

Chemistry Review for Final 1: Measurement
Presentation
•
Science, Chemistry
•
11th Grade
•
Medium
Standards-aligned
William Jared Lovering
Used 6+ times
FREE Resource
27 Slides • 27 Questions
1
Chemistry Review for Final
By William Lovering
2

3

4
Significant Figures Rules
To determine the number of significant figures in a number use the following 3 rules:
1. Non-zero digits are always significant
2. Any zeros between two significant digits are significant. Example: 201
2. A final zero or trailing zeros in the decimal portion ONLY are significant. Example: 0.500 or 0.632000 the zeros are significant 0.006 or 0.000968 the zeros are NOT significant.
5

6
Significant figures in addition and subtraction
For addition and subtraction use the following rules: Count the number of significant figures in the decimal portion ONLY of each number in the problemAdd or subtract in the normal fashionYour final answer may have no more significant figures to the right of the decimal than the LEAST number of significant figures in any number in the problem.
7

8
Calculations with Sig Figs
Know your rules for adding/subtracting and multiplying/dividing!

9
Significant Figures in Multiplication and Division
For multiplication and division use the following rule:
The LEAST number of significant figures in any number of the problem determines the number of significant figures in the answer. (You are now looking at the entire number, not just the decimal portion)
*This means you have to be able to recognize significant figures in order to use this rule*
Example: 5.26 has 3 significant figures; 6.1 has 2 significant figures
10

11
Calculations with Sig Figs
Know your rules for adding/subtracting and multiplying/dividing!

12

13
The correct way to measure a value...
Measurements should always reflect the degree of accuracy that a device is capable of producing.
If you know the value of the smallest division marked on a measurement device, any values read from that device MUST contain that level of precision plus one estimate.
For example, if a ruler is marked into tenths of a centimeter then the recorded value of a measurement would be to the hundredth of a centimeter.
14
Making Measurements w Sig Figs
Be sure to read all of the digits (numbers) from the measuring device and then estimate one digit (number)!


15
Significant Figures and Measurement
Which numbers count?

16
Percent Error Calculations
The formula for calculating % error is on Reference Table T!

17

18

19
Scientific Notation is a shorthand method of writing incredibly large and incredibly small numbers

20
The integer (often represented by M) must be between 1 and 10.
If the power of 10 (often represented by n) is negative, the number is incredibly small. If it is positive, then it is incredibly large.

21

22
Addition and Subtraction Procedure
First, each power of ten must match. Do this by moving the decimal point of one number, changing the power of ten so that the number's actual value does not change.
Add or subtract the M values and keep the same power of ten.
Write the answer in correct scientific notation (M x 10n where M is between 1 and 10).

23
Multiplication and Division Procedure
Multiply or divide the M values.
Multiply or divide the powers of ten (which means adding or subtracting the exponents.
Write the answer in correct scientific notation (M x 10n where M is between 1 and 10).

24

25

26

27

28
Open Ended

Six students used the ruler in Model 1 to measure the length of a metal strip. Their measurements
are shown at the right. Were all of the students able to agree on a single value (1, 2, 3…)
for any digit (ones place, tenths place, etc.) in the measurement? If yes, which value and digit did
they agree on? (Click the picture if you need to see it more closely)
29
Open Ended

The students obtained an even better ruler, shown in Model 3.
Were the students able to agree on a single value for any of the digits in their measurements using the new ruler? If yes, what value(s) did they agree on in which digits?
30
Fill in the Blanks
Type answer...
31
Fill in the Blanks
Type answer...
32
Fill in the Blanks
Type answer...
33
Multiple Choice
Which metric unit is used to measure length?
Meters
Liters
Grams
34
Multiple Choice
Which metric unit is used to measure mass?
Meters
Liters
Grams
35
Fill in the Blanks
Type answer...
36
Multiple Choice
Which digits are significant?
10.512
10.512
10.512
10.512
10.512
37
Multiple Choice
Which digits are significant?
36000.
36000.
36000.
36000.
36000.
38
Multiple Choice
Select the answer reported to the correct number of significant digits.
520 - 92.1
430
428
427.9
42
39
Multiple Choice
What is the correct numerical setup to calculate the percent error for a measurement of 49.67 g if the accepted value is 50.00 g ?
49.67 g − 50.00 g
(50.00 g49.67 g −50.00 g)
(50.00 g49.67 g −50.00 g) x 100
40
Multiple Choice
What is the error if the measured value is 2.65 g/cm3 and the accepted value is 2.70 g/cm3 ?
0.5 g/cm3
0.05 g/cm3
-0.5 g/cm3
-0.05 g/cm3
41
Multiple Choice
What is the percent error if the measured value is 2.65 g/cm3 and the accepted value is 2.70 g/cm3 ?
1.85 %
5 %
1.89 %
0.05 %
0.0185 %
42
Multiple Choice

What is the mass (in grams) pictured on this triple beam balance?
37.3
37.30
7.3
37.300
43
Multiple Choice

What is the mass (in grams) pictured on this triple beam balance?
263.49
623.5
263.490
263.5
44
Multiple Choice

Which of the following would best represent the volume of the liquid in this graduated cylinder?
20.6
27.0
25.9
26
45
Multiple Select
This number has 5 significant figures
10000
0.00025
20200
0.021000
46
Multiple Choice
.000457
47
Multiple Choice
(3.4 x 105)(1.2 x 10-3)
48
Multiple Choice
(2.1×102) × (3.0×104)
49
Multiple Choice
(9 x 10-6) / (3 x 10-3)
50
Multiple Choice
51
Multiple Choice
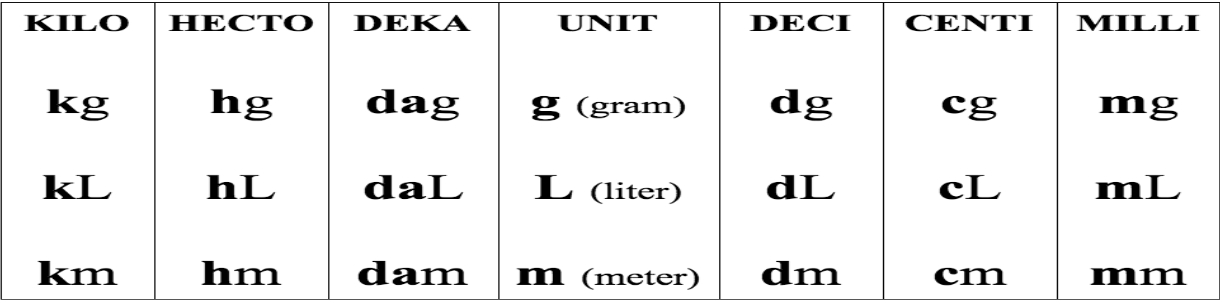
64,600,000 cm = ____ km
646
64.6
6,460
6.46
64,600
52
Multiple Choice
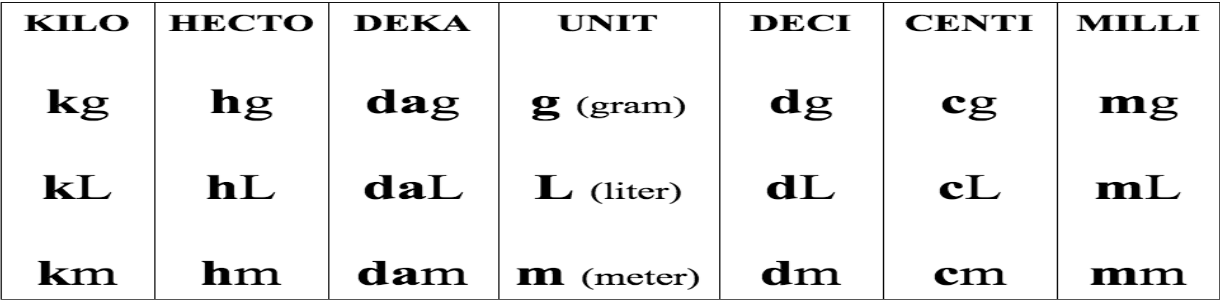
425,000 mL = ___ L
425
42.5
4,250
4.25
42,500
53
Multiple Choice
How many kilograms of calcium are there in 173 pounds of calcium? (1 pound = 454 grams; 1 kg = 1000 g)
1.10 kg
78.5 kg
110 kg
78500 kg
54
Multiple Choice
Chemistry Review for Final
By William Lovering
Show answer
Auto Play
Slide 1 / 54
SLIDE
Similar Resources on Wayground

49 questions
1.1-1.3
Presentation
•
11th Grade

48 questions
Chemical and Mechanical Weathering
Presentation
•
11th Grade

48 questions
Weathering, Erosion, and Deposition Review
Presentation
•
11th Grade

48 questions
Weathering and Erosion
Presentation
•
11th Grade

48 questions
Weathering and Deposition by Water
Presentation
•
11th Grade

48 questions
Factors Contributing to Weathering of Rocks
Presentation
•
11th Grade

50 questions
Endogenic Processes II - Earthquakes and Volcanoes
Presentation
•
11th Grade

49 questions
Alternative Energy
Presentation
•
11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

20 questions
Frog Dissection Quiz
Quiz
•
11th Grade

20 questions
Final Review Thermochemistry
Quiz
•
9th - 12th Grade

11 questions
Explore Homeostasis and Feedback Mechanisms
Quiz
•
9th - 12th Grade

99 questions
PS Semester 2 Review
Quiz
•
9th - 12th Grade

49 questions
Florida Biology EOC Practice Test
Quiz
•
9th - 12th Grade

40 questions
Anatomy and Physiology Final Exam Review Part 1
Quiz
•
11th Grade