

Midterm Review
Presentation
•
Science
•
University
•
Easy
Miss TN
Used 2+ times
FREE Resource
10 Slides • 81 Questions
1
Thermodynamics:
Midterm Review
by Tessa Noora
2
Temperature and Thermometers
3
MINI QUIZ 1
4
Multiple Choice
5
Multiple Choice
Railroad tracks have gaps between rails so they don't buckle in hot weather because of
6
Multiple Choice

More rapidly at an increased distance from each other.
7
Multiple Choice

8
Multiple Choice
9
Multiple Choice
Which of the following is NOT a part of the Kinetic Theory of Matter?
All matter is made up of tiny particles.
Particles are always moving in random paths
Occasionally, particles stop moving.
The type of movement of particles depends on the amount of energy.
10
Multiple Choice

What happens to the average kinetic energy of matter when it is heated?
It increases
It stays the same
It decreases
It cannot be determined
11
Multiple Choice
The random motion of the particles in a substance is greatest when the substance is:
condensed
gas
frozen
liquid
12
Multiple Choice
What is the measure of the average kinetic energy of the particles in an object?
Heat
Motion
Thermal Energy
Temperature
13
Multiple Choice
When particles collide with each other they...
stop completely still
transfer kinetic energy
lose all their energy
become smaller
14
Multiple Choice
Absolute zero is the temperature (in Kelvin), at which, only in theory...
space and time merge together
energy goes to infinity
time stands still
all particles stop moving
15
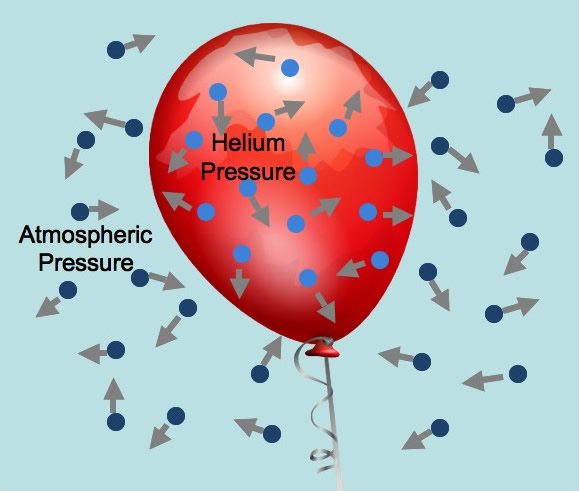
Multiple Choice
An inflated balloon is placed in a refrigerator. Which statement the movement of the gas particles in the balloon?
The particles move faster and become further apart.
The particles move more slowly and become further apart.
The particles move faster and become closer together.
The particles move more slowly and become closer together.
16
Multiple Choice
Which of the following statements about the particles of a gas is true?
The particles vibrate about a fixed position.
The particles are far apart and move freely.
The particles are packed close together, but in a disorderly arrangement.
The particles are stationary and orderly.
17
Multiple Choice
When gas particles collide with other particles, they ...
Travel in a spiral.
Lose energy and move slowly.
Stop motion.
Rebound in a straight line.
18
Multiple Choice
A substance Y has a melting point of -7 degrees Celsius and a boiling point of 63 degrees Celsius. At which temperature is Y a liquid?
-25
25
75
150
19
Multiple Choice
The state of matter with definite volume but no definite shape.
solid
liquid
gas
plasma
20
Multiple Choice
Which of the following is true when particles gain thermal energy?
Particles move more slowly.
Particles move closer together.
Particles stop moving.
Particles move further apart.
21
Multiple Choice

This diagram represents particles in a __________ state.
solid
liquid
gas
plasma
22
Multiple Choice
373 K
23
Multiple Choice
Convert 45 degree Celsius into degree Fahrenheit
77
113
102
110
24
Thermal Expansion
25
MINI QUIZ 2
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice

The picture shows a bimetallic strip in a fire alarm. When there is a fire outbreak, the bimetallic strip will get hot, ....
bend downwards, complete the circuit and the alarm rings.
bend upwards, open the circuit and the alarm will not ring.
becomes longer, open the circuit and the alarm will not ring.
29
Multiple Choice
What is Thermal Contraction
The movement of particles.
A decrease in a materials volume when its temperature decreases
The movement of fluids in a cycle because of convection
A material through which thermal energy does not flow easily.
30
Multiple Choice
31
Multiple Choice

32
Multiple Choice

33
Multiple Choice
Take the coefficient of expansion to be 25 x 10-6
34
Multiple Choice
α = 25 x 10-6
35
Multiple Choice
What is the purpose of the expansion joints in bridges?
They contract and expand with weather changes
To avoid collapse due to high winds
To stop bridges from expanding and contracting
They allow the bridge to expand and contract freely
36
Multiple Choice
Is the following statement true or false?
An object that expands more when heated, will also contract more when the temperature is reduced
True
False
37
Multiple Choice
The volume of the water contract with increase in temperature between......
5°C to 10°C
10°C to 20°C
0°C to 4°C
-0°C to -4°C
38
Multiple Choice
The length of copper wire at 20oC is 4.5m. Calculate its new length when the temperature is 95oC. Given α = 1.7×10-5 oC-1
4.55m
5.41m
5.11m
4.505m
39
Multiple Choice
A silver plate of 16cm2 is heated from 20 degrees to 100 degrees. What is the increase of area? assume α=2×10−5
0.051
16.051
0.0256
16.0256
40
Multiple Choice

During the Investigation "Ready, Set, Collide" on p.13, why did the particles diffuse quicker in hot water.
The particles in the hot water diffused quicker because the water molecules were moving faster, colliding, and moving further apart.
The particles in the hot water diffused slower because the water molecules were moving slower and not colliding as much.
41
Multiple Select

Which of the following statements are true about temperature? Choose all the true statements
It is the measure of kinetic energy
Temperature is measuring the speed of the particles
Temperature increases when kinetic energy increases
Temperature decreases when kinetic energy decreases
None of the statements are true
42
Multiple Select

Which statement is true about how gas particles behave?
(p.19) - Choose all the TRUE statements
When the balloon was placed in the freezer, the gas particles moved slower, closer together, and thermal contraction occurred.
When the balloon returned to room temperature, the gas particles moved faster, farther apart, and thermal expansion occurred.
Thermal expansion and contraction only happen in solids and not in gases.
43
Multiple Choice
Assuming that the thermal energy supplied to each of them is same, which of following states of matter would expand the least?
Solid
Liquid
Gas
44
Multiple Choice
Contraction can be a problem in
Power cable
Railway tracks
Fire alarm
45
Multiple Choice
Which of the images best describe thermal expansion of solid?
46
Gas Laws
47
MINI QUIZ 3
48
Multiple Choice
49
Multiple Choice
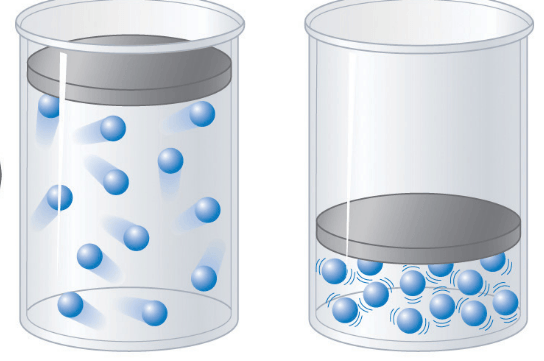
50
Multiple Choice
51
Multiple Choice
52
Multiple Choice
53
Multiple Choice
A student measures the pressure and volume of an empty water bottle to be 1.4 atm and 2.3 L. She then decreases the pressure to 0.65 atm. What is the new volume?
2.1 L
5.0 L
8.2 L
3.9 L
54
Multiple Choice
A scientist needs to store 28.0 moles of xenon gas at STP in the lab. What size gas tank will he need?
628 L
473 L
219 L
376 L
55
Multiple Choice
A sample of gas containing 8.1 moles at STP would fill a container of what volume?
342 L
703 L
475 L
182 L
56
Multiple Choice
A water bottle at STP is cooled to -155°C. What is the new pressure?
3.2 atm
0.43 atm
1.8 atm
0.27 atm
57
Multiple Choice
The pressure of a 6.5 L sample of oxygen gas is measured to be 3.8 atm. If the gas is transferred into a 12 L tank, what is the new pressure?
6.6 atm
3.9 atm
4.5 atm
2.1 atm
58
Multiple Choice
A student transfers a gas at STP from a 11.0 L tank to a 25.0 L tank. If the pressure remains constant, what is the new temperature?
620 K
393 K
254 K
530 K
59
Multiple Choice
What is the pressure of a car tire that had an initial pressure of 1.8 atm but was heated from 38°C to 123°C?
0.9 atm
2.1 atm
1.6 atm
3.4 atm
60
Multiple Choice
A sample of gas containing 9.2 moles is transferred from an 13 L tank to a 23 L tank. What is the new number of moles that can be stored in the tank?
10.2 mol
13.7 mol
19.1 mol
16.3 mol
61
Multiple Choice
What is the volume of a balloon that contains 3.7 moles of helium at 75°C and 5.1 atm?
37 L
13 L
45 L
21 L
62
Multiple Choice
Avogadro's Law explains the relationship between
Pressure and Volume
Pressure and Temperature
Volume and Temperature
Volume and Moles
63
Multiple Choice
If a hairspray can is heated, what can be expected of the pressure of the gas inside the can?
The pressure will increase
The pressure will decrease
The pressure will remain constant
The pressure will equalize
64
Multiple Choice
65
Multiple Choice
Determine the temperature required for 0.0470 mol of gas to fill a balloon to 1.20 L under .998 atm pressure.
0 K
107 K
207 K
307 K
66
Multiple Choice
67
Multiple Choice
68
Heat
69
MINI QUIZ 4
70
Multiple Choice
For the formula: Q= m c ∆T The units for specific heat are:
g /J C
°C/g J
kJ/g
J/g°C
71
Multiple Choice
The KE of all the molecules in the system
U
Q
T
C
72
Multiple Choice
Which of the following is the primary factor in diffusion?
concentration gradient
temperature gradient
presence of external force
none of the given choices
73
Multiple Choice
For a skillet, used for cooking, do you want a high or low specific heat?
High, so food can be cooked faster
Low, so food can be cooked faster
High, so the skillet would be hotter
Low, so the skillet would be hotter
74
Multiple Choice
Water molecules have the greatest kinetic energy in ________________
Ice at 0 °C.
Water at 373 K.
Water at 98 °C
Steam at 150 °C.
75
Multiple Choice
A high specific heat means...
It heats up quickly with energy added
It requires more energy to change temperature
76
Multiple Choice
A metal cube at temperature of 10°C immersed in a liquid at temperature of 70°C. What is the temperature of the metal cube when thermal equilibrium is achieved between the cube and the liquid?
Between 10°C and 70°C
More than 70°C
Less than 10°C
Same as the room temperature
77
Multiple Choice
What is the specific heat of an unknown substance if 100.0 g of it at 200.0 °C reaches an equilibrium temperature of 27.1 °C when it comes in contact with a calorimeter of water. The water weighs 75. g and had an initial temperature of 20.00 °C? (Specific heat of water is 4.18 J/g°C)
0.111 J/g°C
1.29 J/g°C
0.129 J/g°C
22225.85 J
78
Multiple Choice
A sample of iron receives 50.J of heat energy that raises the temperature of the iron by 25.0°C. If iron has a specific heat of 0.10 J/g°C, what is the mass of the iron sample?
25g
30g
20g
50g
79
Multiple Choice
A piece of metal with a mass of 32.8 g is heated to 100.5°C and dropped into 138.2 g of water at 20.0°C. The final temperature of the system is 30.2°C. What is the specific heat capacity of the metal?
2.56 J/g°C
0.391 J/g°C
5.29 J/g°C
3.50 J/g°C
80
Multiple Choice
The specific heat of water is 4.18 J/g°C.
If 980. J of energy is added to 6.20 g of water at 18.0 °C, what is the final temperature of the water?
37.8 °C
-19.8 °C
19.8 °C
55.8 °C
81
Multiple Choice
Calorimeters use insulating materials to:
encourage affordable materials in science experiments in schools.
preserve the heat of the environment outside the Styrofoam cup.
prevent heat from escaping to the environment.
82
Multiple Choice
What does "q" mean?
A measure of heat energy
A change in heat energy
A measure of kinetic energy
A change in temperature
83
Multiple Choice
In order to make a cup of milk coffee at 78°C, 200 g of coffee is mixed up with 40 g of milk. If the original temperature of the coffee is 80°C, find the original temperature of the milk. Assume that there is no heat lost to the surroundings. Given that the specific heat capacity of coffee and milk are 4000 J kg-1 °C-1 and 3900 J kg-1 °C-1 respectively.
10.3°C
20.7°C
45.2°C
67.7°C
84
Multiple Choice
Danny pours some 95°C water into a cup of instant noodles. The final temperature of the mixture is 85°C. If the masses of the hot water and noodles are 500 g and 150 g respectively, find the specific heat capacity of the noodles.
Give that the original temperature of the noodles is 25°C and the specific heat capacity of the water is 4200 J kg-1 °C-1.
500 J kg-1 °C-1
1.08 x 103 J kg-1 °C-1
2.33 x 103 J kg-1 °C-1
6.00 x 103 J kg-1 °C-1
85
Multiple Choice
Find the energy released by a copper block when it is cooled from 100°C to 20°C. Given that the heat capacity of the block is 770 J°C-1.
7.70x104 J
8.61x104 J
6.16x104 J
5.02x104 J
86
Multiple Choice
Water has a specific heat of 4184 J/KgºC. Wood has a specific heat of 1760 J/KgºC. What material needs more energy to raise the temperature 1ºC
Wood
Water
Both are the same
87
Multiple Choice
Which description defines heat?
total thermal energy of an object or substance
change in the temperature of a substance in degrees Celsius
energy transferred between objects at different temperatures
average kinetic energy of the particles that make up a substance
88
Multiple Choice
There are four cups of hot cocoa. The cup sizes (from smallest to largest) are tall, grande, venti, and trenta. The temperature of the cocoa in each cup is 25 degrees celsius. Which cup has the MOST thermal energy?
Large cup (Trenta)
Small cup (Tall)
Venti
Grande
All four are the same temperature so they have same thermal energy
89
Multiple Choice
Which of the following containers of sand has atoms/molecules with the greatest average kinetic energy?
1 kg at 20oC
2 kg at 20oC
4 kg at 20oC
none has a greater amount because all have the same average kinetic energy
90
Poll
After answering all the questions in the mini-quizzes, how ready are you to take the Midterm Exam next week?
100% ready
Needs only a little review
Needs some review, but confident that I'll pass
Who needs to review? Not me!
Needs to study all over again!!!
91
Thank you and God bless!
Thermodynamics:
Midterm Review
by Tessa Noora
Show answer
Auto Play
Slide 1 / 91
SLIDE
Similar Resources on Wayground

83 questions
STUDENT NOTES THE CONSTITUTION PART 1
Presentation
•
12th Grade

84 questions
105 Planning and overseeing PPT 1
Presentation
•
KG - University

87 questions
WWI DC
Presentation
•
University

87 questions
Anemias Nutricionales
Presentation
•
University

86 questions
20k người lạ
Presentation
•
KG - University

87 questions
Sky Science
Presentation
•
KG - University

87 questions
Topic 5 & 6: Design Theory Part I
Presentation
•
University

88 questions
BIO110 Chapter 12
Presentation
•
University
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

20 questions
Guess The App
Quiz
•
KG - Professional Dev...

11 questions
NFL Football logos
Quiz
•
KG - Professional Dev...

19 questions
Minecraft
Quiz
•
6th Grade - Professio...

40 questions
8th Grade Math Review
Quiz
•
8th Grade - University

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...

10 questions
Would you rather...
Quiz
•
KG - University

40 questions
Flags of the World
Quiz
•
KG - Professional Dev...

14 questions
Superhero
Quiz
•
1st Grade - University