

Lesson - 4th 6th Wks
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+3
Standards-aligned
Kyle Pearson
Used 20+ times
FREE Resource
7 Slides • 39 Questions
1
4th 6th Weeks - Final
Empirical and Molecular Formulas
Stoichiometry
Limiting Reactant
Percent Yield
Kinetic Molecular Theory
Pressure
Boyle's Law
Charles' Law
Avogadro's Law
Ideal Gas Law
Dalton's Law

2
Empirical & Molecular Formulas
Empirical Formulas are the reduced/simplest ratio of atoms
Molecular Formulas are the actual ratio of atoms (not reduced)
REMINDER- chemical formulas can be both empirical and molecular
example - water and carbon dioxide
3
Multiple Choice
A molecule of carbon, hydrogen, and oxygen have an empirical formula C5H8O2
What is the molecular formula if the molar mass is about 200g?
C5H10O2
C10H16O4
C2H4O
4
Multiple Select
Find all of the formulas that would be considered in their molecular formula.
H2O
C6H12O6
MgCl2
CO2
5
Fill in the Blanks
Type answer...
6
Multiple Select
Click on all of the compounds that would be in their empirical formula form?
Ca2S2
H2O
NaCl
C6H12O6
7
Stoichiometry
Stoichiometry is used to help chemists determine how much reactant and product is used in the reaction.
MUST have a BALANCED EQUATION
Coefficients from the balanced equation gives you mole ratios
grams to moles conversion factor is found on the Periodic Table
moles to particles conversion factor 1 mole = 6.02e23
moles to volume conversion factor is 1 mole = 22.4 L
8
Fill in the Blanks
Type answer...
9
Fill in the Blanks
Type answer...
10
Limiting Reactants Concept
the reactant that runs out first
the reagent that stops the reaction
the reactant that is completely consumed
the reagent that limits the amount of product that can be made
11
Multiple Select
Click all the explanations that talk about the limiting reactant or reagent?
the reactant that will run out and limit the amount of product you can make
the reactant that you have excess of when making the product
the reagent that stops the reaction
the limiting reactant (or limiting reagent) is the reactant that gets consumed first in a chemical reaction
12
Multiple Choice
2Al + 3I2 ------> 2AlI3
Which reagent will be completely consumed first if you have 10 moles of each reagent?
aluminum
iodine gas
aluminum iodide
13
Percent Yield
Actual Yield / Theoretical Yield x 100 = Percent Yield
14
Multiple Choice
What side of the arrow do you find any yield (actual or theoretical) in a balanced chemical equation?
where ever the question puts it
on the reactant side
on the product side
can be on either side
15
Multiple Choice
Which yield will be the smallest most of the time?
actual yield
theoretical yield
percent yield
16
Fill in the Blanks
Type answer...
17
Fill in the Blanks
Type answer...
18
Fill in the Blanks
Type answer...
19
Kinetic Molecular Theory
How do gasses behave?
20
Multiple Choice
Gas pressure is created by
gas molecules hitting other gas molecules
molecules hitting other molecules
21
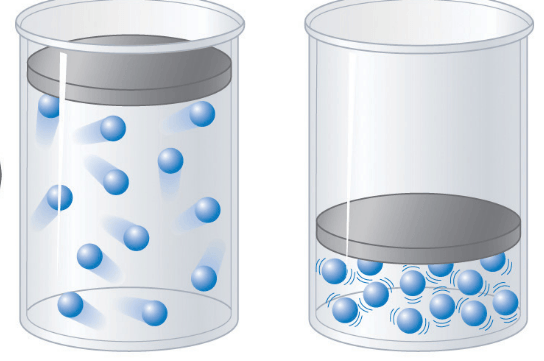
Multiple Choice

Flask 1 contains the most molecules
Flask 2 contains the most molecules
Flask 3 contains the most molecules
Flask 4 contains the most molecules
22
Multiple Select

23
Multiple Choice
A hypothetical gas that perfectly fits all the assumptions of the kinetic molecular theory is known as
real gas
ideal gas
imaginary gas
perfect gas
24
Multiple Select
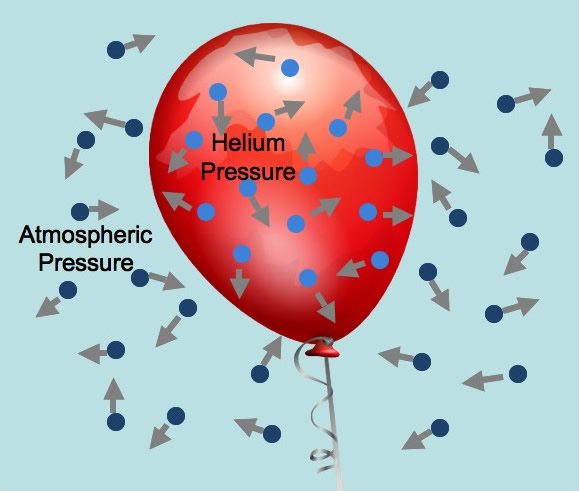
The particles hitting each other
the particles hitting the INSIDE walls of the balloon
temperature
volume
the particles hitting the OUTSIDE of the balloon
25
Gas Laws

26
Multiple Choice
27
Multiple Choice
Boyle's Law says when PRESSURE increases then the VOLUME must...
28
Multiple Choice
29
Multiple Choice
Gases can be compressed.
30
Multiple Choice
31
Multiple Choice
What is the formula for Charles' Law?
V1 /T1=T2 /V2
T1V1/T2V2
V1V2=P2V1
V1/T1=V2/T2
32
Multiple Select
What gas laws must you use Kelvin temperature in or your calculations will be wrong?
Boyle's Law
Charles' Law
Avogadro's Law
Ideal Gas Law
Pressure
33
Multiple Choice
When the temperature of a gas decreases (with pressure and amount held constant), does the volume will ....
be unaltered
remains constant
Increases
decreases
34
Multiple Choice
Which of the following uses the concept of Charles’ Law?
Hot air balloon
Syringe
Drinking straw
35
Multiple Choice
36
Multiple Choice
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
40
Multiple Select
A 28.965 L sample of Kr experiences a change in pressure from 358.34 kPa to 729.71 kPa. The resulting volume is 14.224.
Name the law being used and is the data following the law?
yes
no
Boyle's Law
Charles' Law
Ideal Gas Law
41
Multiple Choice
Which is a correct unit for R?
L.atm/mol.k
L.atm
mmHg/mol.K
L.atom/mol
42
Multiple Choice
when pressure increases
volume decreases
when temperature increases
volume increases
when the moles of a gas decrease
volume decreases
43
Multiple Select
If 3.25 mol of Ar occupies 100. L at a particular temp and pressure.
2.89 moles of Ar occupy 435 liters under same condition.
Define the law and is the law followed based on the data?
yes
no
Charles' Law
Boyle's Law
Avogadro's Law
44
Multiple Select
A mole of any substance is equal to ____.
6.02e23
6.02 x 1023
6.02 x 10-22
6.02 x 10-23
45
Multiple Choice

What would a closed bag of chips do in outer space?
explode
implode
nothing
46
Multiple Select
When using the Ideal Gas law, what units CANNOT be used in your calculations when using the variable "R" constant 0.0821?
atmospheric pressure
(atm)
torr
mmHg
psi
Celsius
(C)
kiloPascals
(kPa)
Kelvin
(K)
4th 6th Weeks - Final
Empirical and Molecular Formulas
Stoichiometry
Limiting Reactant
Percent Yield
Kinetic Molecular Theory
Pressure
Boyle's Law
Charles' Law
Avogadro's Law
Ideal Gas Law
Dalton's Law

Show answer
Auto Play
Slide 1 / 46
SLIDE
Similar Resources on Wayground

41 questions
Covalent Bonding
Presentation
•
8th - 12th Grade

44 questions
Introduction to Moles
Presentation
•
9th - 12th Grade

41 questions
Lewis Dot Structures
Presentation
•
9th - 12th Grade

42 questions
Chapter 13 Vocab Review - Thermochemistry
Presentation
•
10th - 12th Grade

41 questions
Arrhenius & Bronsted Lowry Acids & Bases
Presentation
•
9th - 12th Grade

43 questions
4/22 Solutions in Chemistry
Presentation
•
9th - 12th Grade

40 questions
Mixtures, Solutions, and Chemical Equations
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade