

Wednesday's class 2-2
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Standards-aligned
Connor Boyd
Used 4+ times
FREE Resource
17 Slides • 8 Questions
1
Wednesday's class 2-2
By Connor Boyd
2
Announcements
Quiz moved to Friday
Activity planned moved to next Friday
Some text here about the topic of discussion
3
Atomic structure
Protons, electrons, neutrons, energy levels
Periodic table
Isotopes, atomic mass, atomic number, electronegativity , empirical vs molecular formula
Molecular geometry
Ionic compounds
Writing chemical formulas and characteristics
Covalent compounds
Polarity and characteristics
Topics
Quiz Friday will cover..
4
Today we will cover
Empirical formulas
Isotopes
Atomic mass
Some text here about the topic of discussion
5
MOLECULAR FORMULA
The Octet Rule is important in predicting the chemical formula of simple binary covalent molecular compounds.

6
Multiple Choice
What is the empirical formula for C4H6?
CH
CH3
C2H3
C4H6
7
Multiple Choice
What is the empirical formula for N2S3?
NS
N3S2
N2S3
N1S1.5
8
Multiple Choice
What is the empirical formula of a substance with the molecular formula C2H4?
C2H2
C2H2
C1H1
CH2
9
Atomic Mass and Mass Number
Module 3 - Physical Science

10
Atomic Number, Atomic Mass and Isotopes

11

12

13


14

15
Multiple Choice

How many neutrons are there in one atom of magnesium?
12
6
24
16
Multiple Choice
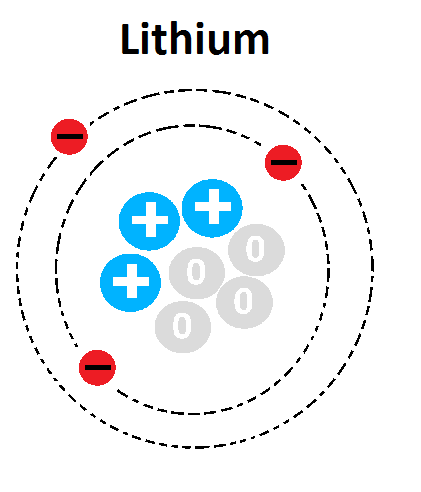
17
Multiple Choice
What is the atomic mass defined as?
18
Review
Atomic Mass = Protons + Neutrons
We round the atomic mass to the nearest whole number, 4
Since we already determined that Helium has two protons, that means: 4=2+Neutrons
There must be 2 Neutrons in one atom of Helium

19

20
Isotopes
atoms with the SAME number of PROTONS but DIFFERENT number of NEUTRONS
21
Carbon 12
Consider the Atom Carbon, the atomic number tells us the number of protons.
How many protons in Carbon 12?

22
Multiple Choice
What is the correct definition for isotopes?
atoms with the same number of protons and neutrons
Atoms with the same number of protons but different number of neutrons
Atoms with the same number of neutrons but different number of protons
Atoms with no mass
23
Lithium isotopes
What do you notice with these 3 isotopes of Lithium?

24
Multiple Choice

What is the difference between these 3 Lithium isotopes?
no difference
they have the same atomic number
they have the same mass number and atomic number
they have different mass numbers
25
Today's Concepts
Introduction to empirical formula, atomic mass and Isotopes
If you are do not fully understand any of these concepts, ask questions!
On the back table there is practice problems
Some text here about the topic of discussion
Wednesday's class 2-2
By Connor Boyd
Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

20 questions
The Structure of An Atom
Presentation
•
11th Grade

21 questions
Isotopes and Avg. Atomic Mass Notes
Presentation
•
9th - 12th Grade

19 questions
Review Periodic Table
Presentation
•
10th - 12th Grade

21 questions
Mass and The Mole Part 1
Presentation
•
10th - 12th Grade

19 questions
Ionic Bonds
Presentation
•
9th - 12th Grade

20 questions
Average Isotope Mass
Presentation
•
9th - 12th Grade

21 questions
IONIC BONDING
Presentation
•
9th - 12th Grade

21 questions
Electricity: Circuits
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade