

Chemistry review for AP Bio
Presentation
•
Biology, Chemistry
•
9th - 12th Grade
•
Easy
+1
Standards-aligned
Hannah Hathaway
Used 15+ times
FREE Resource
3 Slides • 24 Questions
1
Multiple Choice
The smallest particle of a substance is......
atom
element
molecule
compound
2
Multiple Choice
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
What type of charge does a neutron have?
negative
positive
no charge/neutral
7
Watch this video to review the different types of chemical bonds.
You can click on the title in top left of the video to open it larger in another tab.
8
Multiple Choice
Bond where electrons are shared
Ionic Bonds
Covalent Bonds
Both bonds
Neither bond
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice

The most common atoms in biology are carbon, hydrogen, oxygen and nitrogen.
These atoms are....
metals
non-metals
13
Multiple Choice

What kind of atoms are in this molecule of water?
metals only
non-metals only
both metals and non-metals
14
Multiple Choice

What kind of intramolecular bonds are in this molecule of water?
(intramolecular bonds are the bonds that hold together atoms within a single molecule)
ionic bonds only
covalent bonds only
both ionic and covalent bonds
15
Multiple Choice

What kind of intramolecular bonds are in this molecule of glucose?
ionic bonds only
covalent bonds only
both ionic and covalent bonds
16
Multiple Choice

What kind of intramolecular bonds are in this molecule of sodium chloride (NaCl)?
ionic bonds only
covalent bonds only
both ionic and covalent bonds
17
Watch this video to review polarity.
You can click on the title in top left of the video to open it larger in another tab.
18
Fill in the Blanks
Type answer...
19
Multiple Choice
The attraction that an atom has for shared electrons in bonds is
electron affinity.
electronegativity.
electropositivity.
electromagnetism.
20
Multiple Choice
In a polar covalent bond, electrons are shared:
equally
unequally
between two metals
between a metal and a non-metal
21
Multiple Choice
Polar molecules have...
no charges.
partial positive and negative charges on opposite ends of the molecules.
only partial positive charges.
either partial positive or negative charges, but not both.
22
Multiple Choice
Electronegativity is a measurement of the ability of an atom to:
attract electrons
repel electrons
attract protons
attract other atoms
23
Multiple Choice
In order to determine if a molecule is polar, you must consider the
polarity of bonds and symmetry of the molecule.
number of electrons and number of bonds.
polarity of bonds only.
total charge of each ion and number of multiple bonds.
24
Multiple Choice

Is this molecule of NH3 polar or nonpolar?
polar
nonpolar
25
Multiple Choice
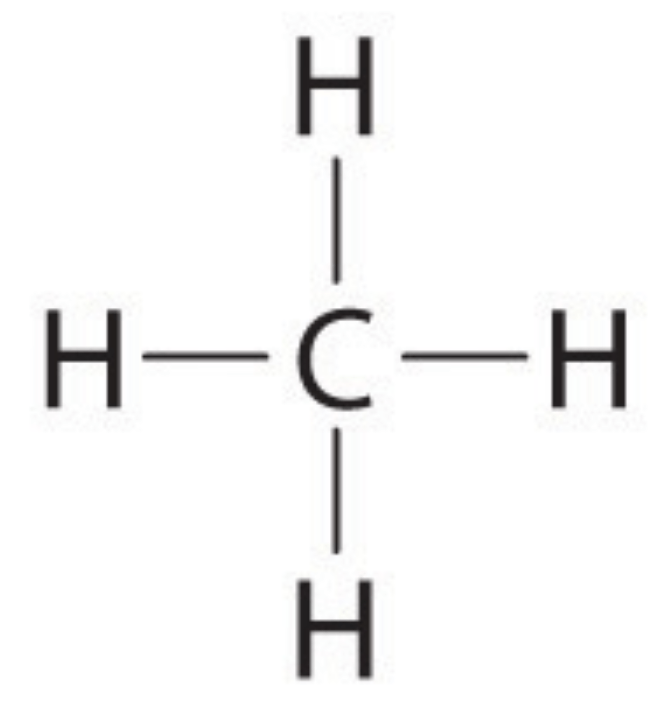
Is this molecule of CH4 polar or nonpolar?
polar
nonpolar
26
Multiple Choice

Is the long hydrophobic chain (highlighted in purple in the picture) of a fatty acid polar or nonpolar?
polar
nonpolar
27
Nice work!!
You made it through the chem review lesson, good job!!
If you feel like you need more practice here is a youtube playlists of helpful videos to review essentials from chemistry:
https://bit.ly/chem-review
The smallest particle of a substance is......
atom
element
molecule
compound
Show answer
Auto Play
Slide 1 / 27
MULTIPLE CHOICE
Similar Resources on Wayground

22 questions
U6L5 - Lecture Notes - Electronegativity and Polarity
Presentation
•
9th - 12th Grade

21 questions
LESSON - Classifying and Balancing Reactions
Presentation
•
9th - 12th Grade

22 questions
Parts of a Cell Lesson
Presentation
•
9th - 12th Grade

22 questions
Solutions Lesson Part 1
Presentation
•
9th - 12th Grade

22 questions
Lactose and Lactase
Presentation
•
9th - 12th Grade

22 questions
DNA structure
Presentation
•
10th - 12th Grade

24 questions
Metric System
Presentation
•
9th - 12th Grade

23 questions
Stock System - Naming Ionic Compounds
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Biology

50 questions
Biology EOC Review
Quiz
•
9th Grade

15 questions
Biology EOC Review P2
Quiz
•
10th Grade

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade

20 questions
Biology EOC Review 1
Quiz
•
10th Grade

18 questions
Population Dynamics
Quiz
•
7th - 12th Grade

50 questions
NC EOC Biology Review part 1
Quiz
•
9th - 12th Grade

25 questions
photosynthesis and cellular respiration
Quiz
•
9th Grade

10 questions
Ameoba Sisters: Introduction to cells
Interactive video
•
9th - 12th Grade