

Extraction of Metals
Presentation
•
Chemistry
•
9th - 10th Grade
•
Medium
Angela Toh
Used 11+ times
FREE Resource
14 Slides • 49 Questions
1
By Angela Toh
S4 Pure Chemistry - Extraction of Metals
2

3
Multiple Choice

In this reactivity series, which of the following is the most reactive element?
Gold
Calcium
Zinc
Potassium
4
Multiple Choice
Metal A is more reactive than Metal B. The following displacement reaction takes place.
Metal A + Metal B in compound ---> Metal B + Metal A in compound.
True or False.
True
False
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
The reactivity series of metals is applied in the ............. of the metals from their ore.
Melting
Extraction
Freezing
Crystalization
8
Multiple Choice
Use the following observations to select the correct reactivity series (highest to lowest).
calcium + water: slow
sodium + water: fast
calcium + dilute acid: very fast
gold + dilute acid: no reaction
aluminium + dilute acid: fast
calcium, sodium, aluminium, gold
sodium, calcium, aluminium, gold
sodium, aluminium, calcium, gold
gold, aluminium, calcium, sodium
9
Multiple Select
Many metals are extracted from their ores by heating them with carbon in huge furnaces. Which metal(s) cannot be extracted using this method?
Aluminum
Calcium
Zinc
Copper
10
Multiple Choice
11
Multiple Choice
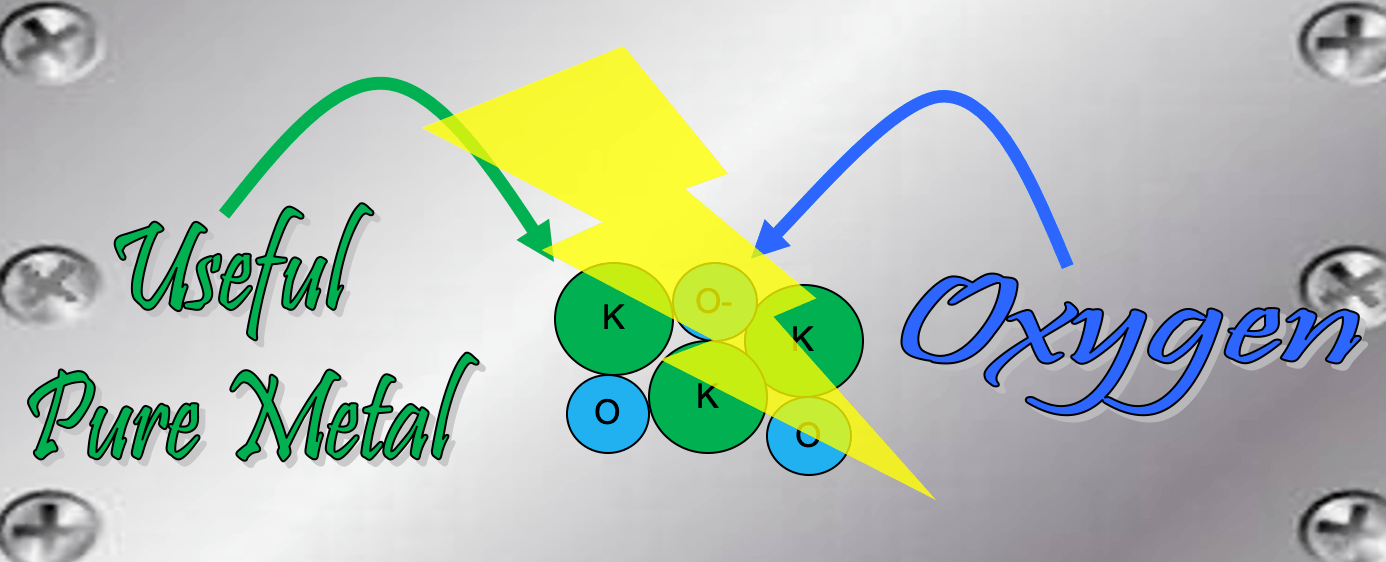
What is the name of the process that uses electricity to separate the elements in a compound?
reduction
electrolysis
extraction
mining
12
Multiple Select
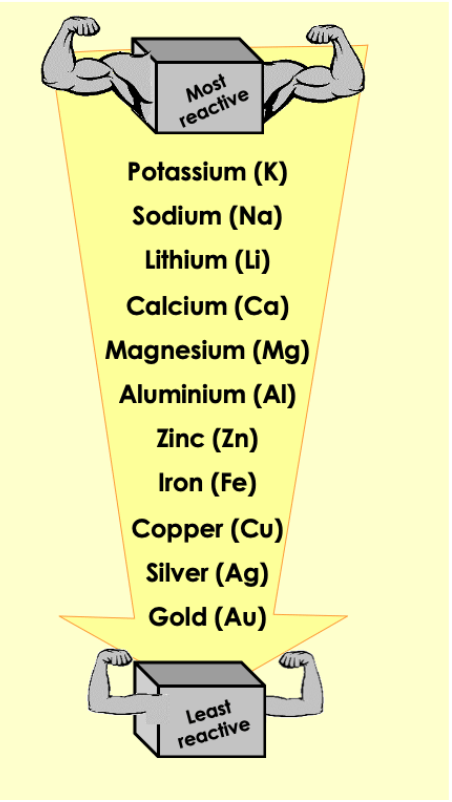
Which of these elements are unlikely to be found uncombined in the earth's crust?
gold
potassium
aluminium
silver
13
Multiple Choice
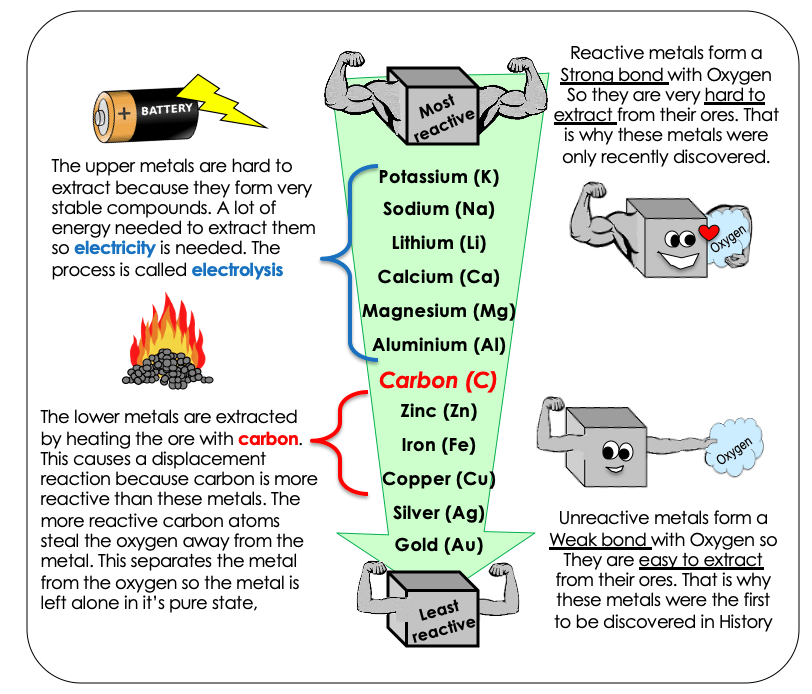
Which element cannot be extracted from its ore by reduction with carbon?
iron
aluminium
copper
zinc
14
Multiple Choice
15
Multiple Choice
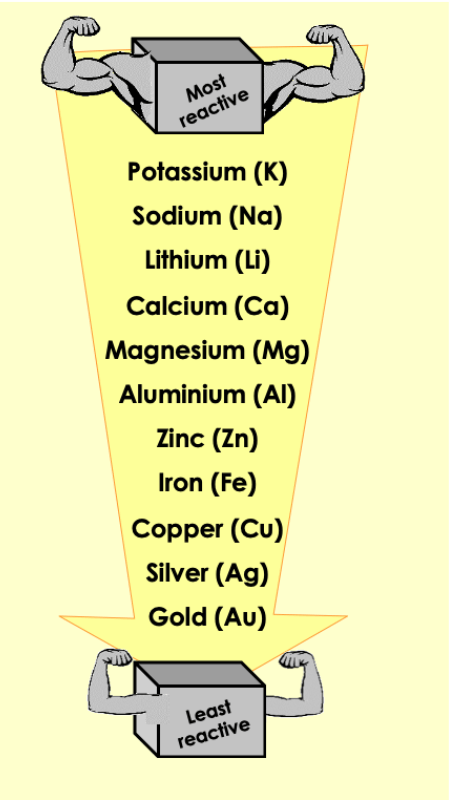
How would you best extract iron from haematite (iron ore)?
reduction by carbon
electrolysis
decomposition
oxidation
16

17

18

19

20
Multiple Choice
21
Multiple Choice

What is the purpose of adding iron ore?
As a source of iron
To remove impurities
To react with coke to form carbon monoxide
To reduce iron oxide to iron
22
Multiple Choice

What is the purpose of adding coke?
As a source of iron
To remove impurities
To react with coke to form carbon monoxide
To reduce iron oxide to iron
23
Multiple Choice

What is the purpose of adding limestone?
As a source of iron
To remove impurities
To react with coke to form carbon monoxide
To reduce iron oxide to iron
24
Multiple Choice

What is the purpose of adding hot air?
As a source of iron
To remove impurities
To react with coke to form carbon monoxide
To reduce iron oxide to iron
25
Multiple Choice
26
Multiple Choice
27
Multiple Select

Which are the correct 4 Raw Materials used in a blast furnace?
Hot Air
Coke
Limestone
Iron Ore
Cryolite
28
Multiple Choice

Which gas reduces iron ore?
CO
CO2
O2
C
29
Multiple Choice

What is the name for the waste products formed in the Blast Furnace?
Slag
Debris
Haematite
Cryolite
30
Multiple Select

Which 2 equations correctly show the formation of carbon monoxide in the blast furnace?
C(s) + O2(g) -----> CO2(g)
2C(s) + O2(g) -----> 2CO2(g)
C(s) + CO2(g) -----> CO(g)
C(s) + CO2(g) -----> 2CO(g)
31
Multiple Select

Which 2 equations correctly show the formation of iron in the blast furnace?
Fe2O3(s) + 3CO(g) -----> 3CO2(g) + 2Fe(s)
2Fe2O3(s) + 6CO(g) -----> 6CO2(g) + 2Fe(s)
Fe2O3(s) + 3C(s) -----> 3CO2(g) + 2Fe(s)
Fe2O3(s) + 3C(s) -----> 3CO(g) + 2Fe(s)
32
Multiple Select

Which 2 equations correctly show the formation of slag in the blast furnace?
CaCO3(s) -----> CO2(g) + CaO(s)
CaCO3(s) -----> CO(g) + CaO2(s)
CaO2(s) + SiO(s) -----> CaSiO3(s)
CaO(s) + SiO2(s) -----> CaSiO3(s)
33

34

35
Multiple Choice
Steel is ... than iron
stronger
weaker
36
Multiple Choice

In order to make steel stronger, ... is added
carbon
coal
37
Multiple Choice

Steel which doesn't rust is called...
stainless steel
rust-free steel
38
Multiple Choice

steel can be used to produce wires because it is...
brittle
ductile
39
Multiple Choice
Stainless steel is so called because of its -------------------
High strength
High corrosion resistance
High ductility
Brittleness
40
Multiple Choice
Which is the primary element used for making stainless steel alloy?
Chromium
Zirconium
Vanadium
Indium
41
Multiple Choice
Which conditions are necessary to make mild steel from iron?
add calcium oxide and blow oxygen through it
heat with calcium oxide
heat with carbon and limestone
heat with nickel and chromium
42

43

44
Multiple Choice
Which metals rusts?
Copper
manganese
iron
potassium
45
Multiple Choice

A rusting bicycle
physical change
chemical change
46
Multiple Choice
What environmental element assists rusting?
nitrogen
oxygen
carbon
hydrogen
47
Multiple Choice

What conditions are required for rusting?
dry cold conditions
dry hot conditions
hot damp conditions
48
Multiple Choice

Which of the test tube(s) in Diagram will iron nail rust?
X only
Y only
X and Z only
Y and Z only
49
Multiple Choice

Which nail rusts first?
Nail in test tube A
Nail in test tube C
Nail in test tube E
Nail in test tube F
50
Multiple Choice
Which metal when attached to an iron nail will cause the iron nail to corrode fastest?
Magnesium
Silver
Lead
Tin
51

52

53
Multiple Choice
What prevents Rusting
Painting , Oiling , Galvinising
Washing with water
54
Multiple Choice

How do you prevent this food can from rusting?
painting
apply oil or grease
coating with plastic
plating with a less reactive metal
55
Multiple Choice
Which of the following does not prevent rusting of iron?
Painting , Oiling , Galvinising
Attach to a piece of copper
connect to a piece of magnesium with wire
cover entirely with copper
56

57

58
Multiple Choice
What is recycling?
putting an object into a recycling bin
making something new out of resources that have already been used
using an object again, either for its original purpose or a different purpose
59
Multiple Choice
How are metals recycled?
They are washed and then broken into small pieces. The pieces are then melted and made into new things.
They are washed in soapy water. It is then mixed and rolled out before being left to dry.
60
Multiple Select
Which two of these resources are finite?
fossil fuels
oxygen
metal ore
fresh water
61
Multiple Choice
Why is recycling so important?
The world's resources are unlimited.
The world's resources are unprotected.
The world's resources are illegitimate.
The world's resources are limited.
62
Multiple Choice

Is it recyclable?
Yes
No
63
Multiple Select
What are the advantages of recycling metals? Select all correct options.
Conserves metals as they are finite resources.
Reduces waste and problems in its disposal.
Takes time to collect and sort waste metals.
Reduces pollution due to extraction activities.
By Angela Toh
S4 Pure Chemistry - Extraction of Metals
Show answer
Auto Play
Slide 1 / 63
SLIDE
Similar Resources on Wayground

61 questions
Inv 1. Lesson 2 and 3 - Earth's Interior and Plate Tectonics
Presentation
•
10th Grade

59 questions
Matter and Energy
Presentation
•
9th - 11th Grade

57 questions
Skimming and Scanning
Presentation
•
9th Grade

60 questions
Повторення матеріалу 8 класу. Урок №3-4
Presentation
•
9th - 11th Grade

58 questions
Latin America
Presentation
•
9th Grade

54 questions
Part 4 EOC Review
Presentation
•
10th Grade

56 questions
Biology EOC Review
Presentation
•
9th - 10th Grade

56 questions
Naming Ionic Compounds - Type I, II, and III
Presentation
•
10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade

34 questions
Nuclear Chemistry Review
Quiz
•
9th - 12th Grade