

Titrations
Presentation
•
Chemistry
•
10th Grade
•
Hard
Anonymous Anonymous
Used 20+ times
FREE Resource
12 Slides • 3 Questions
1
Titrations

2
Acid – Base Titration
•Calculate the concentration of sodium hydroxide using the known concentration of hydrochloric acid.
3
1.Use a pipette to measure 25cm3 of alkali of unknown concentration.
2.Put the alkali into a conical flask with phenolphthalein indicator. Place the conical flask on a white tile.
3.Put the acid into the burette. Run a small amount through the tap and then turn off. Take the starting reading of the volume of acid
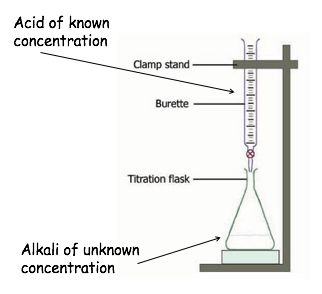
4

5
Carrying Out a Titration
4.Add the acid to the alkali a bit at a time, giving the flask a regular swirl.
5.Watch the colour of the indicator. As it starts to get longer for the colour to reappear, you are getting towards the endpoint of the reaction.
6

7
Carrying Out a Titration
4.Add the acid to the alkali a bit at a time, giving the flask a regular swirl.
5.Watch the colour of the indicator. As it starts to get longer for the colour to reappear, you are getting towards the endpoint of the reaction.
6.As you approach the endpoint, start to add the acid drop by drop.
7.Record the volume of acid used to neutralise the alkali.
8.Repeat at least 3 times.
8
If, for example, your results were:
25.0cm3 of sodium hydroxide required 20.0cm3 of 0.100 mol dm-3 hydrochloric acid for neutralisation.
What is the concentration of the sodium hydroxide?
9

10

11

12

13
Fill in the Blanks
Type answer...
14
Fill in the Blanks
Type answer...
15
Fill in the Blanks
Type answer...
Titrations

Show answer
Auto Play
Slide 1 / 15
SLIDE
Similar Resources on Wayground

11 questions
CIRCULATORY SYSTEM
Presentation
•
10th Grade

11 questions
Parallel Structure
Presentation
•
10th Grade

11 questions
Solubility curves
Presentation
•
10th Grade

12 questions
Gerunds & Infinitives
Presentation
•
10th Grade

13 questions
Tata Nama Senyawa
Presentation
•
10th Grade

10 questions
Evolution
Presentation
•
10th Grade

10 questions
Preposition
Presentation
•
10th Grade

11 questions
Lesson 5: Slope
Presentation
•
10th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade