

Separating a Mixture
Presentation
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Easy
Standards-aligned
Michaela Bratsch
Used 223+ times
FREE Resource
10 Slides • 15 Questions
1
Separating a Mixture

Mixtures can be separated through physical means. These are some common ways to separate a mixture.
2

3
Multiple Choice
Substances in a mixture can be separated by physical methods.
TRUE
FALSE
4

5
Multiple Choice
This method separates substances due to differences in the sizes of their particles.
Filtration
Evaporation
Distillation
Chromatography
6
Multiple Choice
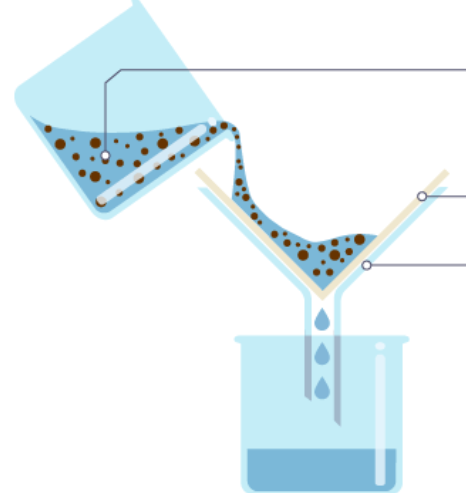
Which method is this a picture of?
Filtration
Evaporation
Distillation
Chromatography
7
Multiple Choice
What method of separation would work best to separate water and rubber pellets?
filtration
sifting
evaporation
magnetic attraction
8

9
Multiple Choice
What tool can be used to separate plastic beads and metal paper clips?
filter
magnet
strainer
cloth
10
Multiple Choice

You can boil them.
You can use centrifugation.
11

12

13
Multiple Choice
Which method separates substances based on them having different boiling points?
Filtration
Centrifugation
Distillation
Chromatography
14
Multiple Choice

The process of separating the substances from a liquid mixture by using selective boiling and condensation is called _____.
Decomposition
Filtration
Distillation
Magnetism
15
Multiple Choice
John wants to separate salt from water. What separation technique should he use?
boiling/distillation
filtration
decanting
magnetism
16

17

18
Multiple Choice
magnetism
distillation
19
Multiple Choice
The process where a mixture is separated through spinning is called ______.
Magnetism
Chromatography
Distilling
Centrifuging
20
Multiple Choice

What separation technique is used for separating blood into its 4 components?
Evaporation
decantation
filtration
centrifuging
21

22

23
Multiple Choice
This method separates dissolved substances based on differences in how well they dissolve in a solvent.
Filtration
Evaporation
Distillation
Chromatography
24
Multiple Choice
If a substance is insoluble in water, it ___________.
dissolves in water
cannot dissolve in water
forms a solution when mixed with water
cannot be separated from water
25
Multiple Choice
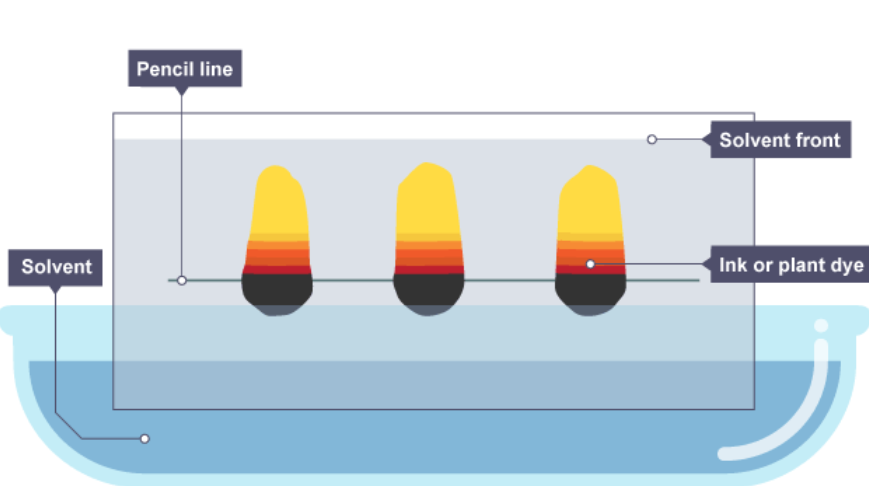
Which method is this a picture of?
Filtration
Evaporation
Distillation
Chromatography
Separating a Mixture

Mixtures can be separated through physical means. These are some common ways to separate a mixture.
Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

20 questions
Gases, Liquids, and Solids - Section 2
Presentation
•
10th - 12th Grade

21 questions
Chemical Reactions Intro
Presentation
•
9th - 12th Grade

21 questions
Foss Chemical Reactions
Presentation
•
9th - 12th Grade

19 questions
Emission Spectra Chemistry
Presentation
•
10th - 12th Grade

14 questions
Particle Diagrams
Presentation
•
9th - 12th Grade

19 questions
group 2, group 17, nitrogen and sulfur
Presentation
•
10th - 11th Grade

18 questions
Safety
Presentation
•
10th Grade - University
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade