

1.1 a: Separation Techniques
Presentation
•
Science, Chemistry
•
8th - 11th Grade
•
Easy
Erin Hannan
Used 35+ times
FREE Resource
11 Slides • 6 Questions
1
Separation Techniques
Year 12
2
What we will cover
physical properties of matter
separation techniques
flow charts
3
Physical properties of matter
We use physical properties to separate mixtures, chemical properties are used in relation to chemical changes/reactions
Homogenous and heterogenous mixtures
Colour (not useful)
Magnetism
Particle size
Melting and boiling points
Density
4
Types of Mixtures

5
Physical properties of matter
Particle size
not useful in identifying substances, but useful for separating
use sieves and/or filter paper
Melting and boiling points
melting point = lowest temp for a solid to change into a liquid
boiling point = lowest temp for liquid to change into a gas
use as a test for purity (pure = 'sharp' melting point)
Density
either by measurement or by displacement
useful for identification
6
Separation Techniques
Filtration
using filter paper
based on particle size
Solubility
using solubility to separate two solids
i.e. sand from salt. Dissolve in water, filter off the sand.


7
Separation Techniques
Separation based on boiling points
Dissolved solid from solution
evaporating the liquid until only solid is left, as liquid has much lower boiling point than the solid
Distillation
solution is boiled, then vapour is condensed back into a liquid and therefore separated from the mixture
used to separate liquids with different boiling points
FRACTIONAL distillation is used when the boiling points are closer together (less then 40 degrees)


8
Separation techniques
Separations based on density
Decanting
when a sediment is formed, the liquid can be decanted off
Separating funnel
when two immiscible liquids (when mixed do not form a homogenous liquid, i.e. oil and water), one will settle on top of the other and then can be separated out via the funnel

9
Separation techniques (less common)
Fractional evaporation - separating gases based on their different boiling points
Magnetic separation - exploiting magnetism in different substances
Centrifuge - density of particles in a liquid


10
Separation Flowchart
It is important to represent your flowchart of separation correctly in your answers. Indicating what substances you have and what is being separated out.
Use diamonds for a separation technique, and rectangles for a substance that is being separated out by that technique, or a substance that is being added.
E.g. Given a mixture of dry salt and salt, outline the procedure to obtain dry sand and dry salt.

11
Multiple Choice
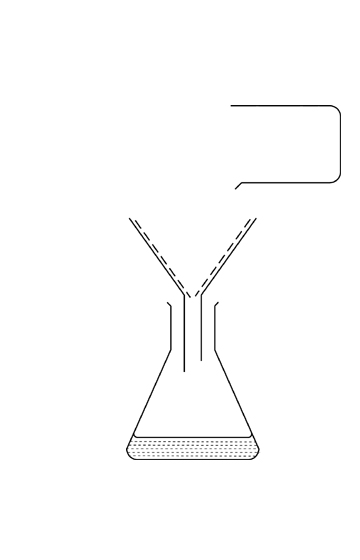
What is the name of this separation technique?
distillation
filtration
evaporation
chromatography
12
Multiple Choice
What is the physical property on which fractional distillation depends?
different boiling points
different melting points
different solubilities
different densities
13
Multiple Choice
During filtration, the solid that is trapped in the filter paper is called ..A.. and the liquid that has passed through the filter paper is called ..B..
A: residue B: filtrate
A: residue B: filterate
A: filtrate B: residue
A: filterate B: residue
14
Multiple Choice
15
Multiple Choice
16
Multiple Choice
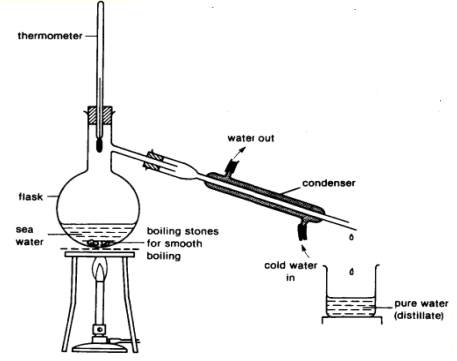
What is the name of this separation technique?
Simple distillation
Fractional distillation
Boiling
Evaporation
17
Next content
gravimetric analysis
nomenclature review
Separation Techniques
Year 12
Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

13 questions
Biochemical Cycle
Presentation
•
8th - 12th Grade

13 questions
Carbon Moving in the Spheres of the Earth
Presentation
•
8th - 12th Grade

17 questions
Unit 7: Heat Transfer
Presentation
•
9th - 12th Grade

12 questions
Writing a Line in Slope Intercept Form Lesson
Presentation
•
8th - 11th Grade

13 questions
6.4/6.5 Triangle Similarity by AA, SSS, and SAS
Presentation
•
8th - 11th Grade

11 questions
Changes in Matter Review
Presentation
•
6th - 8th Grade

16 questions
Geosphere Refresher
Presentation
•
9th - 12th Grade

13 questions
Significance of Carbon
Presentation
•
8th - 10th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Science

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

20 questions
Earth Day Trivia
Quiz
•
9th - 12th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
Earth Day
Quiz
•
3rd - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

100 questions
GMAS Science Review
Quiz
•
8th Grade