

Chemistry: Unit 1 Module 2 - What is Chemistry
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+1
Standards-aligned
Ryan McCluskey
Used 44+ times
FREE Resource
27 Slides • 9 Questions
1
What is Chemistry?
Unit 1 Lesson 2
2

3

4

5
Physical Sciences

Chemistry-and Physics
Physics is the study of energy and motion.
6
Physical Sciences

Chemistry-and Physics
Chemistry can be defined as the study of matter and all of it’s interactions.
7
But What is Matter then?
Basically Matter is defined as anything that has MASS and takes up SPACE.
But I think it is sometimes easier to describe what isn't matter
Everything is matter except for ENERGY. I.E. Heat, Light, Electricity.
8
So what is Mass and how is it different from weight?
9
Takes up Space?
Besides mass matter all takes up some sort of space.
This space is defined as the volume of matter.
We will measure volume in several different ways but it is simply a measure of the space occupied by a from of matter.

10
Multiple Choice

Which of these would be a measure of mass?
10 meters
10 grams
10 pounds
10 seconds
11
Multiple Choice

Chemistry is often defined as the study of __________.
Matter
Energy
Magic
Motion
12

In chemistry we strive to identify matter and separate it by it's properties.
Chemistry Measurements
Properties can be sorted into two categories.
Intensive - Don't change based on amount
Extensive - Do change based on amount
13

Intensive properties are very important for separating matter or classifying it. In chemistry one of the most common intensive properties used is Density.
Chemistry Measurements
Properties can be sorted into two categories.
Intensive - Don't change based on amount
Extensive - Do change based on amount
14

Density is the ratio of a substances mass to it's volume. Two very common measurements taken in a chemistry lab.
Chemistry Measurements
Density is intensive because the ratio stays the same no matter how much or how little of a substance you have.
Density is commonly used to separate substances that look very similar.
15

Chemistry Measurements
A gold sluice can separate the gold from sand because of it's very high density.

16

17

18

19

20

21
Multiple Choice
The volume of a candy bar is 55cm3.The mass of the candy bar is 70g. What is the density of the candy bar?
2.3g/cm3
3850g/cm3
.8g/cm3
1.3g/cm3
22
Multiple Choice

2 g/cm3
4 g/cm3
6 g/cm3
8 g/cm3
23
Multiple Choice

Show all your work on your answer document. D=m/v
0.33 g/cm3
15 g/cm3
3 g/cm3
9 g/cm3
24
Multiple Choice
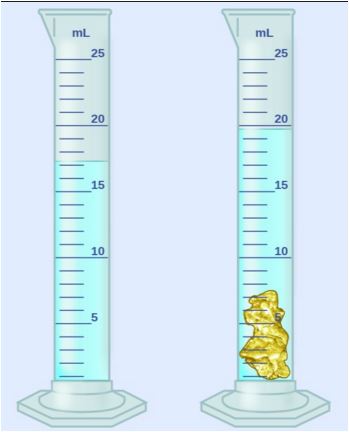
9 g/mL
10.5 g/mL
6.5 g/mL
8 g/mL
25
Properties and Changes of Matter

26
Properties of Matter
In order to classify matter we have to be able to identify properties of matter.
These properties can be measured with our senses or with chemical processes and equipment.

27
Physical Properties
Anything you can observe about an object or substance without changing the substance which makes it up.
Examples: Density, shape, size, color, magnetism, melting and boiling point

28
Physical Properties
Physical Properties can be Qualitative or Quantitative.
Qualitative properties are described without using numbers or value. They describe the quality of the sample.

29
Physical Properties
Physical Properties can be Qualitative or Quantitative.
Quantitative properties are described by using numbers or value. They measure some quantity of the sample.

30
Physical Changes
Physical changes are those changes which do not change the composition of the substance only the shape or state.
The most commonly mistaken are phase changes. Boiling water till it evaporates is only a physical change.

31
Chemical Properties
Chemical properties are those properties which indicate whether a substance will undergo specific chemical processes.
Examples: Flammability, reactivity with acids or bases or combustibility.

32
Chemical Changes
Any change that can change the composition of the substance.
Since we can't see the composition of the substance we look for common evidence of a chemical change.

33
Chemical Changes
Evidence a chemical change has occurred:
Production of gas - smoke, bubbles, odor
Production of light - flames, sparks, glow sticks
Change in temperature - hot or cold without added source
Formation of a precipitate - a solid forms when two liquids are mixed
Change in color - color is not added but is result of reaction

34
Multiple Select
Which of the following is an intensive property?
Freezing Point
Mass
Color
Length
Volume
35
Multiple Choice
An ice cream cone melts in a kid's hand while walking outside on a hot summer day. What is this an example of?
Physical property
Chemical property
Physical change
Chemical change
36
Multiple Choice
Water boils at 100˚C. What is this an example of?
Physical property
Chemical property
Physical change
Chemical change
What is Chemistry?
Unit 1 Lesson 2
Show answer
Auto Play
Slide 1 / 36
SLIDE
Similar Resources on Wayground

29 questions
Properties of Parallelograms
Presentation
•
9th - 12th Grade

31 questions
Periodic Table Structure
Presentation
•
9th - 12th Grade

29 questions
Thermochemistry & Kinetics Review
Presentation
•
9th Grade - University

29 questions
Lessons 12.2 and 12.3
Presentation
•
9th - 12th Grade

35 questions
Balancing Chemical Equations
Presentation
•
9th - 12th Grade

31 questions
Chemical & Physical - Changes & Properties
Presentation
•
9th - 12th Grade

25 questions
Introduction to Electrons
Presentation
•
9th - 12th Grade

28 questions
Introduction to Kinetic Molecular Theory
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade