

The Periodic Table
Presentation
•
Chemistry
•
11th - 12th Grade
•
Medium
Hector Mendoza-Arias
Used 4+ times
FREE Resource
25 Slides • 18 Questions
1
The Periodic Table
Mr. Mendoza

2

3
Dmitri Mendeleev (1869)
Mendeleev(Russinan Chemist) and Lothar Meyer (Germany) published nearly identical classification schemes for elements known to date. (The Periodic Table)

4

Dont forget about me!!
5
The Periodic Table
Based on the similarity of properties and reactivities exhibited by certain elements.

6
Multiple Choice
In Mendeleev's periodic table, elements in each column had similar
atomic masses.
properties.
atomic numbers.
symbols.
7
Henri Moseley (England,1887-1915)
established that each elements has a unique atomic number, which is how the current periodic table is organized.

8
Multiple Choice
Which Scienticst established that each elements has a unique atomic number, which is how the current periodic table is organized.
Mendeelv
Moseley
Meyer
Rutherford
9
NOW!
A map of the building block of matter.

10
Down the Periodic Table
Group/Family: Are arranged vertically down the periodic table (columns or group, 1- 18 or 1-8 A,B)
These elements have the same number of electrons in the outer most shells, the valence shell.

11
Across the Periodic Table
Periods: Are arranged horizontally across the periodic table (rows 1-7)
These elements have the same number of shells or energy levels.

12
Multiple Choice
The order of elements in the modern periodic table is based on an element's
atomic number.
name.
chemical symbol.
atomic mass.
13
Multiple Choice
Each column of the periodic table is
an element.
a group.
an isotope.
a period.
14
Multiple Choice
Atoms of elements that are in the same group have the same number of
protons.
neutrons.
valence electrons.
protons and neutrons.
15
Open Ended
Describe a period of the periodic table.
16
Periodic Table: The three broad Classes
Main, Transition, Rare Earth
Main (Representative), Transition metals, lanthanides and actinides (rare earth)

17
Notable families of the Periodic Table and some important members:
Alkali Metals (Group 1)
Alkaline Earth Metals(Group 2)
Halogens (Group 7)
Noble Gasses(Group 8)

18
Multiple Choice
Which of the following elements is an alkali metal?
calcium
magnesium
mercury
sodium
19
Multiple Choice

Which elements have 1 valence electron?
period 1
group 1
group 4
20
Multiple Choice
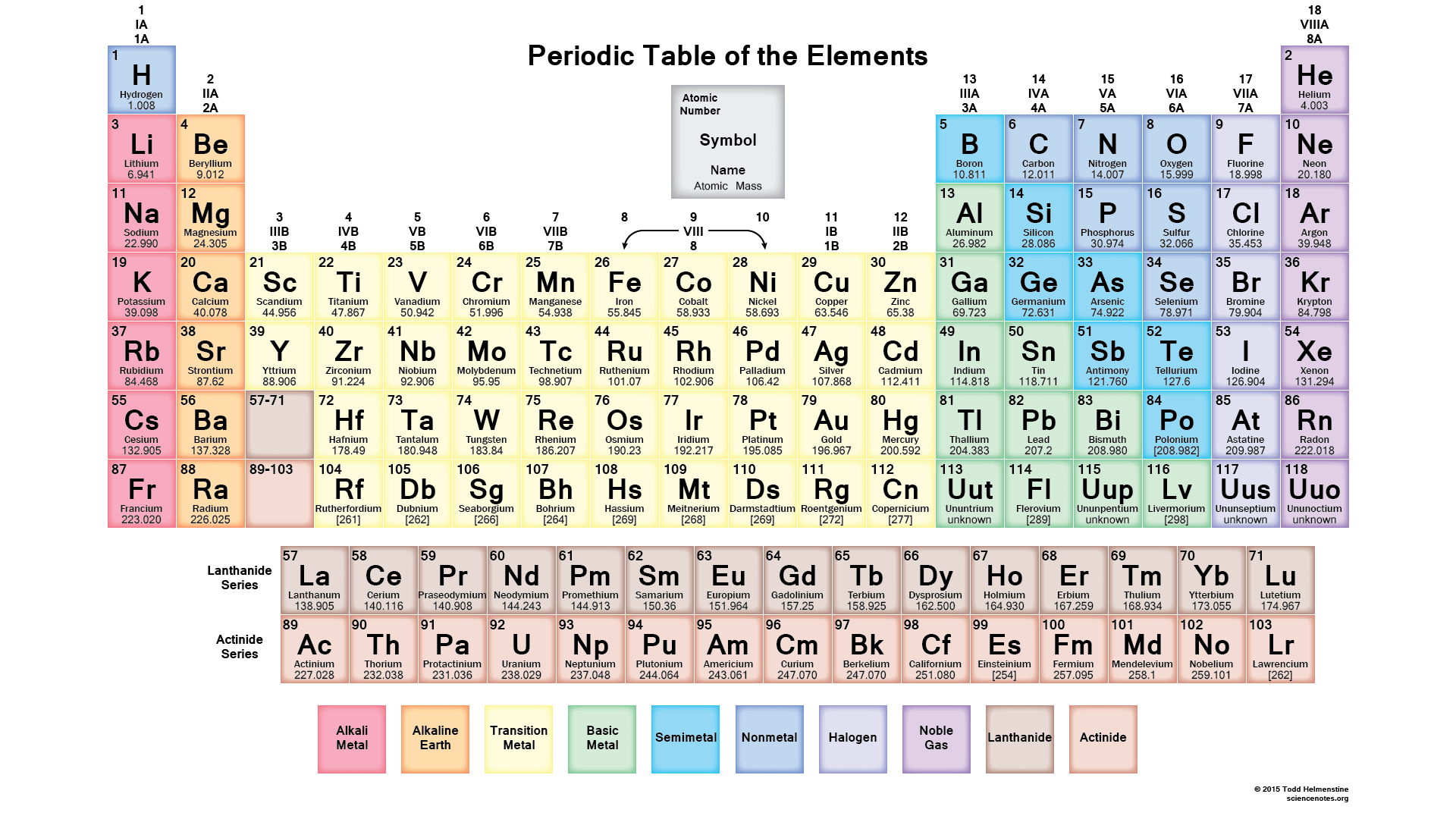
21
Metals
Metals are good conductors of heat and electricity, malleable and ductile (they can be drawn into wire).
Most of the metals are solids at room temperature, with a characteristic silvery shine (except for mercury, which is a liquid).
80%

22
Non Metals
Poor conductors of heat and electricity, and are not malleable or ductile; many of the elemental nonmetals are gases at room temperature
Exist in all three states of matter.
The solids are not lustrous.

23
Metalloids
intermediate in their properties
In their physical properties, they are more like the nonmetals, but under certain circumstances, several of them can be made to conduct electricity.

24
Metalloids
A jagged black line along the right side of the table separates the metals from the nonmetals
metals are to the left of the line (except for hydrogen, which is a nonmetal), the nonmetals are to the right of the line, and the elements immediately adjacent to the line are the metalloids.

25

26
Multiple Choice
Which of these elements are a Non-Metal?
Copper
Gold
Oxygen
27
Multiple Choice
28
Multiple Choice
29

30

31
Alkali Metals
All of these elements have a similar configuration of outer-shell electron
Very reactive metals( water), always combined with something else in nature (like in salt).

32
Multiple Choice
Is hydrogen an alkali metal?
no
yes
maybe
33
Multiple Choice
How many electrons do the alkali metals have in their outer shell?
1
2
8
7
34
Alkaline Earth Metals
“Alkaline”: compounds of these elements form basic (pH greater than 7) or alkaline solutions when dissolved in water.
less reactive than the alkali metals
have two electrons in that outer shell.

35
Transition Metals
Elements in groups 3-11
Less reactive harder metals
Includes metals used in jewelry and construction.
Metals used“as metal.”

36
Halogens
7 Outer-shell electrons, very reactive, non-metals, poor conductors, never uncombined in nature, tend to form salts.

37
Multiple Choice
Which answer has the Chemical Symbols for the elements in Group 7?
F, Cl, Br, I, At, Uus
N, P, As, Sb, Bi, Uup
Be, Mg, Ca, Sr, Ba, Ra
Ab, Cd, Ef, Gh, Ij, Kz
38
Multiple Choice
What group are the Halogens?
6.9
7
8
3
39
Multiple Choice
How many electrons are found in the outer shell of an alkaline earth metal?
1
2
3
4
40
The Noble Gases
Elements whose atoms have full outer shells so they are very unreactive, colorless, odorless gases

41
Multiple Choice

42

43

https://www.pbs.org/video/nova-hunting-the-elements-promo/
The Periodic Table
Mr. Mendoza

Show answer
Auto Play
Slide 1 / 43
SLIDE
Similar Resources on Wayground

39 questions
AP Chemistry Review Lesson
Presentation
•
11th - 12th Grade

38 questions
Electron Distribution
Presentation
•
11th - 12th Grade

38 questions
Fundamental Counting Principle, Permutations & Combinations
Presentation
•
10th - 12th Grade

38 questions
Pre-Icfes intensivo 2
Presentation
•
11th Grade

38 questions
Succession
Presentation
•
10th - 12th Grade

34 questions
Chemistry: Unit 1 Module 5 Dimensional Analysis
Presentation
•
10th - 12th Grade

40 questions
World War I
Presentation
•
11th - 12th Grade

40 questions
Macromolecules
Presentation
•
10th Grade - University
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

20 questions
Le Chatelier's Principle
Quiz
•
9th - 12th Grade

40 questions
AP Chem Review: Units 6-9
Quiz
•
12th Grade

10 questions
AP Chemistry - Unit 2 Review
Quiz
•
11th Grade

6 questions
DOL: Endothermic and Exothermic Reactions
Quiz
•
9th - 11th Grade

20 questions
Uranium: Twisting the Dragon's Tail
Quiz
•
10th - 12th Grade