

Limiting Reactants
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+2
Standards-aligned
Benjamin Flo
Used 20+ times
FREE Resource
9 Slides • 11 Questions
1
Limiting Reactants
Essential Questions:
1. What does the amount of reactant have to do with the amount of product in a chemical reaction?
2. How can the limiting reactant be determined given only mole or gram amounts of a chemical reaction?

2
Multiple Choice

What type of reaction involves 2 substances combining to form 1 new compound?
Decomposition Reaction
Single Replacement Reaction
Double Replacement Reaction
Synthesis Reaction
3
Multiple Choice

What type of reaction involves the breaking down of a substance into simpler substances?
Single Replacement Reaction
Double Replacement Reaction
Synthesis Reaction
Decomposition Reaction
4
Multiple Choice
*Question from Yesterday!*
What is the largest number of moles of Al2O3 that
could result from reacting 6 moles of O2?
4 Al (s) + 3 O2 (g) → 2 Al2O3 (s)
2 mol
6 mol
4 mol
1 mol
5
Open Ended
What's your good thing for today OR one thing you're grateful for?
Please don't type N/A and don't forget to type your name after your entry!
6
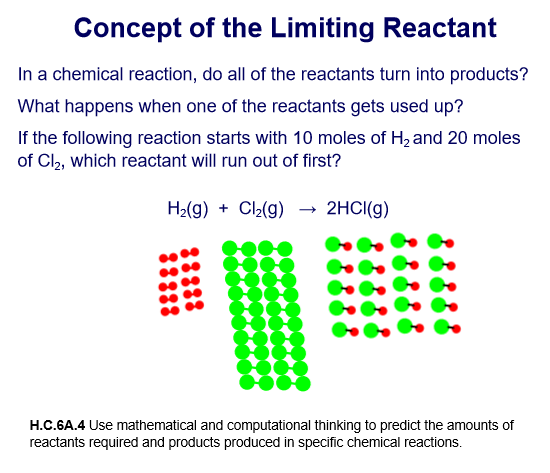
7
Multiple Choice

In the following reaction, which reactant will run out of first?
H2
Cl2
8

9
Open Ended
How would you describe what a limiting reactant is in your own words?
10
Open Ended
Based on what you understand about limiting reactants, when would a reaction stop?
11

12

13

14

15

16
Fill in the Blanks

Type answer...
17
Multiple Choice
When 33 L of nitrogen gas react with 12 grams of
hydrogen gas to make ammonia @STP, the hydrogen
gas will be the excess reactant.
N2(g) + 3H2(g) → 2NH3(g)
True
False
18
Multiple Choice
4NH3+6NO --> 5N2 + 6H2O
19
Multiple Choice
20

Limiting Reactants
Essential Questions:
1. What does the amount of reactant have to do with the amount of product in a chemical reaction?
2. How can the limiting reactant be determined given only mole or gram amounts of a chemical reaction?

Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

15 questions
Chapter 7 Section 3.2: Molar Mass & Percentage Composition
Presentation
•
10th - 12th Grade

18 questions
Acids and Bases: React
Presentation
•
9th - 12th Grade
![[No Qs]Balancing Chemical Reactions & Stoichiometry Practice](https://cf.quizizz.com/image/image-loader.svg)
19 questions
[No Qs]Balancing Chemical Reactions & Stoichiometry Practice
Presentation
•
9th - 12th Grade

17 questions
Percent Composition
Presentation
•
9th - 12th Grade

16 questions
Mole Conversions & Percent Composition Practice
Presentation
•
10th - 12th Grade

13 questions
Molecular Basis of Specific Heat
Presentation
•
9th - 12th Grade

18 questions
Laws of Motion
Presentation
•
9th - 12th Grade

13 questions
Molecular Empirical Lesson
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

120 questions
2nd Semester Final Review
Quiz
•
11th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade