

Air and Atmosphere
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Medium
Leong Yew Sheng Nicholas
Used 11+ times
FREE Resource
9 Slides • 9 Questions
1
Air and Atmosphere
- take out your C17 Notes on Air and atmosphere
- take out C17B worksheets (referring to some of the questions)

2
Multiple Choice

Heated copper reacts with oxygen to form black copper(II) oxide. The original volume of air was 50.0cm3.
Which graph shows how the volume of air in the gas cylinder changes during the experiment?
3
Multiple Select
Which air pollutants correctly matched to its source?
(Tick more than 1 answers)
carbon monoxide produced from complete combustion of fossil fuels
nitrogen oxides from internal combustion engines at high temperature
nitrogen oxides from exhausts of car engines
sulfur dioxide from volcanic eruptions
sulfur dioxide from the burning of fossil fuels
4
Open Ended

Explain why you choose your option for the previous question.
5
Explain why
carbon monoxide is only produced from the incomplete combustion of carbon-containing fuels. During complete combustion of carbon containing fuel, carbon dioxide is formed instead.
6
Open Ended
The burning of hydrogen is a clean energy. The burning of fossil fuels is not a source of clean energy. Suggest the meaning of the term clean energy. (O TYS 2016)
7
Clean energy
Two key points:
1. does not produce pollutants such as ... (give examples)
2. that does not harm the environment
Source of energy that does not produce pollutants such as sulfur dioxide, and it does not harm the environment.
8
Multiple Choice
The table gives the relative concentration of polluting gases in the air in four different industrialised cities.
In which city are limestone buildings most threatened by pollution?
sulfur dioxide: 17
nitrogen dioxide: 46
carbon monoxide: 23
sulfur dioxide: 32
nitrogen dioxide: 33
carbon monoxide: 30
sulfur dioxide: 38
nitrogen dioxide: 40
carbon monoxide: 11
sulfur dioxide: 45
nitrogen dioxide: 14
carbon monoxide: 21
9
Open Ended

Explain why you choose your option for the previous question.
10
Higher concentration
What does it mean?
- There are more pollutant particles present per unit volume.
Which is the most polluted city based on the diagram on the right?

11
Open Ended
Explain why the incomplete combustion of carbon-containing fuels can be dangerous to people?
12
1. Which pollutant is formed during the incomplete combustion of carbon-containing fuels?
- carbon monoxide
13
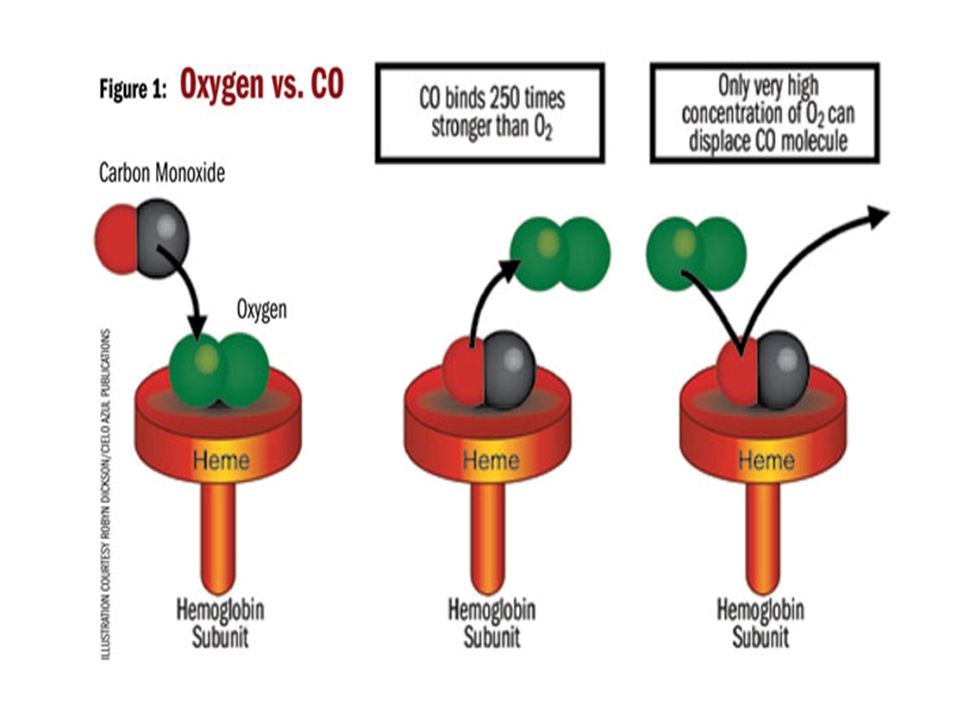
2. Explain why CO is dangerous to people?
- combined irreversibly with haemoglobin to form carboxyhaemoglobin
- prevent red blood cell from transporting oxygen to the rest of the body
- leading to respiratory difficulties and even death

14
Open Ended
Explain why the combustion of fossil fuels can eventually damage building.
15
1. Which pollutant is formed during the combustion of fossil fuels?
- sulfur dioxide (SO2),
- oxides of nitrogen (NO, NO2)

16
2. How does SO2 , NO, NO2 damage building?
- dissolve in rainwater to form acid rain
- acid rain corrodes limestone or metal structures on buildings.

17
Open Ended
Suggest how does the acid rain react with the limestone or metal structure on building?
(Hint: limestone = calcium carbonate)
18
HW
IP day on 15 Mar: O level Practical Practice
(No make-up for missing the practical, no spare time to do make-up)
- annotate 2017 O level Practical before next mon, 15 Mar 2021.
- ask yourself why are we doing each step
- watch the video afterwards to check your answer. (Wait for link)
Air and Atmosphere
- take out your C17 Notes on Air and atmosphere
- take out C17B worksheets (referring to some of the questions)

Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

13 questions
Gerunds and Gerund Phrases
Presentation
•
10th Grade

13 questions
Slopes of Parallel And Perpendicular Lines
Presentation
•
10th Grade

14 questions
Has/ have (Simple Present Tense)
Presentation
•
10th Grade

14 questions
Incomplete Dominance & Codominance
Presentation
•
10th Grade

12 questions
Gerunds & Infinitives
Presentation
•
10th Grade

13 questions
Tata Nama Senyawa
Presentation
•
10th Grade

17 questions
Law of Conservation of Mass
Presentation
•
10th Grade

17 questions
Introduction to Molar Mass
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade