

Chemistry Semester 1 Credit Recovery
Presentation
•
Chemistry
•
10th - 12th Grade
•
Medium
Alison Bowers
Used 1+ times
FREE Resource
20 Slides • 84 Questions
1
Chemistry Semester 1 Credit Recovery
Work through this Quizizz, watching videos and clicking on any links that you need to use. There are questions embedded as you go and you must get above an 80% to gain your credit back.

2
Big Idea #1: Stuff is made of atoms.
Anything in the universe that has mass and takes up space is called matter. Matter can be converted to energy and energy can be converted to matter, but neither can be created or destroyed.
The smallest particles of matter that we want to understand through chemistry are called atoms. Atoms have particular properties, depending on the number and arrangement of their protons, neutrons, and electrons.
3
4
Multiple Choice
Which best describes a theory?
A guess
A well-tested set of ideas that explains many disparate observations
an educated guess
5
Multiple Choice
The constant, random movement of atoms/molecules/particles smacking into each other is called
Brownian motion
Einstein's theory of relativity
kinetic molecular theory
thermodynamics
6
Multiple Choice
The word "atoms" comes from the Greek atomos, which means "_______."
inedible
irrefutable
indivisible
invisible
7
Multiple Choice
Which is NOT one of the three subatomic particles?
electrons
protons
neutrons
diatoms
8
Multiple Select
The word root "sub" means
below
within
outside of
above
9
Multiple Choice
Which two subatomic particles are found in the nucleus of an atom?
protons and electrons
electrons and neutrons
protons and neutrons
10
Multiple Choice
Which best describes the relationship between protons and neutrons?
They are roughly the same mass, but have different charges.
They have the same charges, but different masses.
They have different masses and different charges.
11
Multiple Choice
Which particle has a mass of 1amu and a positive charge?
protons
neutrons
electrons
12
Multiple Choice
Which particle has a mass of 1amu and a neutral charge?
protons
neutrons
electrons
13
Magnesium's Spot on the Periodic Table
Name
Atomic Number (= # of protons)
Symbol (always 1-2 letters, 1st letter always capitalized)
Relative atomic mass (number of protons + number of neutrons, averaged between the most common isotopes)

14
Multiple Choice
Which number determines what element an atom is?
its mass number
its oxidation number
its atomic number
its electronegativity number
15
Multiple Choice
Looking at your periodic table, an atom that has 18 protons would be
oxygen
neon
fluorine
carbon
16
Multiple Choice
Looking at your periodic table, how many protons does lithium have?
1
3
4
6.94
17
Multiple Choice
T/F: An atom of oxygen always maintains its chemical properties, even when it is bonded to something else to form a compound.
True
False
18
Multiple Choice
What is the chemical symbol for the element with 5 protons?
Ar
B
C
F
19
Multiple Choice
When can the nucleus of an atom change?
During physical changes
During chemical reactions
During nuclear reactions
Never
20
Multiple Select
Which best describes what it means to be an isotope?
Two elements with similar chemical properties
Two atoms of the same element
Two atoms of the same element, but with different numbers of neutrons
Two atoms of the same element, but with different mass numbers
21
Multiple Choice
How do you find an atom's mass number?
Number of protons + electrons
Number of protons + neutrons
Number of electrons + neutrons
22
Multiple Select
Which are TRUE about Carbon-12 and Carbon-14?
They are both the element carbon.
They both have 12 or 14 protons.
They both have 6 protons.
They have different numbers of electrons.
They have different numbers of neutrons.
23
Big Idea #2: Fundamental Laws
Mass is never created or destroyed, but it can change from one form to another.
24
25
Multiple Choice
The Law of Conservation of Mass used to be called
Einstein's Law
Lavoisier's Law
Coulomb's Law
Mendeleev's Law
26
Multiple Choice
27
Multiple Choice
Proust expanded on Lavoisier's work and noticed that chemical compounds always contain the same proportions of elements. This is now called the
Law of Chemical Compounds
Law of Definite Proportions
Law of Proportional Change
Law of Chemical Proportions
28
Multiple Select
Dalton was the first person to show evidence that chemical compounds exist and the Law of Definite Proportions worked. However, he
did the math incorrectly.
didn't believe that chemical compounds existed.
thought atoms were the same thing as molecules.
believed that two atoms could combine to form one new atom, rather than two atoms bonding together to make a compound.
29
Multiple Choice
Gay-Lussac gets credit for collecting air samples and figuring out which piece of info that is obvious to us today?
Oxygen is a gas
Oxygen is O2, or two atoms of oxygen bonded together to act like a compound
Oxygen is a key ingredient for combustion
Oxygen is necessarily for animals' survival
30
Multiple Choice
Whose law states that any two gases at equal temperature, pressure, and volume would have the same number of gas particles?
Gay-Lussac's
Dalton's
Charles'
Avogadro's
31
Multiple Choice
A substance that is made up of two or more elements bonded together in definite proportions is called a(n)
compound
allotrope
element
mixture
32
Multiple Choice
A substance that is made up of one substance that cannot be broken down any further during a chemical reaction or process is a(N)
compound
allotrope
element
mixture
33
Multiple Choice
A substance that is made up of multiple atoms of the same element, but acts differently than an individual atom of that element, is a(n)
compound
allotrope
element
mixture
34
Multiple Choice
Oxygen gas is O2 (two oxygen atoms bonded together), but ozone is O3 (three atoms of oxygen bonded together). These are examples of
compounds
allotropes
elements
mixtures
35
Multiple Choice
Oxygen and hydrogen atoms can bond together to form water (H2O) and hydrogen peroxide (H2O2). These are examples of
compounds
allotropes
elements
mixtures
36
Multiple Choice
Hydrogen atoms have 1 proton and helium atoms have 2. They have different atomic numbers, different masses, and different chemical properties. These are
compounds
allotropes
elements
mixtures
37
38
Multiple Select
Which are TRUE about Mendeleev's childhood?
Father passed away
Mother re-opens an abandoned tavern to support the family
House burns down
Mom thinks he's a smarty-pants, gets him enrolled in a university
Mom immediately dies
39
Multiple Choice
Mendeleev began to notice similarities between the chemical behaviors of different elements. Noticing repeated properties or events in science is the cross-cutting concept
patterns.
energy & matter.
systems and system models.
cause and effect.
40
Multiple Select
Mendeleev became so certain that he understood the pattern that he did what? Select ALL that are true.
left gaps where new elements would/should exist
argued with some French scientist that he himself had pre-discovered one of these elements IN HIS OWN MIND
published a paper saying the French scientist's data was WRONG
created new elements in his lab to fill these gaps.
41
Multiple Choice
42
Multiple Choice
43
Multiple Choice
44
Multiple Choice
45
Multiple Choice
46
Multiple Choice

47
Multiple Choice

48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
51
Multiple Choice
52
Multiple Choice
53
Multiple Choice
Elements which are shiny, conduct electricity and heat are called
metal
nonmetal
metalloid
nonexistent
54
Multiple Select
Which are TRUE about the periodic table?
You can find an element's number of protons on it.
You can find out which elements are similar to each other in chemical properties.
You can use the periodic table to help balance equations.
Groups are horizontal, periods are vertical.
The patterns in the periodic table are caused by electron behavior.
55
56
Electron Configuration
Where electrons are arranged around the nucleus
Found in orbitals
Each orbital holds 2 electrons
57
Arrangement of Orbitals
- Lowest levels are closest to the nucleus
- Number refers to energy level
- Letter refers to shape of the orbitals

58
Rules for Filling Orbitals
Aufbau Principle: Electrons will fill the lowest energy level first
Hund's Rule: Electrons will fill each orbital before combining
59
Writing Electron Configuration
Write the energy level (1s, 2s, 2p, etc.)
Write the number of electrons in the level as an exponent
60
Multiple Choice
How many electrons can the first energy level hold?
1
2
8
0
61
Multiple Choice
1s22s22p63s2
62
Multiple Choice

What's the electron configuration for this atom?
1s22s1
1s1
1s22s22p1
63
Multiple Choice

What's the electron configuration for this atom?
1s22s1
1s1
1s22s22p1
64
Multiple Choice
Which of these is Li? *Atomic Number 3*
65
Multiple Choice

How many electrons are in this atom?
5
8
10
12
66
Multiple Choice

Which rule is broken here?
Aufbau Principle
Hund's Rule
No Rule is Broken
67
Multiple Choice
How many electrons are in each orbital
1
2
3
4
68
Multiple Choice

Which rule is broken here?
Aufbau Principle
Hund's Rule
No rule is broken
Both
69
Multiple Choice
What does the 1 in
3p1 stand for?Energy Level
Shape
Number of electrons
70
Multiple Choice
What does the p in
3p1 stand for?Energy Level
Shape
Number of electrons
71
Multiple Choice
What does the 3 in
3p1 stand for?Energy Level
Shape
Number of electrons
72
73
Chemical Bonds
Bonds form when electrons from one atom are interacting with another atom
Two atoms sharing pair(s) of electrons = covalent
One atom transferring 1 or more valence electron to another atom = ionic
Sharing very equally = nonpolar
Sharing very unequally = polar; one side of the molecule hogs the electrons and gets a negative charge. The other side gets little electron attention and gets a positive charge.
74
Multiple Choice
Atoms always try to reduce their "stress" by being in the lowest possible energy state. They do this by
removing all of their electrons
balancing repulsive and attractive forces
emitting nuclear radiation
forming negatively charged ions
75
Multiple Choice
What happens when two nuclei are too close (or when two protons are too close to each other in the nucleus)?
Like charges attract
Like charges repel
Opposite charges attract
Opposite charges repel
76
Multiple Choice
77
Multiple Choice
78
Multiple Choice

79
Multiple Choice

80
Multiple Choice
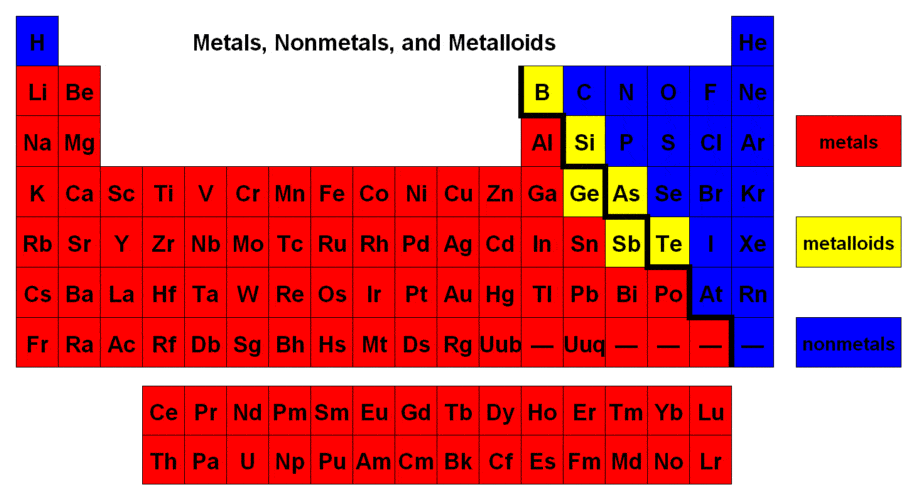
81
Multiple Choice
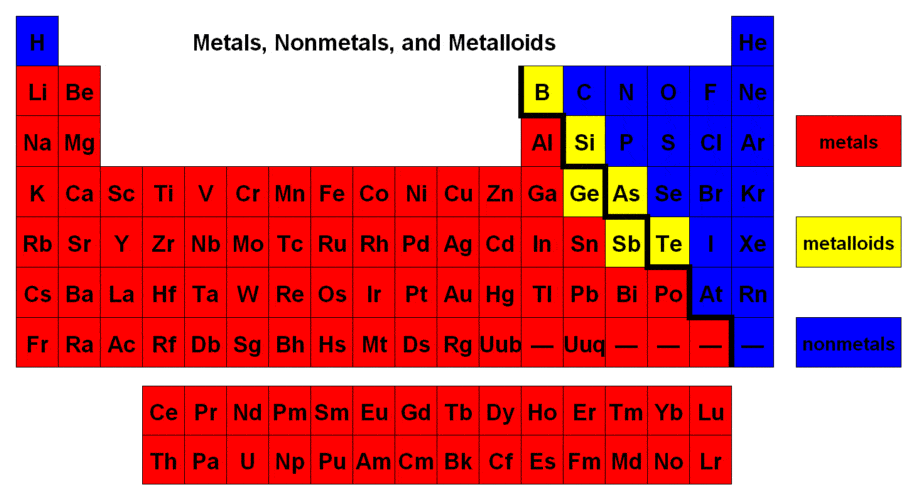
What elements generally make an ionic bond?
metal and nonmetal
2 or more nonmetals
metal
none of the above
82
Multiple Choice

When you have H-Cl, what is the polarity?
nonpolar
polar
ionic
83
Multiple Choice

When you have Br-Br, what is the polarity?
Polar
nonpolar
Ionic
84
Chemical Reactions
Chemical reactions occur when old bonds are broken and/or new bonds are formed.
This involves electrons being transferred and/or shared, but the nucleus of each atom STAYS THE SAME.
Reactants (right side) have their bonds broken and the new bonds created form new products (left side).
Example: Hydrogen gas and oxygen gas (H2 and O2) react to form water (H2O).
The H-H bond and the O-O bond are broken, the H-O-H bonds are formed.
85
Chemical Equations
Must be balanced! The same number of atoms of each element MUST be the same on either side of the arrow.
If you start with 2 oxygen atoms on the right, you better have 2 oxygen atoms on the left.
Mass cannot be created or destroyed!
86
Chemical Equations
Coefficient is the number in front of each chemical formula
Subscripts are the small numbers behind each chemical symbol
EX: 6 H2O
6 = coefficient; 2 = subscript
There are 6 molecules of the compound, H2O.
Therefore there are 6x2= 12 atoms of hydrgen and 6x1=6 atoms of oxygen.
87
Multiple Choice
Ex: 2H2 + O2 --> 2H2O
88
Multiple Choice
How many Mn atoms are found in the reactants of the following equation as indicated by the coefficients?
3 Cr(MnO4)6 + 6 Na3N ----> ______________
3
4
6
18
89
Multiple Choice
4Fe + 3O2 --> 2Fe2O3
90
Multiple Choice
Al + O2 --> 2Al2O3
91
Multiple Choice
92
Multiple Choice
How many iron (Ag) atoms are reactants in this equation?
4Ag + 3O2 → 2Ag2O
4
2
3
0
93
Multiple Choice
Identify the product(s) in the following chemical reaction:
Chromium oxide + Carbon → Chromium + Carbon monoxide
Chromium and carbon.
Chromium and carbon monoxide.
Chromium and carbon dioxide.
Chromium oxide and carbon.
94
Multiple Choice
95
Multiple Choice
96
Multiple Choice
97
Multiple Choice
98
Stoichiometry
Creates a bridge between looking at individual atoms and molecules in a reaction to macroscopic quantities that we can actually measure in the lab, such as grams or kilograms
1 mole of a substance = 6.022x1023 particles of that substance
We know equations must be balanced because mass can't be created or destroyed. Stoichiometry allows us to know what quantity of tiny particles we are working with when we use larger measuring quantities such as grams.
99
Multiple Choice
Balance this equation:
Li + Cl2 → LiCl
2Li + Cl2 → 4LiCl2
2Li + Cl2 → LiCl2
2Li + Cl2 → 2LiCl
2Li + 2Cl2 → 2 LiCl2
100
Multiple Choice
If the balanced equation 2Li + Cl2 → 2LiCl is accurate, how many ATOMS of Li must react with 1 molecule of Cl2?
1 atom of Li per 1 molecule of Cl2
2 atoms of Li per 1 molecule of Cl2
1 atom of Li per 2 molecules of Cl2
2 atoms of Li per 2 molecules of Cl2
101
Multiple Choice
If the balanced equation 2Li + Cl2 → 2LiCl is accurate, how many MOLES of Li must react with 1 MOLE of Cl2?
1 mole of Li per 1 mole of Cl2
2 moles of Li per 1 mole of Cl2
1 mole of Li per 2 moles of Cl2
2 moles of Li per 2 moles of Cl2
102
Molar Mass
pretty straightforward, it's just the mass (in grams) of 1 mole of a substance
Find it by adding up all of the atomic masses for each atom in an element
Atomic masses are found on your periodic table
You'll need this for gas laws in Semester 2, so you may wish to take a picture of the next slide or screenshot it
103

104
Multiple Choice
Calculate the molar mass of HNO3.
63.01
4
32
18.04
Chemistry Semester 1 Credit Recovery
Work through this Quizizz, watching videos and clicking on any links that you need to use. There are questions embedded as you go and you must get above an 80% to gain your credit back.

Show answer
Auto Play
Slide 1 / 104
SLIDE
Similar Resources on Wayground

97 questions
Minerals, Rocks, & Nat. Resources Review ES.4, 5, & 6
Presentation
•
9th - 11th Grade

93 questions
Political Parties
Presentation
•
11th - 12th Grade

96 questions
BIOTEKNOLOGI KURIKULUM MERDEKA
Presentation
•
9th - 12th Grade

98 questions
Training 2
Presentation
•
KG

95 questions
STAAR Revising and Editing (2025)
Presentation
•
9th - 12th Grade

97 questions
Kirchoff's Law
Presentation
•
9th - 12th Grade

98 questions
WH Question
Presentation
•
KG - University

101 questions
Culture, Race, and Gender
Presentation
•
KG - University
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade