

Elements, Compounds and Mixtures
Presentation
•
Chemistry, Science
•
7th - 10th Grade
•
Easy
Standards-aligned
X Y
Used 1+ times
FREE Resource
23 Slides • 18 Questions
1
Elements, Compounds and Mixtures

2
3
Learning goals
At the end of this section, you should be able to:
Write the definition of element
State the chemical symbol of elements using Periodic Table
Subject | Subject
Some text here about the topic of discussion
4
It is a pure substance that cannot be broken down into two or more simpler substances by chemical processes.
Some text here about the topic of discussion.
Element

5
Poll
Is water an element?
Yes
No
not sure
6
Open Ended
Is air an element? (just a yes or no respond will do)
7
Open Ended
Is Sugar an element?
8
Sugar is not an element.
When heated, it breaks down into carbon and water vapour.
Some text here about the topic of discussion.
Is Sugar an element?


9
Two key questions to check whether a substance is an element.
Can the substance be broken down into two or more simpler substances?
Can it be found in the periodic table?
Subject | Subject
Some text here about the topic of discussion
10
Chemists use chemical symbols to represent elements.
Each symbol is unique, consisting of one or two letters.
Some text here about the topic of discussion.
Chemical symbols of elements

11

Elements can be classified into:
Metals
Metalloids
Non-metals
Some text here about the topic of discussion.
Classifying elements
12

Elements are classified based on:
Metallic and non-metallic properties
Physical states (at r.t.p)
Some text here about the topic of discussion.
Classifying elements
13

Some text here about the topic of discussion.
Classifying elements
14
Multiple Choice
Using the Periodic Table, find the symbols of Aluminium.
AL
al
Al
15
Multiple Choice
Using the Periodic Table, find the symbols of Magnesium.
MG
mg
Mg
16
Multiple Choice
Using the Periodic Table, find the symbols of Lead.
PB
pb
Pb
17
Multiple Choice
Using the Periodic Table, find the symbols of Copper.
Cu
CU
cu
18
Multiple Choice
Using the Periodic Table, find the symbols of Nitrogen.
N
n
19
Multiple Choice
Using the Periodic Table, find the symbols of Sulfur.
S
s
20
Elements exist in atoms and molecules.
Some text here about the topic of discussion.
Atoms and Molecules

21
Atoms are the smallest particles of an element that have the chemical properties of that element.
The atoms of an element are different from that of another element.
Some text here about the topic of discussion.
Atoms and Molecules

22

A molecule is a group of two or more atoms that are chemically combined (joined together).
Molecules exist in diatomic or polyatomic.
Some text here about the topic of discussion.
Atoms and Molecules
23
Draw
Circle all the molecules in the picture
24
What is a compound?
A compound is a pure substance that contains two or more elements that are chemically combined in a fixed ratio.
Some text here about the topic of discussion.
Compound


25
Ions
Molecules
What are compounds made of?
Some text here about the topic of discussion


26
A compound has different properties from the elements that form it.
Some text here about the topic of discussion.
Properties of Compounds
27
Names of Compounds
Each compound has a chemical name indicating the elements it contains.
Subject | Subject
Some text here about the topic of discussion

28
Chemical Formulas of Compounds
A compound can be represented by a chemical formula.
The chemical formula shows:
the types of elements present;
the ratio of the different atoms present.
Subject | Subject
Some text here about the topic of discussion

29
Composition of Compounds
Every compound has fixed compositions of the different elements it contains.
Chemical formula can be deduced by the ratio of atoms present in a compound.
Subject | Subject
Some text here about the topic of discussion

30
Decomposition of Compounds
A chemical reaction is necessary to separate the elements in a compound.
In thermal decomposition, heat is used to break down compounds into elements or simpler compounds.
Compounds can also be broken down by using electricity. This process is known as electrolysis.
Subject | Subject
Some text here about the topic of discussion

31
Open Ended
Is water an element? Explain your answer.
32
Multiple Choice
33
Learning goals
At the end of this section, you should be able to:
define a mixture;
differentiate between compounds and mixtures.
Some text here about the topic of discussion
34

Mixtures are made up of two or more substances that are not chemically combined.
Some text here about the topic of discussion.
What are mixtures?
35
There are a few scenarios on how substances are mixed together physically:
mixture of elements
mixture of compounds
mixture of compound and element
Type of mixtures

36
Multiple Select

Which of the following is a pure substance? (you can choose more than one answer)
A
B
C
D
37
Multiple Choice

Which substance is a solid?
A
B
C
D
38
Multiple Choice

Which diagram represent a solution?
A
B
None are solutions
39
Multiple Select
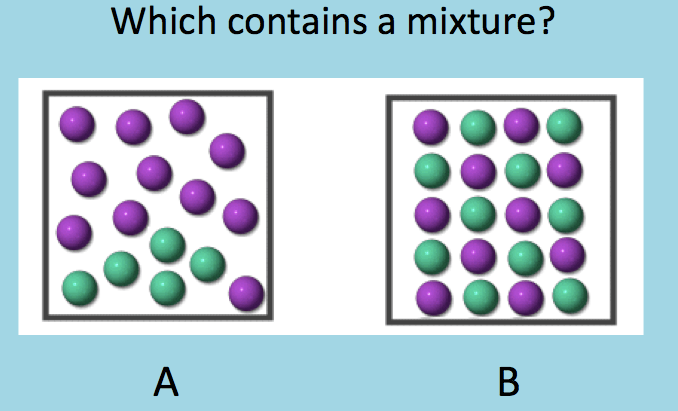
Which of the following is a mixture?
A
B
40
Multiple Select

Which of the following diagram represent a pure substance?
A
B
C
D
41
Multiple Choice

Which of the following contain only elements?
A
B
C
D
Elements, Compounds and Mixtures

Show answer
Auto Play
Slide 1 / 41
SLIDE
Similar Resources on Wayground

39 questions
Lab #2: Measuring Earthquakes
Presentation
•
8th - 10th Grade

37 questions
Force and Motion Review
Presentation
•
7th - 8th Grade

38 questions
sound waves
Presentation
•
7th - 9th Grade

37 questions
Nature of Matter
Presentation
•
7th - 10th Grade

32 questions
Groups of the Periodic Table
Presentation
•
7th - 9th Grade

37 questions
Atomic Structure and the Periodic Table
Presentation
•
7th - 10th Grade

37 questions
Periodic Table Isotopes and Atomic Structure
Presentation
•
7th - 10th Grade

34 questions
Collision Theory & Reaction Rate Experiment
Presentation
•
KG
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

15 questions
Acids and Bases Review
Quiz
•
8th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade