

Ch. 6 L. 2 Chemical Reactions
Presentation
•
Science, Biology, Chemistry
•
7th - 12th Grade
•
Easy
D Olguin
Used 1+ times
FREE Resource
25 Slides • 35 Questions
1
Ch. 6 L. 2 Chemical Reactions
By D Olg
2

3

4

5

6
Multiple Choice

7
Multiple Choice

8
Multiple Choice

9
Multiple Choice

10
Multiple Choice
Which of the following could indicate that a chemical change took place?
Change in color
Production of Energy
Formation of Gas
All are correct
11
Multiple Choice
The difference between a Physical change and a Chemical change is…
Physical change is only a change to the outside appearance.
Chemical change is when matter is changed into something new.
Atoms are rearranged in chemical changes & not in physical.
All are correct.
12
Multiple Choice
What does a chemical change result in?
A change in state of the substance (ex. liquid to solid)
A change in texture of the substance.
A change in the way atoms are bonded to each other.
A change in the appearance of the substance.
13
Multiple Choice
When a new substance is formed with different properties than the original substance it is called a...
Chemical change
Physical change
Change in State
Change in appearance only.
14

15

16

17

18

19
Multiple Choice
During Chemical Reactions, the substances that are used are called?
Precipitate
Corrosion
Reactants
Products
20
Multiple Choice
During Chemical Reactions, the new substances created are called?
Precipitate
Corrosion
Reactants
Products
21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
-->
mean?
28

29

30

31

32

33

34

35

36

37

38
Multiple Choice
39
Multiple Choice
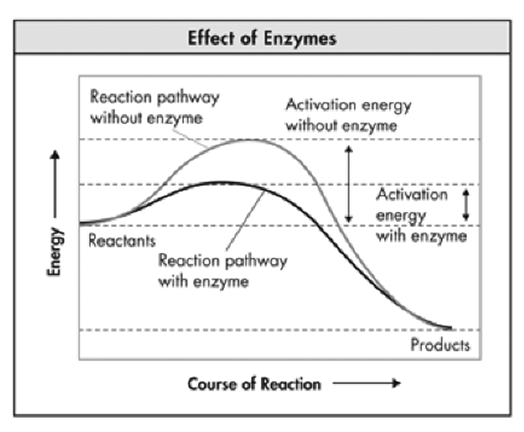
40
Multiple Choice
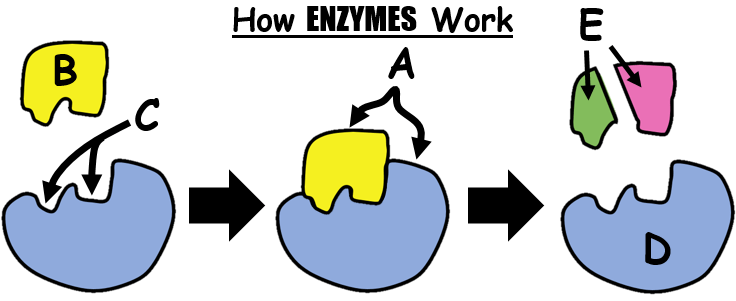
41
Multiple Choice
42
Multiple Choice
43
Multiple Choice
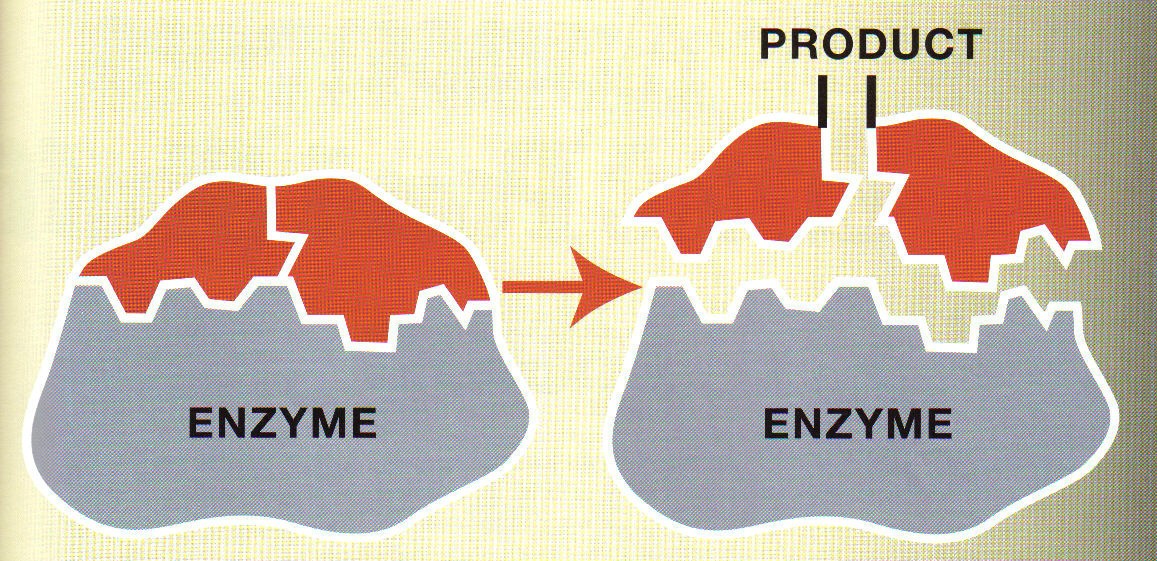
44
Multiple Choice
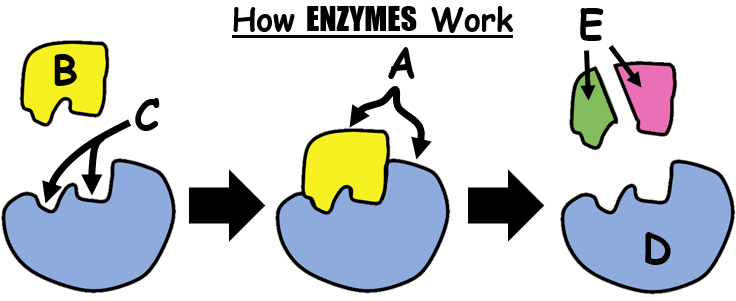
45
Multiple Choice
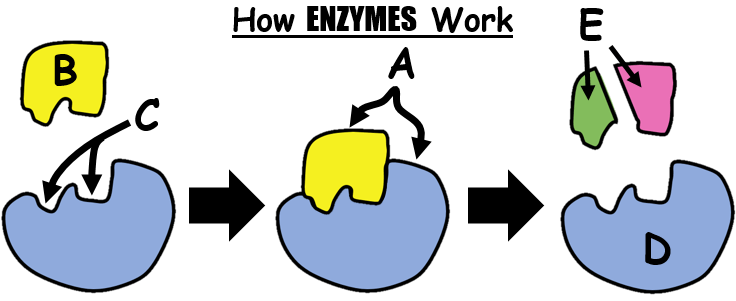
46
Multiple Choice
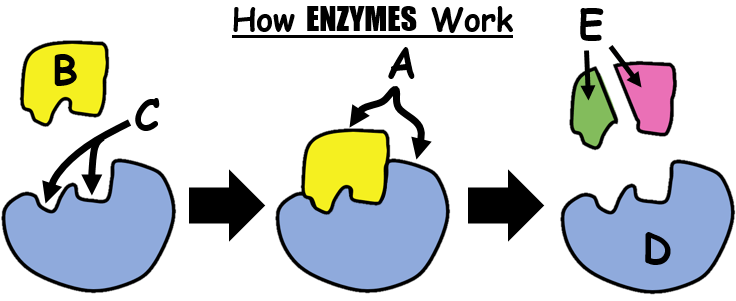
47
Multiple Choice
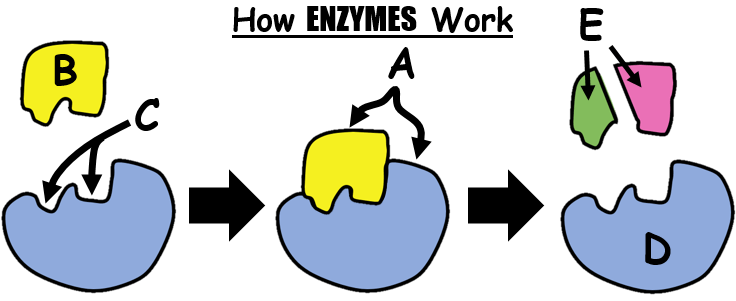
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
What happens when enzymes are heated to a high temperature?
The enzymes die.
The shapes of the enzyme are altered.
The enzymes' amino acid sequence changes.
The enzymes remain the same
51
Multiple Choice
52
Multiple Choice
53
Multiple Choice

54
Multiple Choice
What term is given to a reaction which transfers heat energy to the surroundings?
Endothermic
Reversible
Exothermic
Exotermic
55
Multiple Choice
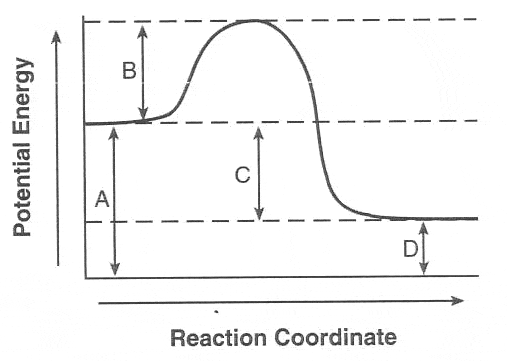
56

57

58

59

60

Ch. 6 L. 2 Chemical Reactions
By D Olg
Show answer
Auto Play
Slide 1 / 60
SLIDE
Similar Resources on Wayground

57 questions
SSA: SPACE SCIENCE (8th GRADE)
Presentation
•
6th Grade - University

51 questions
7th Grade Beginning of the Quarter
Presentation
•
7th Grade - University

55 questions
Cell Structure Review
Presentation
•
7th - 12th Grade

57 questions
Genes and Punnett squares
Presentation
•
9th - 12th Grade

57 questions
Theater Arts
Presentation
•
KG - University

52 questions
Determiners
Presentation
•
7th - 12th Grade

54 questions
Repaso de los Verbos Irregulares en el Presente y Sus Usos
Presentation
•
7th - 12th Grade

57 questions
Lesson | Unit D_Sci8 - Stewardship | Case Study: Sustainable Development and Advanced Battery Technologies
Presentation
•
8th Grade - University
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Abiotic & Biotic Factors
Quiz
•
7th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade