

Unit 2 Review
Presentation
•
Chemistry
•
10th - 12th Grade
•
Medium
Andrew Baldwin
Used 12+ times
FREE Resource
16 Slides • 34 Questions
1
Unit 2 Review

2
Poll
How would you rate the your confidence with this unit?
Very Confident
Confident
Somewhat Confident
Not Very Confident
3

4

5
Multiple Choice
6

7
Multiple Choice

In Rutherford's famous "Gold Foil" experiment, some particles passed through the foil, some were deflected, and some were bounced straight back. This observation made Rutherford conclude
gold atoms have a solid nucleus
gold atoms can conduct electricity
gold atoms are denser than other metals
gold's elections orbit the nucleus at definite distances from the center
8
Multiple Choice
The cathode-ray tube was used to discover the ____ by ____.
electron by Thomson
electron by Dalton
proton by Thomson
proton by Dalton
electron by Rutherford
9
Multiple Choice

In the atomic model nicknamed the "plum pudding" model, what do the plums represent?
the nucleus
the atom
the electrons
the positive material
10

11
Multiple Choice

Look at this picture of Argon off the periodic table. Which of the following is true for this element?
18 protons
40 protons
22 protons
21.9 protons
12

13

14
Multiple Choice
The electron is not included in the calculations for the atomic mass because
It has negative charge
It is located in the outer energy levels of the atom
Its mass is basically zero
It attracts neutral particles
15
Multiple Choice
16
Multiple Choice
Subatomic particles with a negative charge
Electrons
Neutrons
Protons
Quarks
17

18
Multiple Choice

How many protons are in a Chlorine atom?
17
35
35.45
18
19

20
Multiple Choice
How many electrons are present in an atom of Be-9
2
4
5
9
21
Multiple Choice
How many protons, electrons, and neutrons does an atom with an atomic number of 50 and a mass number of 125 contain?
75 protons, 50 electrons, and 70 neutrons
50 protons, 50 electrons, and 75 neutrons
50 protons, 75 electrons, and 120 neutrons
50 protons, 75 electrons, and 50 neutrons
22
Multiple Choice

What is the atomic mass of Neon?
10
20.18
10.18
30.18
23

24
Multiple Choice

25

26
Multiple Choice
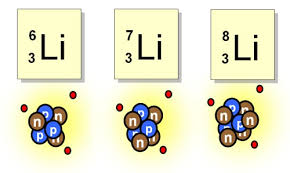
27
Multiple Choice

How many neutrons does the isotope of lithium have?
8
3
4
5
28
Multiple Choice

29
Periodic Table Organization
Organization and groups
30


There are 7 periods.
Each row represents an added electron energy level.
Periods (rows)
There are 18 groups
Each group in the main block elements is equal to the number of valence electrons. (Ex. group 13 = 3 valence electrons
Groups (columns)
31

Elements in the same group have similar properties
The properties are tied to the electron configuration
5 main groups to know their properties
Some text here about the topic of discussion.
Groups/Families
32


As mentioned in the previous slide, as we add rows to the periodic table, we are adding energy levels to the atoms. This is the reason for the pattern seen above. As we proceed down the group, the atoms get larger. But as we go across the period, the atoms get smaller due to the increased nuclear size. The bigger nucleus increases the level of attraction and makes the atom smaller.
Atomic Size
The pattern seen here is inversely related to the image to the left. The graph above shows that as the atoms get bigger it is easier to remove electrons from the valence shell. And as the atoms get smaller, it is harder to remove electrons from the valence shell. This is the same pattern for electronegativity
Ionization energy
33
Electron configuration is used to show the location of electrons in the atom. Use the diagram above to refresh your mind. Remember the four orbital shapes and the four blocks of the periodic table. (refer to your periodic table if you still have it.)
Electron Configuration

34
Multiple Choice
The vertical columns on the periodic table are called:
periods
elements
groups
matter
35
Multiple Choice
The horizontal rows on the periodic table are called:
periods
elements
groups
matter
36
Multiple Choice
Which group are the Halogens in?
1
2
17
18
37
Multiple Choice
Which group are the Noble Gases in?
1
2
17
18
38
Multiple Choice
Which group are the Alkaline Earth Metals in?
1
2
17
18
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
I am a nonmetal
I am in period 2
I am in group 16/6A
Ba
Si
Ba
O
43
Multiple Choice
Of the halogens, which has the smallest radius?
F
Br
He
At
44
Multiple Choice
1s22s22p63s23p64s23d10
45
Multiple Choice
46
Multiple Choice
47
Multiple Choice
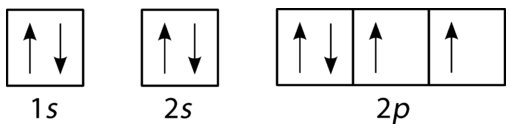
This orbital diagram represents:
Carbon
Boron
Nitrogen
Oxygen
48
Multiple Choice

What is incorrect about this orbital diagram?
Both arrows in the 2p box should be pointing up
There are too many electrons in the 1s orbital
In the 2p boxes, there should only be 1 electron in the first 2p box and one in the 2nd 2p box
All the arrows should be pointing up
49
Multiple Choice

This orbital diagram represents which element?
Sodium
Magnesium
Aluminum
Iron
50
Poll
How would you rate your confidence now?
very confident
confident
somewhat confident
not very confident
Unit 2 Review

Show answer
Auto Play
Slide 1 / 50
SLIDE
Similar Resources on Wayground

45 questions
Water and Aqueous Solutions
Presentation
•
10th - 12th Grade

45 questions
Commonly Confused Words
Presentation
•
10th - 11th Grade

45 questions
Synthetic Materials Tutorial
Presentation
•
9th - 12th Grade

46 questions
Unit 3 Review
Presentation
•
9th - 12th Grade

44 questions
Introduction to Stoichiometry
Presentation
•
9th - 12th Grade

46 questions
Matrices; 11.2
Presentation
•
9th - 12th Grade

44 questions
Bronsted Lowry Acids and Bases
Presentation
•
9th - 12th Grade

47 questions
The Periodic Table
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade