

Electrode and Cell Potentials
Presentation
•
Chemistry
•
University
•
Easy

Luis Bello
Used 9+ times
FREE Resource
10 Slides • 16 Questions
1
Electrode and Cell Potentials
by Luis Bello
2
Previous Class
Galvanic Cells
3

4
Multiple Choice
In a Galvanic Cell, the anode is...
positive
negative
5
Multiple Choice
In a Galvanic Cell, the cathode is...
positive
negative
6
Multiple Choice
Oxidation occurs at the...
anode
cathode
7
Multiple Choice
Reduction occurs at the...
anode
cathode
8
Multiple Choice
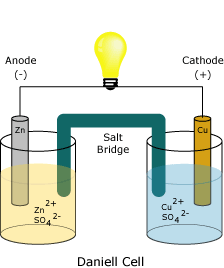
In the above Galvanic Cell, which half cell is undergoing reduction?
Cu | Cu2+
Zn | Zn2+
9
Multiple Choice
A certain galvanic cell has for its spontaneous cell reaction: Zn + HgO → ZnO + Hg
Which is the half-reaction occurring at the cathode?
HgO + 2 e- → Hg + O2-
Hg → Hg2+ + 2 e-
Zn → Zn2+ + 2 e-
ZnO + 2 e- → Zn
10
Multiple Choice

Which of the following deductions about this cell is correct?
11
Multiple Choice

The product(s) of the half-reaction occurring in half-cell 1 is (are)
12
Multiple Choice

For this cell, the overall equation will be
13
Learning Objetives
Describe and relate the definitions of electrode and cell potentials
Interpret electrode potentials in terms of relative oxidant and reductant strengths
Calculate cell potentials and predict redox spontaneity using standard electrode potentials
14
15
The electrode potential
In electrochemistry, the electrode potential is the electromotive force of a galvanic cell built from a standard reference electrode and another electrode to be characterized.
By convention, the reference electrode is the standard hydrogen electrode (SHE). It is defined to have a potential of zero volts.
16

17
The SHE electrode
The SHE electrode can be connected to any other electrode by a salt bridge to form a cell.
If the second electrode is also at standard conditions, then the measured cell potential is called the standard electrode potential for the electrode.
The standard electrode potential for the SHE is zero, by definition.
18
Standard electrode potentials
Standard electrode potentials reflect the relative oxidizing strength of the half-reaction reactant, with stronger oxidants exhibiting larger (more positive) E°X values. Tabulations of standard electrode potentials may be used to compute standard cell potentials, E°cell, for many redox reactions.
19

20
Multiple Choice

-0.41V
-0.27 V
+0.27 V
+0.41 V
21
Multiple Choice

The relevant standard electrode potentials for this cell are shown below.
Cr3+(aq) + 3e- ⇌ Cr(s) E= − 0.74 V
Ag+(aq) + e− ⇌ Ag(s) E = + 0.80 V
Calculate the emf of the cell shown in the diagram.
+1.54 V
-1.54 V
+ 0.06 V
- 0.06 V
22
Multiple Choice

Which species will oxidise Fe2+(aq) to Fe3+(aq)?
Br2 (aq)
Cl-(aq)
I2 (aq)
Mn2+(aq)
23
Multiple Choice

Calculate the emf for the reaction between MnO4– ions and Br– ions in acidic solution.
-0.42 V
+0.42 V
+2.60 V
-2.60 V
24
Multiple Choice

The standard electrode potentials of two half reactions are shown.
Which of the following processes is thermodynamically favourable? The reaction of
Co2+ with Cl2 to form Cl−
Co2+ with Cl− to form Cl2
Co3+ with Cl2 to form Cl−
Co3+ with Cl− to form Cl2
25
Multiple Choice
Calculate the cell potential of a galvanic cell that uses the two half equation below
Sn2+ + 2e- --> Sn4+ E0 = -0.14 V
Mg2+ + 2e- --> Mg E0 = -2.37 V
-2.23 V
-2.51 V
+ 2.23 V
26
Multiple Choice
Using the following standard reduction potentials
Fe3+(aq) + e --> Fe2+(aq) + e E0 = +0.77V
Ni2+ + 2e --> Ni E0 = -0.26V
Ni + 2 Fe3+ --> Ni2+ + 2 Fe2+
Calculate the standard cell potential for the above galvanic cell
-1.8V
+1.8V
+0.51 V
Electrode and Cell Potentials
by Luis Bello
Show answer
Auto Play
Slide 1 / 26
SLIDE
Similar Resources on Wayground

22 questions
PARTICIPIAL ADJECTIVES
Presentation
•
University

20 questions
IDIOMS LESSON
Presentation
•
12th Grade

20 questions
Origins of Russia
Presentation
•
KG

20 questions
Gerund vs. infinitive
Presentation
•
University

23 questions
Waves Electrons
Presentation
•
University

19 questions
INGLES 1 - Sesión 2 Greetings and Farewells
Presentation
•
University

19 questions
General Interview Question
Presentation
•
University

20 questions
Presentation tips
Presentation
•
University
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

55 questions
Post Malone Addtion (Tres)
Quiz
•
12th Grade - University

20 questions
Disney Trivia
Quiz
•
University

50 questions
AP Biology Exam Review 2017
Quiz
•
11th Grade - University

24 questions
5th Grade Math EOG Review
Quiz
•
KG - University

12 questions
Star Wars Trivia - Easy
Quiz
•
KG - University

215 questions
8th Physical Science GA Milestones Review
Quiz
•
KG - University

40 questions
Famous Logos
Quiz
•
7th Grade - University

14 questions
(5-3) 710 Mean, Median, Mode & Range Quick Check
Quiz
•
6th Grade - University