

3.2 e: Galvanic Cells
Presentation
•
Science, Chemistry
•
10th Grade - University
•
Medium
Erin Hannan
Used 21+ times
FREE Resource
13 Slides • 9 Questions
1
Galvanic Cells Basics
Year 11 Chemistry
2
Syllabus dot points
3.2 e conduct investigations to measure and compare the reduction potential of galvanic half-cells
3
What we will cover
What is a galvanic cells/types
Parts of a galvanic cell
How to draw
4
What is a galvanic cell?
A device where a chemical reaction occurs that produces electricity
Batteries are galvanic cells
Conversion of chemical energy into electrical energy
Uses redox reactions because of the movement of electrons
the electrons flow through a set up wire and generate a current/electricity

5
Galvanic or Voltaic Cell?
Both names are used to describe the same device - but why?
Luigi Galvani and Alessandro Volta both came up with the same system!


6
Parts of a Galvanic Cell

Electrodes
Cathode - where reduction occurs
Anode - where oxidation occurs
Solutions
an electrolyte solution that can conduct electricity
Salt bridge
provides electrical contact between the two solutions/half cells
something containing an electrolyte solution
7

8
How does a galvanic cell work?
Metal electrodes are placed in their respective electrolyte solutions
The redox reactions begin
In this example:
The zinc anode is where oxidation occurs - zinc enters the solution from the electrode - loss
The copper cathode is where reduction occurs - copper attaches to the electrode from the solution - gain
Electrons flow between two half cells, from negative terminal to positive terminal
The salt bridge completes the circuit

9
10
Drawing a galvanic cell

two half cells
salt bridge
wire + bulb/voltmeter
labelling:
electrodes
solutions
direction of electrons
movement of ions
sometimes: the half reactions
11
Draw the following galvanic cell:

Include:
all experimental set up (incl. a bulb)
labels of cathode and anode
labels of solutions
direction of electron flow
half reactions occurring at each electrode
the migration of ions within each half cell
12

13
Multiple Choice
In a Galvanic Cell, the anode is...
positive
negative
14
Multiple Choice
In a Galvanic Cell, the cathode is...
positive
negative
15
Multiple Choice
Oxidation occurs at the...
anode
cathode
16
Multiple Choice
Reduction occurs at the...
anode
cathode
17
Multiple Choice
In a Galvanic cell, the electrons flow from the...
anode to the cathode
cathode to the anode
18
Multiple Choice
In a Galvanic Cell, the salt bridge...
completes the circuit so that electrons can flow through the wire
maintains charge balance
is soaked in an unreactive electrolyte such as KNO3
all of the above
19
Multiple Choice
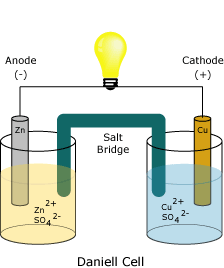
In the above Galvanic Cell, which half cell is undergoing reduction?
Cu | Cu2+
Zn | Zn2+
20
Multiple Choice
A certain galvanic cell has for its spontaneous cell reaction: Zn + HgO → ZnO + Hg
Which is the half-reaction occurring at the anode?
HgO + 2 e- → Hg + O2-
Zn2+ + 2 e- → Zn
Zn → Zn2+ + 2 e-
ZnO + 2 e- → Zn
21
Multiple Choice
A certain galvanic cell has for its spontaneous cell reaction: Zn + HgO → ZnO + Hg
Which is the half-reaction occurring at the cathode?
HgO + 2 e- → Hg + O2-
Hg → Hg2+ + 2 e-
Zn → Zn2+ + 2 e-
ZnO + 2 e- → Zn
22
Up next:
Standard electrode potentials
calculating voltages
Galvanic Cells Basics
Year 11 Chemistry
Show answer
Auto Play
Slide 1 / 22
SLIDE
Similar Resources on Wayground

17 questions
Ion Concentration
Presentation
•
9th - 12th Grade

19 questions
Homeostasis
Presentation
•
11th - 12th Grade

20 questions
Cell Division
Presentation
•
9th - 12th Grade

17 questions
Balancing Equations and Predicting Products Guided Practice
Presentation
•
9th - 12th Grade

18 questions
6.1 c: Acid/Base Reactions
Presentation
•
10th Grade - University

17 questions
REAKSI REDOKS
Presentation
•
10th - 12th Grade

15 questions
8.4-8.7
Presentation
•
10th - 12th Grade

18 questions
Honors Diseases and Disorders of the Integumentary System
Presentation
•
11th - 12th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Science

10 questions
Exploring Light and Waves Concepts
Interactive video
•
6th - 10th Grade

10 questions
Evolution Review
Presentation
•
10th Grade

10 questions
Exploring Plant Structures and Adaptations
Interactive video
•
6th - 10th Grade

5 questions
Evolution (Darwin)
Interactive video
•
9th - 12th Grade

10 questions
Classification and Properties of Matter
Interactive video
•
6th - 10th Grade

10 questions
Exploring Natural Selection Concepts
Interactive video
•
6th - 10th Grade

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

10 questions
Exploring the Phases of the Moon
Interactive video
•
6th - 10th Grade