

MVHS Chemistry Semester 1 Review Day 1
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+4
Standards-aligned
Nicholas Carpenter
Used 1+ times
FREE Resource
22 Slides • 40 Questions
1
MVHS Chemistry Semester 1 Review
Day 1

2
Topics that will be covered on the final
Properties of Matter - Chemical and Physical Properties and Changes - Day 1
Atomic Theory - Composition of atoms, subatomic particles, ions and isotopes, Electron Configurations - Day 1
Periodic Table - Trends and Organization - Day 1
Chemical Reactions - Signs of Reactions, Balancing Equations, Types of Reactions, Predicting Products - Day 2
Bonding - Types of Bonds, Balancing Charges, IMFs, Writing Chemical Names and Formulas - Day 1 and 2
3
Properties of Matter 1
Properties are physical or chemical
Physical Properties can be observed without changing the chemical composition of a substance
Physical properties include mass, volume, density, color, luster, texture, melting point, boiling point, solubility, shape
Chemical Properties can only be determined by changing the composition of the substance (the substance has to undergo a chemical reaction)
Chemical properties include flammability, oxidation numbers, and pH
4
Properties of Matter 2
Mass: the amount of matter in a given object (protons and neutrons); standard unit is kg, but we usually use grams in Chemistry
Volume: The amount of space an object takes up; standard unit is L, but we often use mL (liquids) or cm3 (solids) (1 mL = 1 cm3);
Density: The amount of matter within a given volume; measured in g/mL or g/cm3 (those mean the same thing,
D=Vm ; m=D⋅V ; V=Dm
D: Density; m: mass; V: volume
volume of irregular solids (like rocks) can be measured via water displacement - place in a graduated cylinder of water and measure the volume change
5
Multiple Choice
6
Multiple Choice
Bobbie has a paper clip. It has a mass of 9g and a volume of 3cm3. What is its density?
3 g/cm3
1/3 g/cm3
27 g/cm3
39 g/cm3
7
Multiple Choice
Amos has an eraser. It has a mass of 4 g, and a volume of 2 cm3. What is its density?
8 g/cm3
2 g/cm3
1/2 g/cm3
24 g/cm3
8
Multiple Choice
Mei has a rock. The rock has a mass of 14 g and a volume of 2 cm3. What is the density of the rock?
7 mL
7 g/cm3
28 g/cm3
1/7 g/cm3
9
Multiple Choice

10
Multiple Choice
11
Multiple Choice
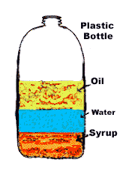
12
Properties of Matter 3
Physical changes can occur without changing the composition - examples include breaking ice, phase changes (melting or boiling)
Chemical changes involve reactions - combustion, neutralizing acids or bases
13
Atomic Theory 1
Composition of atoms: nucleus in center, made of protons (positive charge) and neutrons (no charge); electron cloud around nucleus contains electrons (negative charge)
In neutral atoms, np (# of protons) = ne (# of electrons)
Ions have an unbalanced number of protons and electrons
Cations have more protons than electrons, so they have a positive charge
Anions have more electrons than protons, so they have a negative charge
Calculate charge (q): q = np - ne
14
Multiple Choice
If an atom loses electrons, the charge will be positive.
true
false
15
Multiple Choice
Which types of elements form anions?
metals
nonmetals
metalloids
noble gases
16
Multiple Choice
Which types of elements form cations?
metals
nonmetals
metalloids
noble gases
17
Atomic Theory 2
Electrons are situated around the nucleus in energy levels (also called shells)
Outermost shell is called the valence shell
Valence electrons determine element's chemical properties
Valence shells correspond to rows in the periodic table - if the outermost electrons are in the 3rd shell, the element is in the 3rd row of the periodic table
Shells are subdivided into subshells (or sublevels)
s, p, d, f subshells
subshells are subdivided into orbitals, which can hold 2 electrons each
18
Atomic Theory 3
Electrons have a property known as "spin"
Electrons within the same orbital must have opposite spins (up or down);
s subshell has 1 orbital (2 electrons); p has 3 orbitals (6 electrons); d has 5 orbitals (10 electrons); f has 7 orbitals (14 electrons)
Each orbital in a subshell must have 1 electron in it before any can have 2 (no one gets seconds until everyone has gotten 1)
Elements can be identified by their electron configuration, which tells us where (in the shells and subshells) their electrons are located
Shells and subshells are filled in the same order for most elements, it's just a matter of how many electrons there are to fill the available orbitals
19
Atomic Theory 4 - Electron Configurations
To write an electron configuration, you must find a home for all of the electrons
The coefficient (big number) tells you the shell or energy level; the letter tells you the subshell, and the superscript (little number/exponent) tells you how many electrons are in the subshell (the superscripts in the image tell you how many each subshell can hold, not how many there are - you determine that)

20
Atomic Theory 5 - Electron Configs pt 2
Start at the top of the blue arrows and follow them down; once you reach the end of one arrow, move to the top of the next
Ex: an element has 9 electrons, it's configuration would be 1s2 2s2 2p5
The last number is 5 instead of 6 because we only had 5 electrons left after filling 1s and 2s. Add the superscripts to count the number of electrons

21
Atomic Theory 6 - Electron Configs pt 3
Energy levels in d block are 1 less than s and p; f block energy levels are 2 less
ex: Selenium config is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p4
Valence electrons correspond with columns in periodic table (at least for s and p blocks) - Groups 1 and 2 have 1 and 2 valence electrons, Groups 13, 14, 15, 16, 17, and 18 have 3, 4, 5, 6, 7, and 8, respectively

22

Final terms in the electron configuration of each element
23
Multiple Choice
Each row on the periodic table represents:
an energy level
a sublevel
an electron
an orbital
24
Multiple Choice

25
Multiple Choice

1s22s22p63s23p64s23d10
26
Multiple Choice

27
Multiple Choice
28
Multiple Choice

How many electrons can the first energy level hold?
1
2
8
0
29
Atomic Theory 7 - Isotopes
Atoms of the same element that have different numbers of neutrons are called isotopes
Different isotopes have different mass numbers (number of protons + number of neutrons
Isotopes are identified by that mass number (ex: Carbon-12 and Carbon-14)
30
Atomic Theory 8 - Isotopes
Different isotopes are present in samples and in the universe in different amounts (abundances)
The average mass of all of the isotopes of a given element is the atomic mass that you see on the periodic table
Example calculation on the next slide

31
Atomic Theory 9 - Isotopes
Important - when using % abundance in calculation, you must convert the percentage to a decimal by dividing by 100
ex: 78.99% becomes 0.7899
Multiply the resulting decimals by the masses of the corresponding isotopes and add them all together to get the average atomic mass

32
Multiple Choice

33
Multiple Choice

34
Multiple Choice
The atomic mass of an element is the ___.
average of the mass number and the atomic number for the element
weighted average of the masses of the isotopes of the element
total mass of the isotopes of the element
total number of subatomic particles in the nucleus
35
Multiple Choice
36
Multiple Choice
What is the average mass of this element?
37
Periodic Table 1
Periodic Table is arranged in order of ascending atomic number (number of protons)
Columns are called Groups; Rows are called Periods
Groups are elements that have similar properties (same number of valence electrons)
Periods have the same number of energy levels/shells of electrons
38

39
Periodic Table 2 - Trends
Trends are electronegativity, atomic radius, ionization energy, reactivity
Electronegativity: a measure of an atom's ability to attract electrons
atomic radius: the distance from the center of the nucleus to the valence electrons
ionization energy: the amount of energy required to remove an electron
Reactivity: how readily an atom will react with other atoms
40
Periodic Table 3 - Trends Cont.
Electronegativity increases up and to the right
Atomic Radius increases down and to the left
Ionization energy increases up and to the right
metal reactivity increases down and to the left - with atomic radius
nonmetal reactivity increases up and to the right - with electronegativity
41
Multiple Choice
42
Multiple Choice
43
Multiple Choice
44
Multiple Choice
45
Multiple Choice
46
Multiple Choice
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice

50
Multiple Choice
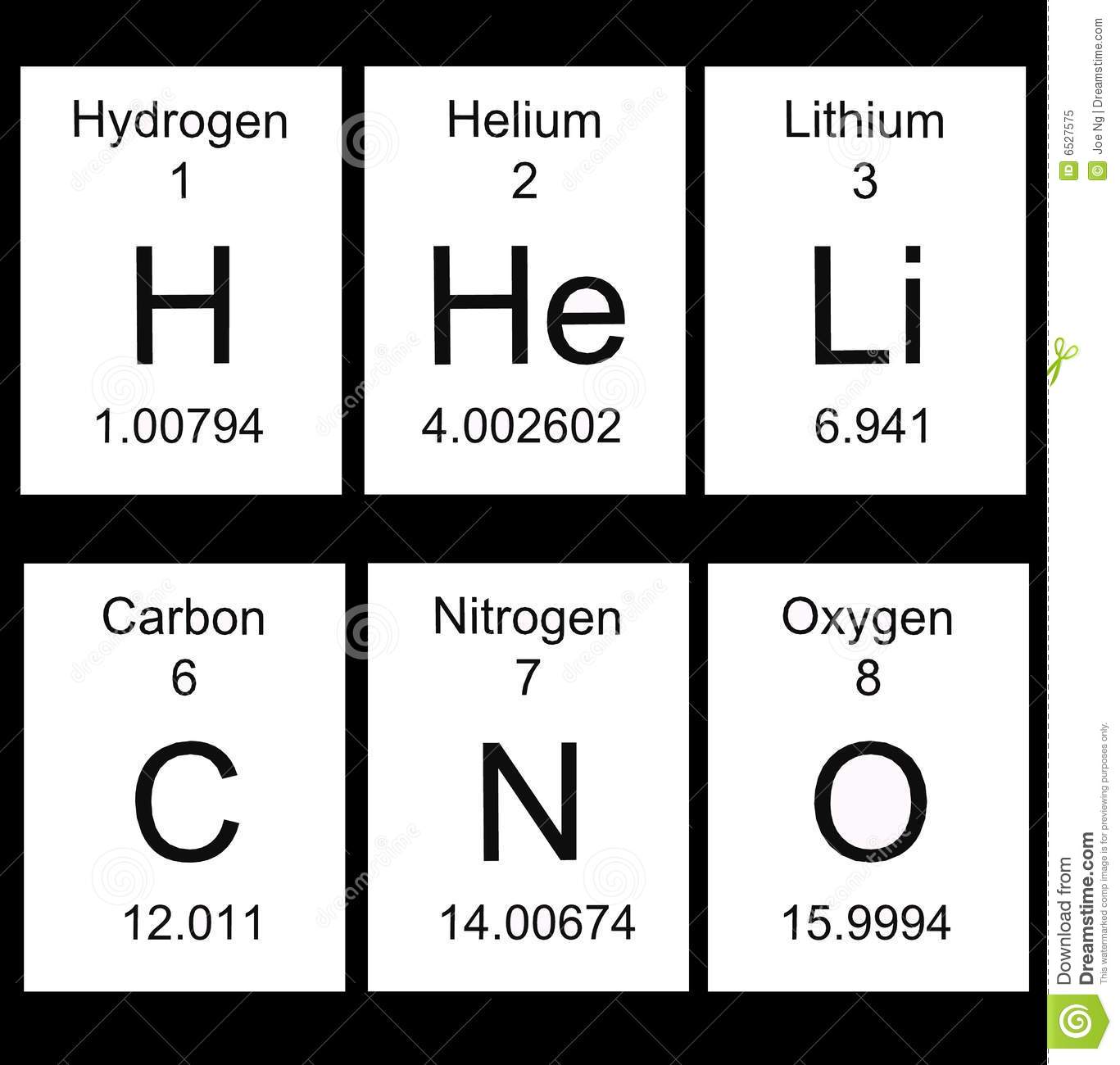
51
Multiple Choice
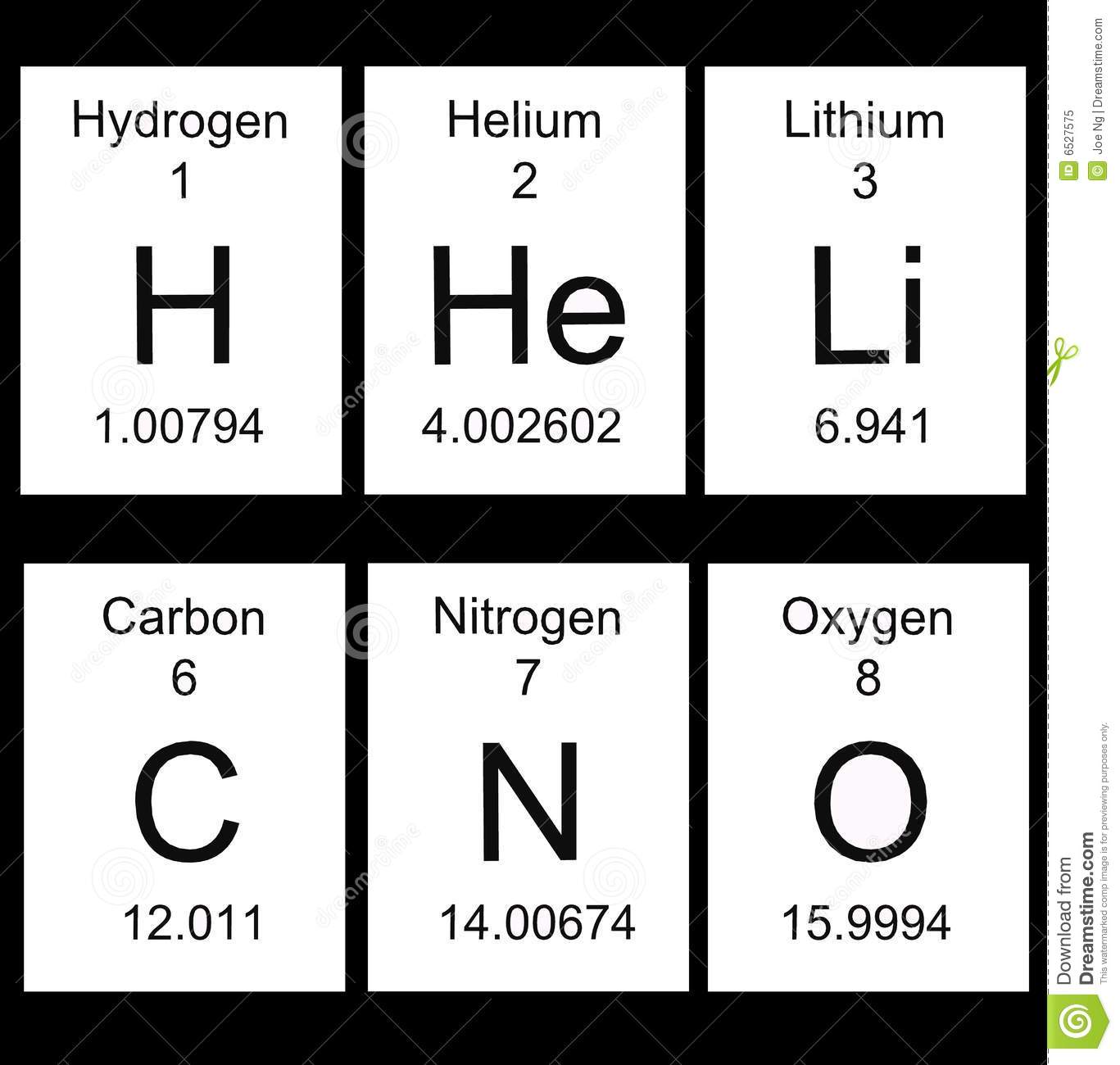
52
Multiple Choice

Elements in the same group or column have the same ...
# of valence electrons
# of shells
# of protons
Mass Number
53
Multiple Choice
What happens to the number of shells as you move down a group?
They decrease by 1
They increase by 1
Nothing
54
Bonding 1 - Types of Bonds
Ionic bond - one atom (a nonmetal) takes the valence electrons of another atom (or atoms) to become an anion and makes the other atom(s) become a cation; these oppositely charged atoms now attract each other and create an ionic compound
There are also polyatomic ions - ions with multiple atoms that are covalently bonded together; these can interact with individual ions or other polyatomic ions
Covalent Bonds: when two or more nonmetals share valence electrons
Metallic Bond: when metals bond with each other creating a "sea of electrons;" valence electrons move freely between atoms
55
Bonding 2 - Properties
Ionic compounds are brittle and have high melting points
Covalent compounds have lower melting points and are not as brittle; some are liquid or even gas at room temperature
Metallically bonded substances (alloys) are ductile (can be pulled into wires) and malleable (can be hammered into shapes) because the "sea of electrons" allows atoms to slide past each other
56
Bonding 3 - IMFs
Polarity: when one atom in a covalent compound has a higher electronegativity than another, it will pull the shared valence electrons closer to it, creating areas of partial positive and negative charges
Dipole: a molecule with a partial positive end and a partial negative end
Hydrogen Bonding: a special type of dipole-dipole attraction featuring hydrogen bonded with high electronegativity elements (N, O, F)
London Dispersion: "temporary dipoles" created by valence electrons in two atoms repelling each other
Relative IMF strength: hydrogen bonding > dipole dipole > London Dispersion
57
Multiple Choice
58
Multiple Choice
Which one has higher melting point? CO2 or H2O
CO2
H2O
59
Multiple Choice
The weaker the intermolecular forces of a substance the _____________ the boiling point
higher
lower
60
Multiple Choice
Which substance has the weakest intermolecular forces?
Substance A, boiling point of 75 °C
Substance B, boiling point of 105 °C
Substance C, boiling point of 25 °C
Substance d, boiling point of 45 °C
61
Multiple Choice
Water has an unusually high boiling point for a molecular compound because it has
hydrogen bonding
ion-ion attractions
a high density
a large gram formula mass
62
Multiple Choice
Which sample has hydrogen bonding?
H2S
CH4
NH3
HI
MVHS Chemistry Semester 1 Review
Day 1

Show answer
Auto Play
Slide 1 / 62
SLIDE
Similar Resources on Wayground

56 questions
Electron Configuration
Presentation
•
10th - 12th Grade

56 questions
Physics Unit 1: Kinematics in One Dimension and Graphing Motion
Presentation
•
9th - 12th Grade

59 questions
Matter and Energy
Presentation
•
9th - 11th Grade

57 questions
Benchmark 4 review day 1 (Unit 10 thermo,K&E)
Presentation
•
10th Grade - University

55 questions
El preterito
Presentation
•
9th - 12th Grade

55 questions
Periodic Table and Periodic Trends (Pre-AP Chem)
Presentation
•
9th - 12th Grade

55 questions
Periodic Table Trends
Presentation
•
9th - 12th Grade

60 questions
Conductivity of Solutions CER
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

36 questions
6th Grade Math STAAR Review
Quiz
•
6th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

10 questions
Exploring Properties and Models of Acids and Bases
Interactive video
•
6th - 10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

16 questions
AP Chemistry Review: Unit 9
Quiz
•
9th - 12th Grade

11 questions
Balancing Chemical Equations
Presentation
•
9th Grade

12 questions
IP: Neutralization Reactions
Quiz
•
10th - 11th Grade

28 questions
Acids and Bases Test Review
Quiz
•
10th Grade