

1.1 c & 1.2 a: Periodic Trends/Isotopes/Unstable Isotopes
Presentation
•
Science, Chemistry
•
10th - 12th Grade
•
Easy
Erin Hannan
Used 11+ times
FREE Resource
17 Slides • 11 Questions
1
Periodic Trends and Properties, and Isotopes
Year 11
2
What we will cover
1.1 c : classify the elements based on their properties and position in the periodic table through their:
– physical properties
– chemical properties
1.2 a: investigate the basic structure of stable and unstable isotopes by examining:
– their position in the periodic table
– the distribution of electrons, protons and neutrons in the atom
– representation of the symbol, atomic number and mass number (nucleon number)
3
Elements, compounds, mixtures etc.
Elements: substance consisting of one type of atom. E.g. diamond
Compound: pure substance consisting of two or more elements. E.g. water
Mixture: substance containing two or more substances that aren't chemically combined.
heterogenous: visibly different substances or phases. E.g. soil, ice in water
homogenous: uniform in appearance and composition . E.g. cordial

4
The Periodic Table


5
there are changes in properties from group to group, across a period, and down a group
the PT can help us determine the 'general' properties of an element based on others we know
i.e. if we are unsure what bromine may react with commonly, we can look to other elements in that group (like chlorine) and assume they will make similar compounds
6
What can we determine from the PT
Metallic character
increases as we go down a group , but decreases as we go across a period
shiny, good conductors, malleable, higher melting point
number of subatomic particles
other trends like atomic radius, ionisation energy, electronegativity

7
Multiple Choice

in alphabetic order
simular physical & chemical properties
their symbols
Just simular physical properties
8
Multiple Choice

Horizontal Rows
Vertical Columns
9
Multiple Choice
The formula used to calculate the number of neutrons is:
Mass Number - Atomic Number
Protons + Electrons
Atomic Number - Mass Number
Protons x2
10
Basic atomic structure
small dense positively charged nucleus, with protons and neutrons
surrounded by an electron cloud, which we represent in shells/orbitals/energy levels
atomic number, Z = number of protons = number of electrons (in a neutral atom)
mass number, A = number of protons + number of neutrons
next lesson: energy levels

11
Multiple Choice
Which of the following particles is located outside of nucleus of the atom?
proton
neutron
electron
12
Multiple Select
Which of the following particles are responsible for most of the mass of an atom? (You may choose more than one answer.)
proton
neutron
electron
13
Multiple Select
Which of the following particles compete to establish the charge of an atom?
proton
neutron
electron
14
Isotopes
differing numbers of neutrons in the nucleus
isotopes of the same element will have the same atomic number, but different mass numbers
named by their mass number
e.g. chlorine-35 and chlorine-37 (contains an extra two neutrons)


15
Relative abundance of isotopes
the percentage of that isotope in the naturally occurring element
for example:

16
Calculating relative atomic mass from isotopic data

17
Multiple Choice
204
206
207
208
18
Unstable isotopes
when an isotopes emits radiation it is unstable
instability comes from a an excess of protons or neutrons
this emission is actually from the nucleus, and there are three different types of emission
He nucleus = two protons + two neutrons


19
Alpha emitting isotopes
Emits a helium nucleus, therefore loses 2 protons and 2 neutrons
For example, a common uranium isotope is
Which has 92 protons and 146 neutrons.
If it loses a He nucleus, it will have 90 protons and 144 neutrons, and will transform into:


20
Beta emitting isotopes
Emits an electron, but from the nucleus.
Confused?
A neutron is decomposed into a proton and an electron, and then the electron is ejected from the nucleus.

For example: cobalt-60 (radiation therapy)

21
Multiple Choice
42He
0-1e
0+1 e
10n
22
Multiple Choice
6027Co = ___ + 0-1e
5625Mn
6028Ni
5823V
5927Co
23
Gamma emitting isotopes
Gamma radiation (on the EM spectrum) is emitted as waves
Generally occurs as a secondary form of decay after alpha or beta radiation, as the daughter particles are usually in an excited state
They need to release some energy to become more stable
The particles remain the same, there is no loss of particles, charge, or mass
Common usage: radiotherapy, sterilisation of equipment
24
Multiple Choice
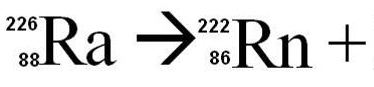
alpha
beta
gamma
none
25
Multiple Choice
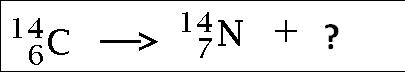
alpha particle
beta particle
gamma particle
neutron
26
Why are some isotopes unstable?
Generally elements with larger nuclei (larger than AN 83) will be unstable, as the forces keeping the nuclei intact are not stable. Or, with smaller nuclei, the ratio of neutrons to protons is unstable.
Two forces keeping the nucleus together:
electrostatic force between positively charge protons
mass-mass attraction between all particles (like a tiny gravitational force)
If n:p ratio is too high = beta emitter
If n:p ratio is too low = alpha emitter
Half-life of isotopes varies (time required for half the atoms of a sample to undergo radioactive decay). E.g. iodine 131 is 8 days, and uranium-238 is 4.5 x 10^9 years
27
Penetrating power of radiation types

28
What's next?
energy levels in atoms
electron configuration
stability of atoms
atomic emission
Periodic Trends and Properties, and Isotopes
Year 11
Show answer
Auto Play
Slide 1 / 28
SLIDE
Similar Resources on Wayground

22 questions
Ion Formation and Ionic Bonding
Presentation
•
10th - 12th Grade

19 questions
Layers of the Earth
Presentation
•
10th - 12th Grade

21 questions
Atmosphere Refresher
Presentation
•
9th - 12th Grade

20 questions
Cell Division
Presentation
•
9th - 12th Grade

20 questions
Atomic Structure Review
Presentation
•
9th - 12th Grade

20 questions
Water, Solubility, Solutions, Molarity
Presentation
•
9th - 12th Grade

23 questions
Ecosystems and Biomes
Presentation
•
11th - 12th Grade

21 questions
Significant Figure and Scientific Notation Review
Presentation
•
10th - 11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

20 questions
Frog Dissection Quiz
Quiz
•
11th Grade

20 questions
Final Review Thermochemistry
Quiz
•
9th - 12th Grade

10 questions
Exploring Animal Adaptations: Physical, Behavioral, and Life Cycle Changes
Interactive video
•
6th - 10th Grade

11 questions
Explore Homeostasis and Feedback Mechanisms
Quiz
•
9th - 12th Grade

99 questions
PS Semester 2 Review
Quiz
•
9th - 12th Grade