

8.5BC Reteach
Presentation
•
Science
•
8th - 10th Grade
•
Practice Problem
•
Medium
Ciara West
Used 9+ times
FREE Resource
18 Slides • 37 Questions
1
What you need to know about PROTONS
1. They have a positive charge!
2. They are located inside the nucleus!
3. They add mass to the atom!
4. They are equal to your atomic number! PROTON=IDENTITY!

2
What you need to know about PROTONS
Remember protons are equal to the atomic number, so that means if the atomic number here is 3, we should have 3 protons!

3
Multiple Choice

How many protons are in Gold
79
197
118
276
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice

Sodium has an atomic number of 11, how many protons does this atom have?
22
11
12
2
7
What you need to know about NEUTRONS
1. They have no charge or a neutral charge
2. They are located inside the nucleus!
3. They add mass to the atom!
4. Mass-Atomic #= Neutrons!

8
What you need to know about NEUTRONS
Remember the formula to find NEUTRONS is MASS - ATOMIC #=NEUTRONS, So Lithium has a mass that rounds to 7, minus the atomic number 3, so you'd have 4 neutrons!

9
Multiple Choice

10
Multiple Choice
11
Multiple Choice
12
What you need to know about ELECTRONS
Remember electrons are equal to protons, so that means if the atomic number here is 3, we should have 3 protons and 3 electrons!

13
Multiple Choice
14
THE PERIODIC TABLE
Groups, Periods, Valence Electrons, Energy Levels, Metals, Nonmetals, Metalloids
15
What you need to know about GROUPS
1. SAME GROUP SAME CHEMICAL/PHYSICAL PROPERTIES!
2. Groups go from 1-18.
3. Groups go UP and DOWN
4. Groups tell the number of valence electrons (#A)

16
What you need to know about GROUPS
Remember, groups tell you the number of VALENCE ELECTRONS! Just look for the number with an A next to it! So group 13, has 3 Valence Electrons!

17
Multiple Choice
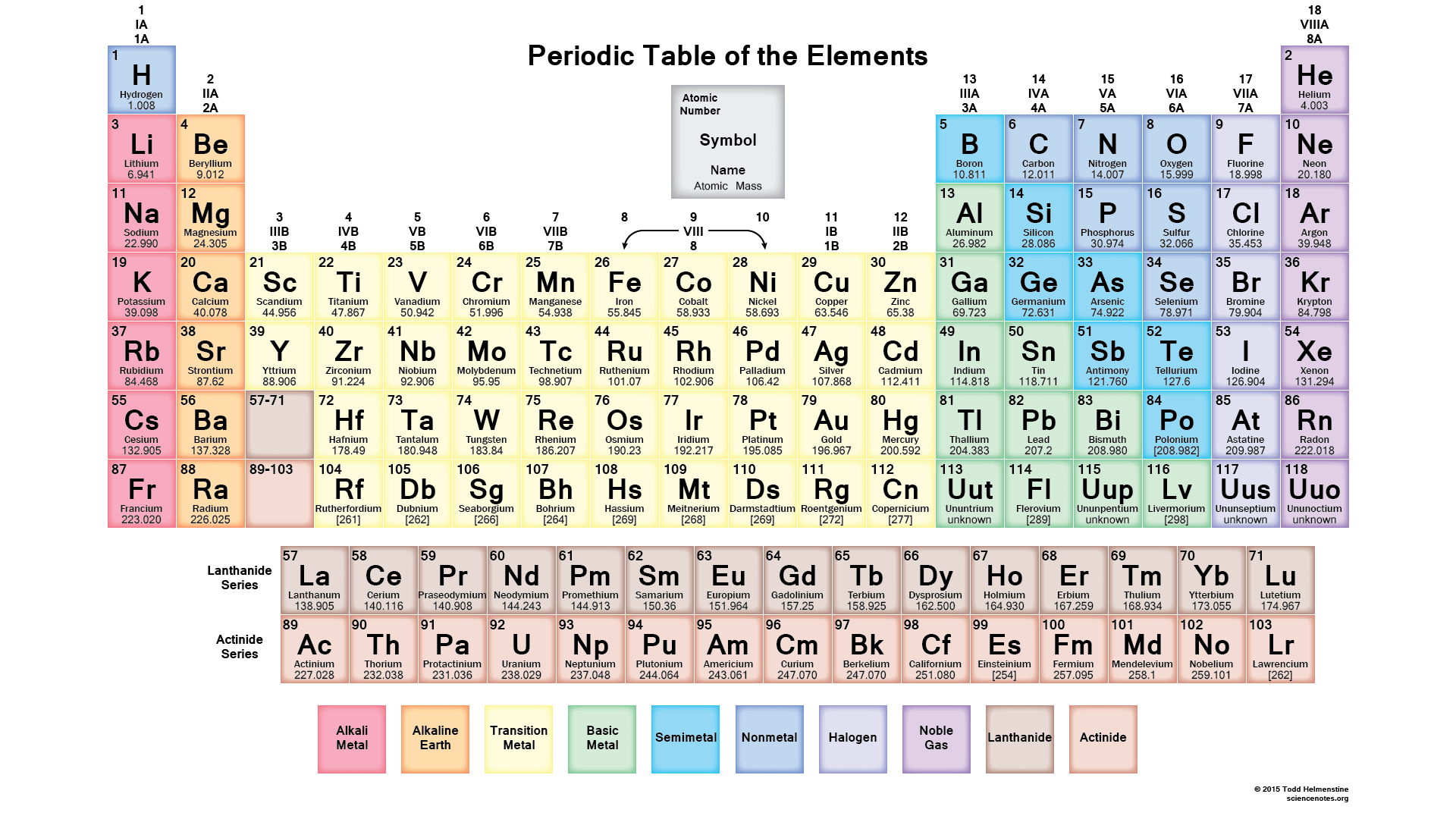
18
Multiple Choice

What are the vertical columns on the periodic called?
groups
periods
nonmetals
metals
metalloids
19
Multiple Choice
20
Multiple Choice

Beryllium is in group number______.
Group 2
Group 3
Group 4
Group 7
21
Multiple Choice

How many valence electrons are in group 16?
16
6
1
22
What you need to know about PERIODS
1. Periods are in rows that go across (side to side)
2. Periods go from 1-7.
3. Periods tell us the number of energy levels

23
What you need to know about PERIODS
Remember, periods tell you the number of energy levels! So period 4, has 4 energy levels!

24
Multiple Choice

energy levels
valence electrons
protons
neutrons
25
Multiple Choice

26
Multiple Choice
Which of these elements can be found in period 2?
Oxygen, O
Sulfur, S
Selenium, Se
Tellurium, Te
27
Multiple Choice
How many energy levels are in period 2?
12
2
22
28
Multiple Choice
Which of these elements can be found in period 4?
Oxygen, O
Sulfur, S
Selenium, Se
Tellurium, Te
29
What you need to know about VALENCE ELECTRONS
1. These are electrons on the OUTSIDE energy level!
2. Valence electrons determine REACTIVITY!
3. VALENCE ELECTRONS GO FROM 1-8
4. Look for the #A to see the number of Valence Electrons! (#A)

30
What you need to know about VALENCE ELECTRONS
5. Elements with 1 or 7 Valence electrons are HIGHLY reactive/ unstable.
6. Elements with 8 Valence electrons are stable/nonreactive.

31
What you need to know about VALENCE ELECTRONS
Remember, valence electrons are on the outside energy level only. AND the # with an A next to it also tells you how many VE there are! So for Carbon, you can count or look at the #A to see it has 4 valence electrons!

32
Multiple Choice
All atoms are most stable with (or would "prefer") how many electrons in their valence shell?
1
2
8
18
33
Multiple Choice
What are valence electrons?
The total number of electrons in an atom
The number of electrons in the outermost shell
The number of electrons in the second shell
The number of protons in the outermost shell
34
Multiple Choice

How many valence electrons does Hydrogen Have?
1 Valence electron
2 Valence electron
3 Valence electron
4 Valence electron
5 Valence electron
35
Multiple Choice

Which two groups on the Periodic Table are the MOST reactive?
Group 1 and Group 17
Group 2 and Group 16
Group 13 and Group 15
Group 17 and Group 18
36
Multiple Choice

Which two groups on the Periodic Table are the MOST reactive?
Group 1 and Group 17
Group 2 and Group 16
Group 13 and Group 15
Group 17 and Group 18
37
What you need to know about ENERGY LEVELS
1. They are the number of shells or circles around the nucleus.
2. They are equal to your periods!

38
What you need to know about ENERGY LEVELS
Remember, it's just the number of circles around the nucleus OR look at the period! As you can see, Sodium should have 3 energy levels!

39
Multiple Choice
Cd and Te have the same number of energy levels.
True
False
40
Multiple Choice
P and Po have the same number of energy levels.
True
False
41
Fill in the Blanks
Type answer...
42
Multiple Choice
The period number tells you the number of
protons
energy levels
valence electrons
43
Fill in the Blanks
Type answer...
44
What you need to know about METALS, NONMETALS, & METALLOIDS
1. Metals are on the left/middle.
2. Nonmetals on are the right.
3. Metalloids are along the staircase.

45
What you need to know about METALS, NONMETALS, & METALLOIDS
Remember these facts about metals, nonmetals, and metalloids!

46
Multiple Choice
47
Multiple Choice
48
Multiple Choice

49
Multiple Choice
Which of these grouping of elements could have the characteristic of luster (shiny)?
Metal
Nonmetal
metalloids
Both metals and metalloids
50
Multiple Choice
Which of these was NOT a conductor?
sulfur
magnesium
zinc
aluminum
51
Let's see what you learned!
Try out these more difficult questions!
52
Multiple Choice

What property does element X have based on its location in the Periodic Table?
Highly reactive nonmetal
Manmade radioactive substance
Nonreactive gas
Metallic solid
53
Multiple Choice
Which of the following best describes the location of the element tin?
Group 4(4B), Period 4
Group 16(6A), Period 5
Group 14 (4A), Period 5
Group 13(3A), Period 6
54
Multiple Choice

A student is shown a picture of the atom below. What element, based on the subatomic particle in the atom, is shown?
Beryllium
Helium
Lithium
Oxygen
55
Multiple Choice
Which of the following atoms is the most reactive?
What you need to know about PROTONS
1. They have a positive charge!
2. They are located inside the nucleus!
3. They add mass to the atom!
4. They are equal to your atomic number! PROTON=IDENTITY!

Show answer
Auto Play
Slide 1 / 55
SLIDE
Similar Resources on Wayground

53 questions
EM Spectrum
Presentation
•
8th Grade

49 questions
Contact and Noncontact Forces
Presentation
•
6th - 9th Grade

51 questions
Topic 3.3 - Electrical Circuits
Presentation
•
9th Grade

50 questions
Saturn
Presentation
•
9th Grade

49 questions
Introduction to Forces
Presentation
•
9th Grade

46 questions
Lesson 2 Properties of Matter
Presentation
•
8th - 9th Grade

49 questions
Unit 2 Cell Division R
Presentation
•
7th - 9th Grade

49 questions
Unit 2: Functions Review
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade

46 questions
Unit 4 Geosphere Test Review
Quiz
•
9th - 12th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade