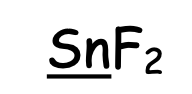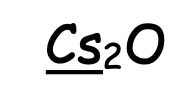Assigning Oxidation Numbers
Presentation
•
Chemistry
•
10th - 12th Grade
•
Medium
•
NGSS
HS-PS1-1, HS-PS1-2, HS-PS1-7
Standards-aligned
Jessica Iseman
Used 63+ times
FREE Resource
3 Slides • 12 Questions
1
2
3
Multiple Choice
4
Multiple Choice
5
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
Show answer
Auto Play
Slide 1 / 15
SLIDE