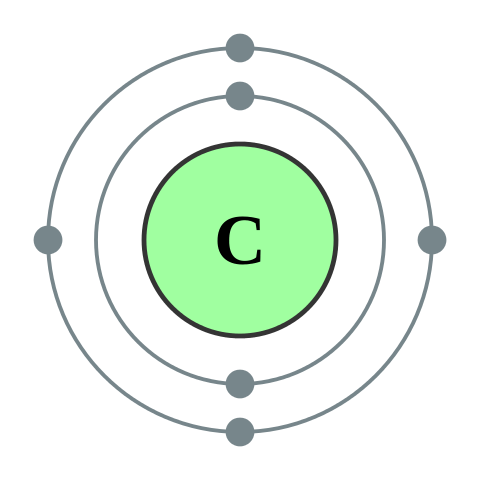
Chemical Formulas and Bonding Review
Presentation
•
Chemistry
•
8th Grade
•
Practice Problem
•
Medium
Standards-aligned
Michael Stange
Used 19+ times
FREE Resource
8 Slides • 12 Questions
1
2
3
4
Multiple Select
5
Fill in the Blank
6
7
Fill in the Blank
8
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
16
17
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

15 questions
Colonial Protest, Boston Massacre, Boston Tea Party
Lesson
•
8th Grade

15 questions
Verb Moods- Review
Lesson
•
8th Grade

15 questions
Microbiology Review Lesson
Lesson
•
8th Grade

15 questions
Solving Multi-Step Equations Practice
Lesson
•
8th Grade

15 questions
The Partitive and Expressions of Quantity in French
Lesson
•
8th Grade

15 questions
Physical/Chemical Changes
Lesson
•
8th Grade

15 questions
Line of Best Fit
Lesson
•
8th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

29 questions
Alg. 1 Section 5.1 Coordinate Plane
Quiz
•
9th Grade

22 questions
fractions
Quiz
•
3rd Grade

11 questions
FOREST Effective communication
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade

10 questions
Identifying Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Counting Atoms
Quiz
•
8th Grade

10 questions
Exploring Acids, Bases, and the pH Scale
Interactive video
•
6th - 10th Grade

20 questions
Periodic Table Basics
Quiz
•
8th Grade

15 questions
Elements, Compounds, and Mixtures Review
Quiz
•
8th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade