

End of Year Review Physical Science
Presentation
•
Science
•
8th Grade
•
Medium
+8
Standards-aligned
Crystal Winner
Used 3+ times
FREE Resource
53 Slides • 57 Questions
1
End of Year Review Physical Science
This review contains: Periodic Table, Scientific Investigation, Graphs, Cycle, Acids and Bases, Photosynthesis
2
Periodic Table Patterns

3
Periodic Table Patterns

4
Now let's talk groups
groups go up and down on the periodic table
there are 18 groups on the periodic table
BUT only groups 1 and 2 and 13-18 follow any sort of pattern.
the pattern is the group number can help you quickly figure out the number of valence electrons
valence electrons are the electrons on LAST energy level
valence electrons are the ones responsible for bonding- so VERY important!
5
The Group Pattern
The group numbers 1,2 13-18 can help to quickly tell how many valence electrons there are for the elements in the group

6
The Group Pattern
The bolded number at the top of the group tell the number of valence electrons the group has.
Group 1 has 1 valence electron
Group 2 has 2 valence electrons

7
The Group Pattern
The bolded number at the top of the group tell the number of valence electrons the group has.
Group 13 has 3 valence electrons
Group 14 has 4 valence electrons
(see the pattern yet?)
Group 15 has 5
Group 16 has 6
Group 17 has 7
Group 18 (mostly) has 8
*Helium in group 18 is an exception - it only has 2 electrons!

8
Multiple Choice
How many valence electrons do element in group 2 have?
1
2
12
9
Multiple Choice
How many valence electrons do element in group 14 have?
1
4
14
10

11

12

13
Multiple Choice

14
The periodic table is FULL of patterns.
If you know the patterns, you can use it to find information quickly about the elements
The two patterns we are going to review are the periods and the group numbers
15
Multiple Choice
16
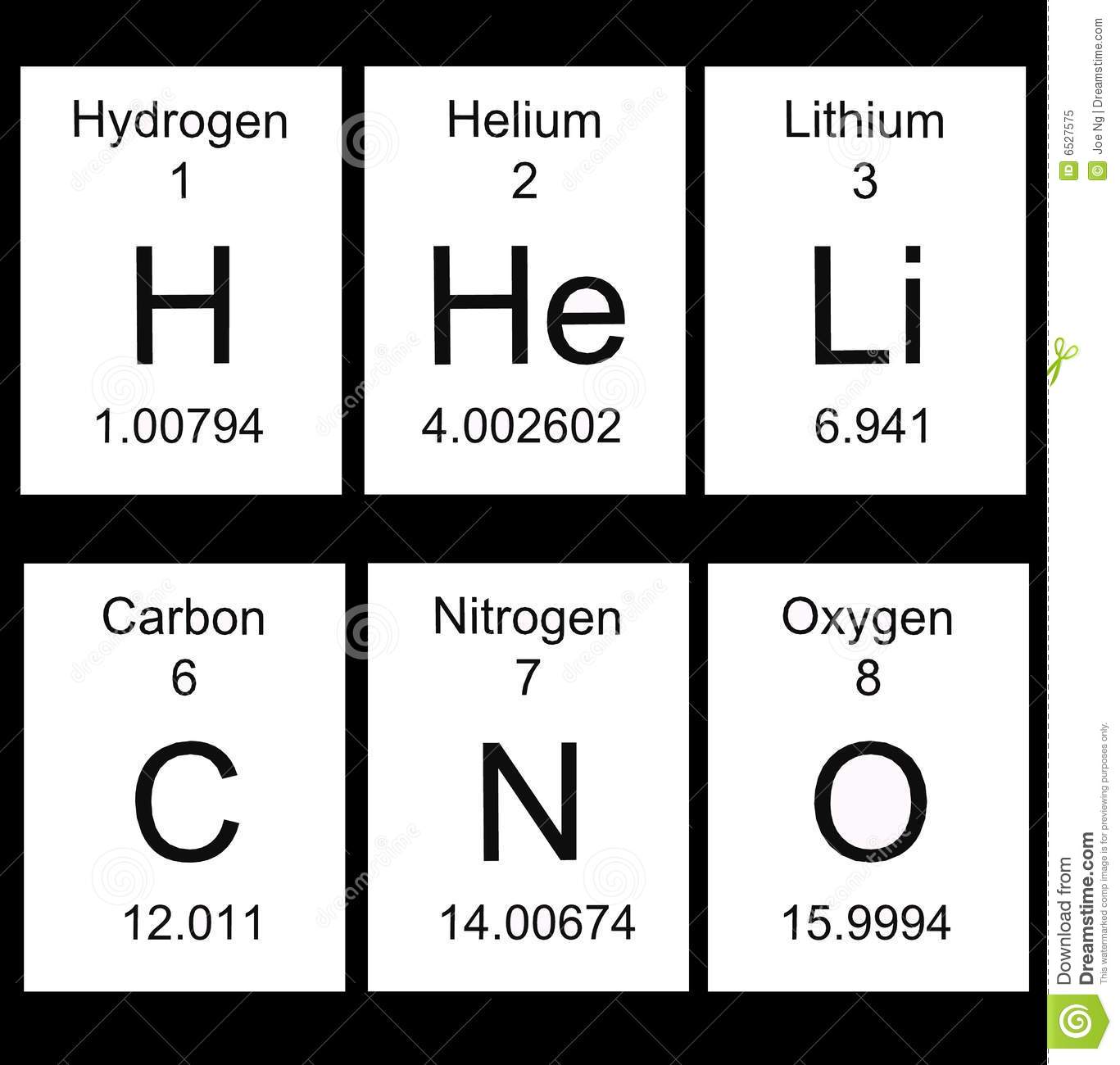
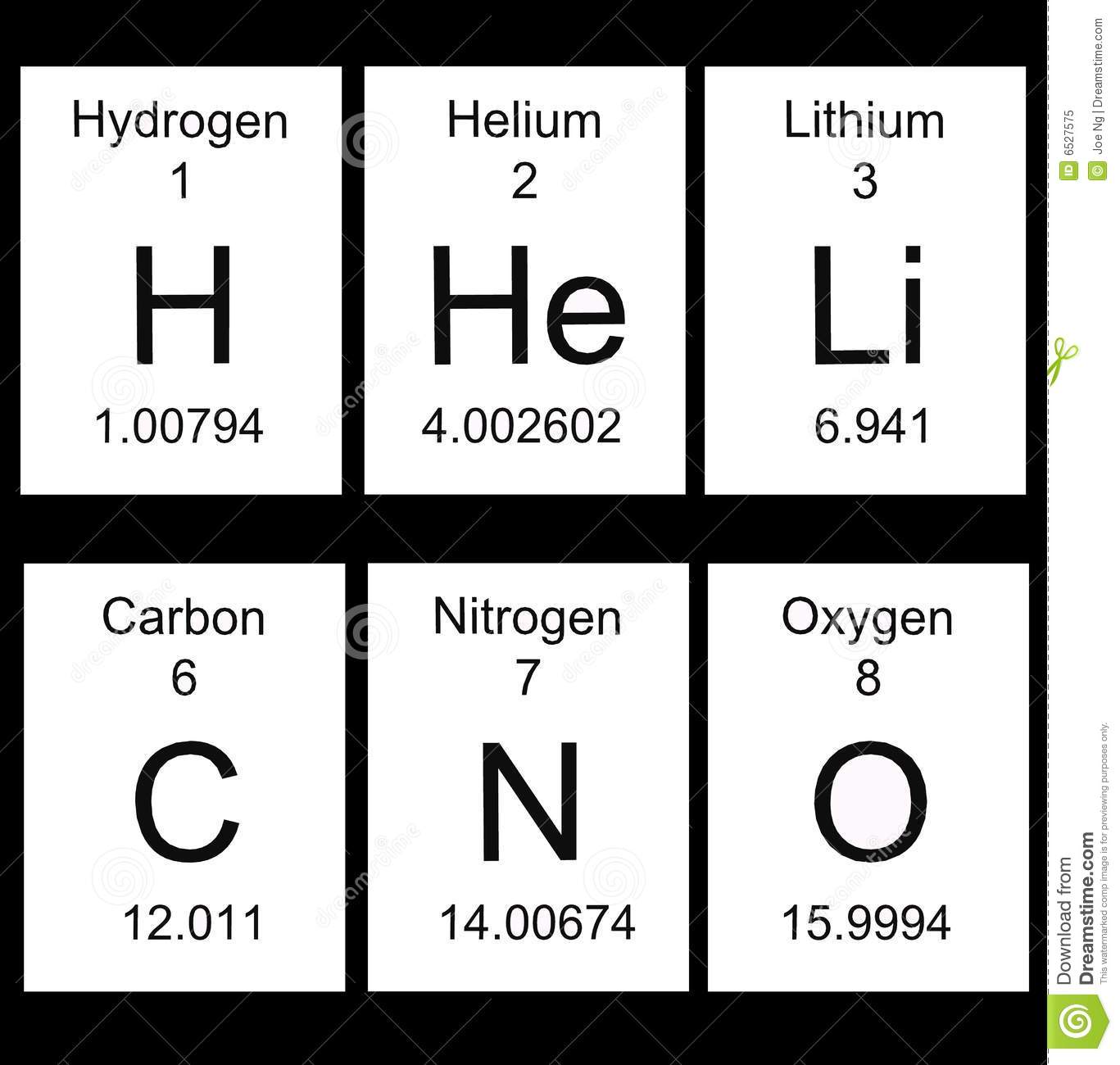
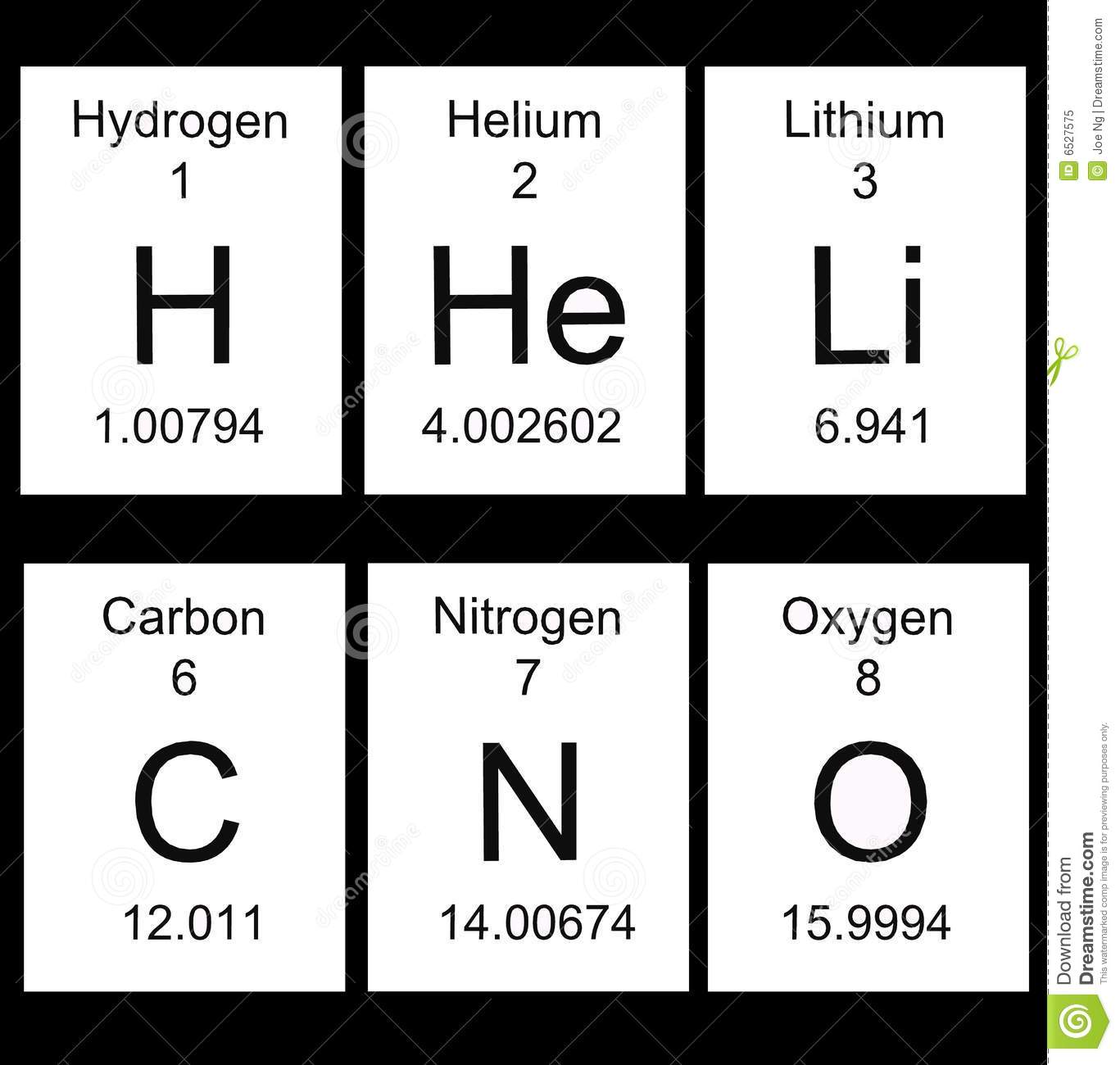
Let's start with the periods
periods are the name for the rows that go across the table
there are 7 rows on the periodic table
the pattern is the elements in a period have the same number of energy levels
as elements get bigger with more electrons, the elements need more space for their electrons.
17
Multiple Choice
18
See?
Energy level 1 with hydrogen and helium have only 1 and 2 electrons
Energy level 2 from Lithium to Neon have two energy levels and have even more electrons
Energy leve3 from sodium to argon have 3 energy levels and still more electrons!

19
Multiple Choice
20
Multiple Choice

How many energy levels does Calcium have in period 4
1
2
3
4
21
Multiple Choice
the positively charged subatomic particle contained in the nucleus of an atom
proton
neutron
electron
matter
22
Multiple Choice

How many energy levels does sodium (Na) have in period 3?
1
2
3
4
23
Multiple Choice
a subatomic particle, contained in the nucleus of an atom, having the same mass as a proton but no electrical charge
proton
neutron
electron
matter
24
Multiple Choice

How many energy levels does Neon have in period 2?
1
2
3
4
25
Multiple Choice
a negatively charged subatomic particle that orbits the nucleus of an atom
proton
neutron
electron
matter
26
Multiple Choice

How many energy levels does helium have in period 1?
1
2
3
4
27
Multiple Choice
a unique number for each element that equals the number of protons in an atom of that element
element name
element symbol
atomic number
atomic mass
28
Okay! So the period number tells us the number of energy levels and element has.
the bigger the number of electrons, the more energy levels it needs
so as you go down the periodic table, the elements have more energy levels
the maximum number of energy levels is 7
29
Multiple Choice

30
Now let's talk groups
groups go up and down on the periodic table
there are 18 groups on the periodic table
BUT only groups 1 and 2 and 13-18 follow any sort of pattern.
the pattern is the group number can help you quickly figure out the number of valence electrons
valence electrons are the electrons on LAST energy level
valence electrons are the ones responsible for bonding- so VERY important!
31
Multiple Choice

32

33

34
Multiple Choice
35
Multiple Choice
How many valence electrons do element in group 2 have?
1
2
12
36
Multiple Choice
37
Multiple Choice
How many valence electrons do element in group 14 have?
1
4
14
38
Multiple Choice
39
Multiple Choice
How many valence electrons do element in group 18 have?
1
8, except Helium only has 2
18
40
Multiple Choice

This is the chemical formula for a molecule of...
Oxygen
Carbon dioxide
Glucose
Water
41
Multiple Choice
Where in a plant cell does photosynthesis take place
Palisade cells
Chlorophyll
Chloroplasts
Vacuole
42
Multiple Choice
What is the purpose of photosynthesis?
To release energy
To create glucose
To grow
To get oxygen for respiration
43
Multiple Choice
44


45

46

47

48

49
Multiple Choice
Which of the following is correct about the carbon cycle?
Photosynthesis of plant life is one of the main producers of CO2
Burning fossil fuels decreases atmospheric CO2
Glucose is a form of carbon storage in plants
Cellular respiration is a process that decreases CO2
50

51

52

53

54
Scientific Laws and Scientific Theories
A theory explains why some natural phenomena occur. A law states the observations of that phenomenon and names them.

55
Multiple Choice
Scientists must think about which of the following when designing experiments?
the steps required
equipment needed
safety precautions
all of the above
56

57
Multiple Choice
A cow is given a growth hormone and then compared to another cow that was not given a growth hormone. Both cows were weighed at 2 years. What is the INDEPENDENT variable?
amount of time
weight
growth hormone
type of cow
58
Multiple Choice
A cow is given a growth hormone and then compared to another cow that was not given a growth hormone. Both cows were weighed at 2 years. What is the DEPENDENT variable?
amount of time
weight
growth hormone
type of cow
59
Control Variables
Some variables in this experiment are kept the same:
- Time (2 years)
- Environment
It is important to keep all variables except the independent variable the same to get accurate results.
60

61

62

63

64

65

66
X-Axis Label
Tells you what the x-axis (horizontal/bottom line) is showing
Independent Variable
Matches the different trails in the experiment

67
Multiple Choice

What is the independent variable based on the graph?
Favorite candy
Type of candy
Popularity
How many people hate it
68
Y-Axis Label
Tells you what the y-axis (vertical line) is talking about
Dependent variable
What we measure/collect data on

69
Multiple Choice

What is the dependent variable based on the graph?
Popularity
Type of candy
Time of trick-or-treating
Number of trick-or-treaters
70
Trends in Graphs
Talk about how one variable changes the other
As the [independent variable] increases, the [dependent variable] ...
71
Trends in Graphs
As the time increases the mass of chalk decreases.

72
Trends in Graphs
As time increases, the speed increases.

73
Multiple Choice

Describe the trend shown in the graph.
As the volume decreases, the mass stays the same.
As the mass decreases, the volume increases.
As the volume increases, the mass increases.
As the mass increases, the volume stays the same.
74
Density
Density is number of mass in a certain volume of an object.
D = m/V
D = density (kg/m3)
m = mass (kg)
V = volume (m3)
75
Multiple Choice
76
Multiple Choice
77
Multiple Choice
78
Multiple Choice
79
Multiple Choice
80
Multiple Choice
81
Multiple Choice
82
Acids and Bases

83
Multiple Choice
The smallest part of an element is
proton
atom
compound
mixture
84
Multiple Choice
Two or more different elements chemically combine to form compounds.
True
False
85

86
Multiple Choice
Which of the following are properties of acids?
They conduct electricity when dissolved in water
They taste sour
They react with metals to produce hydrogen gas
All of the answer choices are correct
87

88

89

90

91

92
Multiple Choice

If the substance is neutral, what would the pH be?
3
5
7
9
11
93
Multiple Choice

Which of the following pH values represents a base?
2.1
4.6
6.8
8.1
94
Multiple Choice

Which is the stronger acid?
pH 1
pH 4
pH 8
pH 13
95
Multiple Choice

The strongest bases have pH values close to
0
14
7
5
96
Periodic Table divisions:
The Periodic Table is divided into groups and periods.
Groups run up and down.
Periods run side to side.
REMEMBER: "The groups! The groups! The groups are in columns! The rows! The rows! The rows are periods!

97
Multiple Choice
Groups run:
up and down
left to right
98
Multiple Choice
Periods run:
up and down
left to right
99
Metals, Metalloids, and Nonmetals
The periodic table is divided into 3 types of chemicals
They are in alphabetical order: 1. Metal, 2. Metalloid, 3. Nonmetal
Metals are the largest group
Metalloids are the smallest group between metals and nonmetals.

100
Multiple Choice
Metals are on the:
Left side
Right Side
Stairstep
101
Multiple Choice
Metalloids are on the:
Left side
Right Side
Stairstep
102
Multiple Choice
Nonmetals are on the:
Left side
Right Side
Stairstep
103
Multiple Choice
A student is studying a mystery substance. It breaks easily. It has a very dull luster. It doesn't conduct any electricity. What is the most likely substance this is?
Metal
Metalloid
Nonmetal
104
Multiple Choice
The same student works with a new substance. He concludes it conducts electricity very well. It has a very shiny luster. When he hammers it, it simply dents and flattens. What is the most likely substance type?
Metal
Metalloid
Nonmetal
105
Multiple Choice
The third substance the student works with is a little brittle. It is just a little shiny. When he tries to conduct electricity through it at first, it doesn't conduct, but upon heating up, it does start to allow electrons to flow. What is the most likely substance type?
Metal
Metalloid
Nonmetal
106

107
Multiple Choice
Valence electrons are
the electrons in the outer most energy level
all the electrons in the atom
108

109
Multiple Choice
The chemical symbol for an element is always represented by only 1 letter
True
False
110

End of Year Review Physical Science
This review contains: Periodic Table, Scientific Investigation, Graphs, Cycle, Acids and Bases, Photosynthesis
Show answer
Auto Play
Slide 1 / 110
SLIDE
Similar Resources on Wayground

106 questions
Overview of Ecology
Presentation
•
6th - 8th Grade

106 questions
Ecology Lessons
Presentation
•
6th - 8th Grade

109 questions
F4 (R) Buy It! L35 2021
Presentation
•
8th Grade

105 questions
9.1 Photosynthesis & Plant Growth
Presentation
•
7th - 9th Grade

104 questions
Rational Numbers and Operations on Rational Numbers
Presentation
•
7th Grade

106 questions
F3 FoodX3 L17
Presentation
•
8th Grade

103 questions
Bài thuyết trình chưa có tiêu đề
Presentation
•
8th Grade

99 questions
Energy Review 3rd 9 weeks
Presentation
•
8th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade