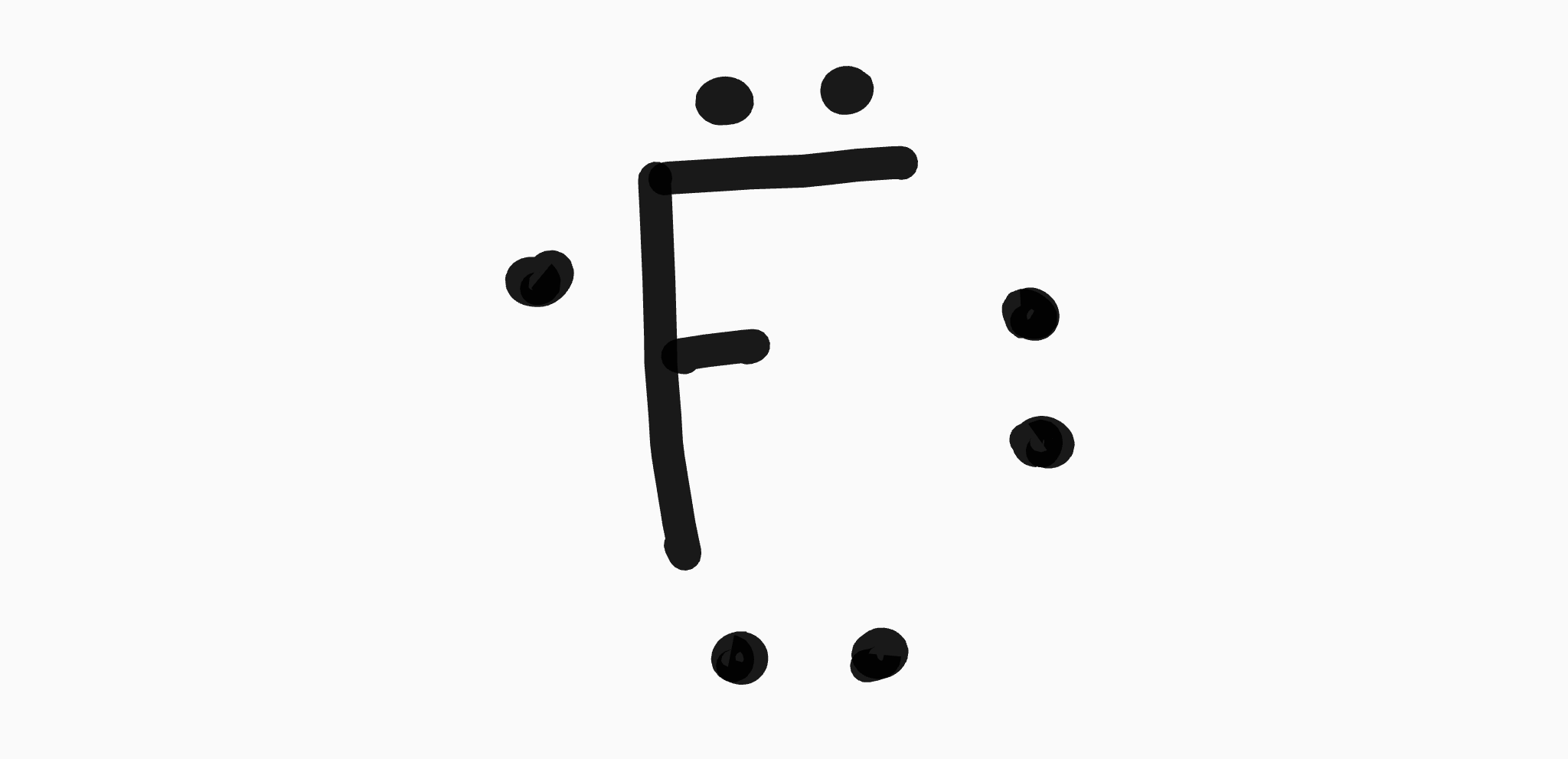
17
Multiple Choice

How many unpaired electrons in the Lewis Dot Diagram of an atom of fluorine? (this will determine how many covalent bonds it needs to form... its valency)
(click the image to enlarge)
1
2
3
18
Multiple Choice

How many covalent bonds does an atom of oxygen need to form to be stable?
(click image to enlarge)
1
2
3
19

20
Lewis Dot Diagrams of nonmetal atoms are the key to determining how they bond with each other
But how to draw them?

21

(Mr. M demonstrating after the Quizizz Lesson concludes)
22
Recap
nonmetals can form covalent bonds with other nonmetal atoms
covalent bonds involve the sharing of valence electrons
Typically, each atom contributes one valence electron into each covalent bond (thus, bonding pairs are formed)
The number of unpaired valence electrons an atom has determines how many covalent bonds it needs (a.k.a. its valency)

23
Multiple Select
Which of the following elements could be involved in covalent bonds?
Select all that apply.
H, hydrogen
Na, sodium
S, sulfur
Fe, iron
24
Multiple Choice
Covalent bonding involves the ...
transferring of electrons from one atom to another
the sharing of electrons between atoms
the attraction between metal cations and a sea of mobile electrons
25
For Definitions and Examples of Covalent Bonding, Lewis Dot Diagrams and Structures... go to Quizlet
"2.9 Covalent Bonding"

2.9 Covalent Bonding
Sharing electrons to achieve stability.
It's how nonmetals bond with other nonmetal atoms.
Molecules are formed. (discrete, electrically-neutral particles of matter... they be small y'all)

Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

20 questions
Average Atomic Mass and Abundance
Presentation
•
9th - 12th Grade

20 questions
Calculating Atomic Mass
Presentation
•
9th - 12th Grade

19 questions
Chemical Bonding
Presentation
•
9th - 12th Grade

21 questions
Reaction Rates
Presentation
•
9th - 12th Grade

23 questions
Periodic Table
Presentation
•
9th - 12th Grade

19 questions
Ohms law
Presentation
•
9th - 12th Grade

21 questions
3.6 Notes: Electron Orbital Diagrams
Presentation
•
9th - 12th Grade

20 questions
Ch. 7.1 - Ions
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade