

Isotopes Lesson
Presentation
•
Chemistry, Science
•
10th - 12th Grade
•
Medium
Standards-aligned
Andrew Baldwin
Used 30+ times
FREE Resource
6 Slides • 11 Questions
1
Isotopes Lesson
by Andrew Baldwin
2
Objectives
Students will be able to explain how the atomic structure of isotopes are similar and different from one another.
3
Multiple Choice
Niels Bohr added _______________ to the model of the atom.
electrons.
a nucleus.
neutrons.
energy levels.
4
Multiple Choice
Which of the following is the best description of J.J. Thomson's model of the atom?
a featureless, indivisible sphere
a sphere of positive material with electrons embedded in it
mostly empty space with electrons randomly orbiting around a positive nucleus
mostly empty space with electrons located in set orbits around a positive nucleus.
5
Multiple Choice
Who proposed the following model for the atom?
"Atoms are solid spheres. Each element is made of a different type of atom."
Bohr
Chadwick
Dalton
Rutherford
6
Multiple Choice

How many protons are in a Chlorine atom? (Mass number = 35)
17
35
35.45
18
7
Multiple Choice

How many neutrons are in a Phosphorus atom? (Mass Number = 31)
15
30.9738
16
31
8

9
Reading the Symbol
Note: This is not reflective of the Periodic table Organization.
Mass Number provided at the top and atomic number sometimes at the bottom.
The only change should be the top number.

10
Anything else?
Sometimes it is given in Hyphen notation.
Element symbol or name hyphen (dash) Mass Number.

11
Multiple Choice
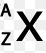
In a correctly written symbol what would be located in the "A" position?
number of neutrons
atomic number
number of electrons
mass number
12
Multiple Choice
What quantities vary between isotopes of an element?
protons, electrons, and atomic mass
protons, electrons, and atomic number
neutrons and electrons
neutrons and atomic mass
13
Multiple Choice

What do these isotopes of carbon all have in common?
neutrons & mass number
atomic number and neutrons
atomic number and electrons
protons, atomic number, and mass number
14
Multiple Choice

How many protons does this isotope of titanium have?
48
22
26
70
15
Multiple Choice
Isotopes are atoms of the same element with different #’s of __________ & therefore different __________.
n0 ; atomic #’s
p+ ; atomic #’s
e- ; atomic masses
n0 ; atomic masses
16
Multiple Choice

If X is the symbol for an element, which of the following two symbols represent isotopes of the same element?
I and II
III and IV
I and IV
I and III
17
Time for Practice
Back to Canvas and Complete Counting Particles Level 2
Isotopes Lesson
by Andrew Baldwin
Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

16 questions
Gas Laws Review (participation)
Presentation
•
10th - 12th Grade

13 questions
Calculating Molar Mass Day 1
Presentation
•
9th - 12th Grade

14 questions
Multiple Ionization Energies
Presentation
•
10th - 12th Grade

12 questions
Particle/Mole Conversions
Presentation
•
9th - 12th Grade

12 questions
Quantum Mechanical Model of the Atom- Introduction
Presentation
•
10th - 12th Grade

15 questions
Stoichiometry Review Lesson
Presentation
•
9th - 12th Grade

11 questions
Naming Organic Compounds
Presentation
•
10th - 11th Grade

16 questions
Bohr Model
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade