

Chapter 13 Vocab Review - Thermochemistry
Presentation
•
Chemistry
•
10th - 12th Grade
•
Easy
+3
Standards-aligned
D'Anne Cribb
Used 16+ times
FREE Resource
1 Slide • 41 Questions
1
Thermochemistry Chapter 13 Vocab Review

2
Multiple Choice
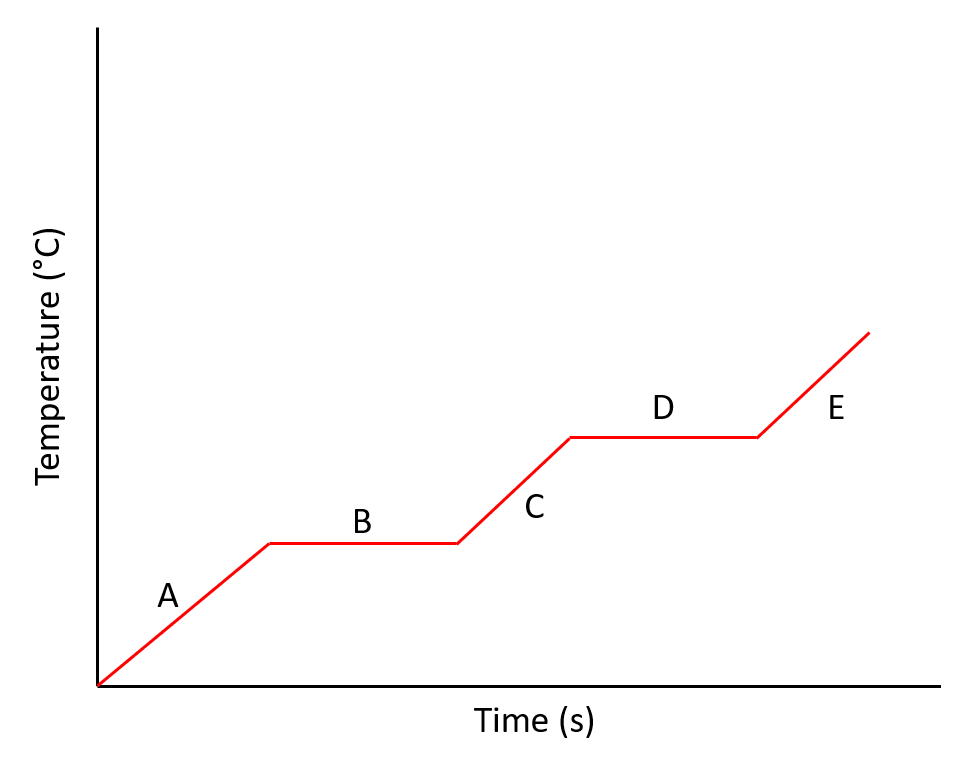
Why are lines B and D flat?
The ice is melting
The ice is freezing
All of the thermal energy is used to break the bonds
They stopped the stopwatch
They stopped heating the ice.
3
Multiple Choice

The diagram shows a heating curve for ice. Which line represents the ice melting?
A
B
C
D
E
4
Multiple Select
Select the state changes that latent heat of vaporization applies to.
melting
evaporating
freezing
condensing
sublimation
5
Multiple Select
Select the state changes that latent heat of fusion applies to.
melting
evaporating
freezing
condensing
sublimation
6
Multiple Choice

7
Multiple Choice

8
Multiple Choice
True or false: melting and freezing occur at the same temperature!
True
False
I don't know
9
Multiple Choice
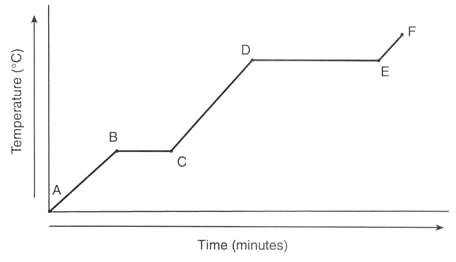
10
Multiple Choice

11
Multiple Choice
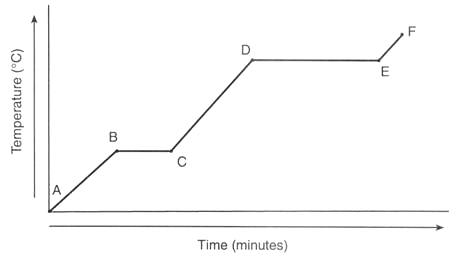
12
Multiple Choice

13
Multiple Choice

14
Multiple Choice

15
Multiple Choice

16
Multiple Choice
For an exothermic reaction, the heat is on the ____ side
reactant
product
either side
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
Durning a phase change the temperature of a substance_________.
always increases
always decreases
always stays the same
varies
20
Multiple Choice
What does temperature measure?
heat
average kinetic energy
space
time
21
Multiple Choice

Convert 42 kJ into joules
0.042 J
175.73 J
42000 J
10.04 J
22
Multiple Choice
What is the study of heat changes that accompany chemical reactions and phase changes?
energy
chemistry
work
thermochemistry
23
Multiple Choice
A cast iron skillet is used to fry bacon. For optimal frying, the pan must be heated to about 178 oC from a room temperature of 22.0 oC. It is known that 1.58 x 105 J of heat energy are absorbed by the pan to reach the desired temperature and the specific heat of iron is 0.450 J/g oC. What must the mass of the skillet be?
12.7 kg
2.25 kg
110 kg
1.97 kg
24
Multiple Choice

The graph represents the uniform heating of a substance, starting with the substance as a solid below its melting point. Which line segment listed below represents an increase in potential energy and no change in average kinetic energy?
Line segment AB
Line segment BC
Line segment CD
Line segment EF
25
Multiple Choice

The potential energy diagram of a reaction is shown. Which statement below is correct relating to this reaction?
#1 represents the enthalpy change for this exothermic reaction.
#2 represents the enthalpy change for this endothermic reaction.
#3 represents the enthalpy change for this endothermic reaction.
#4 represents the enthalpy change for this exothermic reaction.
26
Multiple Choice

27
Multiple Choice
Is this reaction endothermic or exothermic?
28
Multiple Choice

29
Multiple Choice
30
Multiple Choice
Energy cannot be created or destroyed; it can only be transferred refers to the....
31
Multiple Choice
32
Multiple Choice
A chemical reaction that absorbs heat from the surroundings is said to be __________ and has a __________ ΔH at constant pressure.
endothermic; positive
endothermic; negative
exothermic; negative
exothermic; positive
33
Multiple Choice
34
Multiple Choice
The transfer of thermal energy between objects of different temperatures is called...
temperature
heat
internal energy
none of these
35
Multiple Choice
36
Multiple Choice
H2 + Cl2 --> 2 HCl + 1845 kJ
Is this reaction endothermic or exothermic?
Endothermic
Exothermic
37
Multiple Choice
During an endothermic reaction in a beaker if we are part of the surroundings and touched the beaker, it would feel _______.
Warm
Cold
Bubbly
Damp
38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
Is the combustion of gasoline endothermic or exothermic?
Endothermic
Exothermic
41
Multiple Choice
42
Multiple Choice
In an exothermic process the surrounding looses heat.
True
False
Thermochemistry Chapter 13 Vocab Review

Show answer
Auto Play
Slide 1 / 42
SLIDE
Similar Resources on Wayground

38 questions
Intro to Bonds
Presentation
•
9th - 11th Grade

37 questions
Punctuation: Colons and Semicolons
Presentation
•
9th - 12th Grade

35 questions
Acids and Bases
Presentation
•
10th - 12th Grade

37 questions
Balancing Chemical Equations
Presentation
•
9th - 12th Grade

35 questions
Solubility
Presentation
•
9th - 12th Grade

40 questions
Chemical and Physical changes
Presentation
•
10th - 12th Grade

36 questions
Chapter 8: Kinetics & Equilibrium LESSON
Presentation
•
10th - 12th Grade

33 questions
Quizizz Lesson Unit 6 Lesson 4: Periodic Trends
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

120 questions
2nd Semester Final Review
Quiz
•
11th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade