

Electrochemistry remediation
Presentation
•
Chemistry
•
10th - 12th Grade
•
Medium
+5
Standards-aligned
Lynsey Williams
Used 4+ times
FREE Resource
6 Slides • 24 Questions
1
Electrochemistry remediation
By Lynsey Williams
2

Vocabulary
3
Multiple Choice
What is reduction?
It is the gain of electrons.
It is the loss of electrons.
It is the creation of electrons.
It is the destruction of electrons.
4
Multiple Choice
What is oxidation?
It is the gain of electrons.
It is the loss of electrons.
It is the creation of electrons.
It is the destruction of electrons.
5
Multiple Choice
What is a galvanic (voltaic) cell?
It is a cell that destroys electrons on one side and creates electrons on the other side.
It is a cell that contains only one metal bar and one aqueous ion solution.
It is a type of battery that drives a redox reaction when electricity is applied.
It is a type of battery that generates an electrical current from redox reactions.
6
Multiple Choice
What is an anode?
It is the electrode where oxidation takes place.
It is the electrode where reduction takes place.
It is an aqueous solution containing an electrode.
It is the salt bridge that connects half-cells.
7
Multiple Choice
What is a cathode?
It is the electrode where oxidation takes place.
It is the electrode where reduction takes place.
It is an aqueous solution containing an electrode.
It is the salt bridge that connects half-cells.
8
Multiple Choice
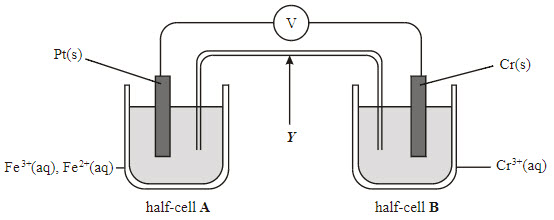
9
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
15
Multiple Choice

As an element is oxidized, its oxidation number
decreases as electrons are lost
increases as electrons are lost
decreases as electrons are gained
increases as electrons are gained
16
Multiple Choice

Mg + PbCl2 → MgCl2 + Pb
Which statement correctly describes the oxidation and reduction that occur?
Mg is oxidized and Pb+2 is reduced
Mg is oxidized and Cl- is reduced
Mg is reduced and Cl- is oxidized
Mg is reduced and Pb+2 is oxidized
17
The element higher on the list is stronger and is therefore oxidized.
Stronger Element is oxidized
Using oxidation table
Some text here about the topic of discussion

18
Multiple Choice
If Al is above Co in the activity series of metals, what is true?
no reaction
both elements are reduced
Al is reduced
Al is oxidized
19
Multiple Choice

Look at the metal activity table (4.5) Who is oxidized?
Cu
Zn
CuSO4
ZnSO4
20
Multiple Choice
21

Electromagnetic Spectrum
Some text here about the topic of discussion.
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice
Light is a form of energy.
True
False
30
Multiple Choice
How do we see an object?
Light reflects off the object and into our eyes.
Light travels from our eyes and reflects off the object
Light reflects off our eyes and enters the object
The object produces its own light
Electrochemistry remediation
By Lynsey Williams
Show answer
Auto Play
Slide 1 / 30
SLIDE
Similar Resources on Wayground

22 questions
Where do electrons live?
Presentation
•
9th - 12th Grade

25 questions
Gas Laws
Presentation
•
9th - 12th Grade

23 questions
Mole Conversions Review
Presentation
•
10th - 12th Grade

22 questions
Lewis Dot Structures
Presentation
•
9th - 12th Grade

23 questions
Valence Electrons, Lewis Dot Diagrams and Ions
Presentation
•
10th - 12th Grade

22 questions
Compound names and formulas
Presentation
•
10th - 12th Grade

24 questions
Nuclear Decay
Presentation
•
9th - 12th Grade

24 questions
Gas Laws Lesson - Part 1
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade