

Periodic Table Review
Presentation
•
Chemistry, Science
•
9th - 12th Grade
•
Easy
Standards-aligned
Megan Ghothane
Used 34+ times
FREE Resource
15 Slides • 22 Questions
1

2

3

4

5

6
Multiple Choice

7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice

The atomic number determines what an element is. A different atomic number makes a different element. What is the atomic number of Neon (Ne)?
10
20.18
9
58
none of these is correct
11
Multiple Choice

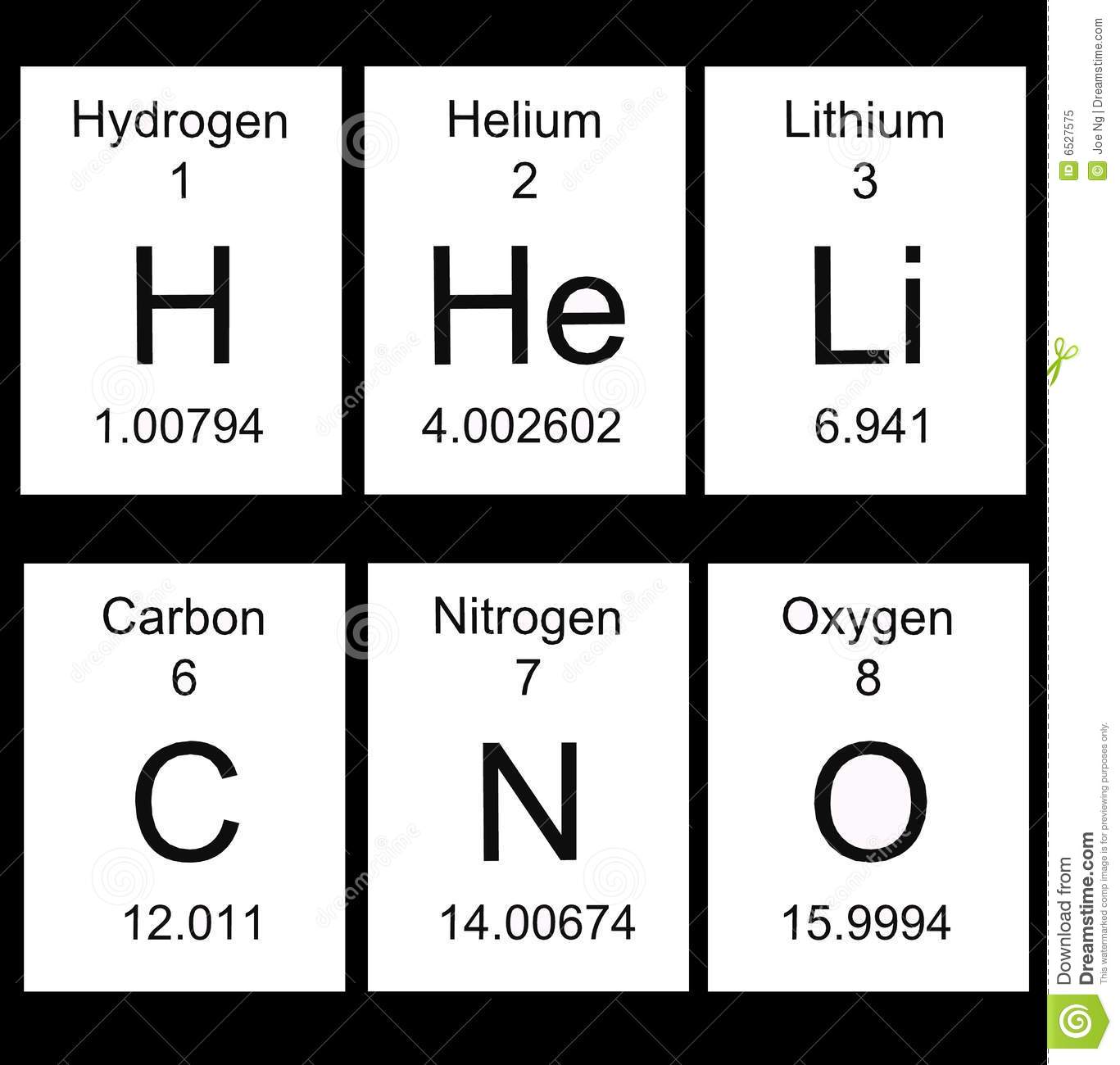
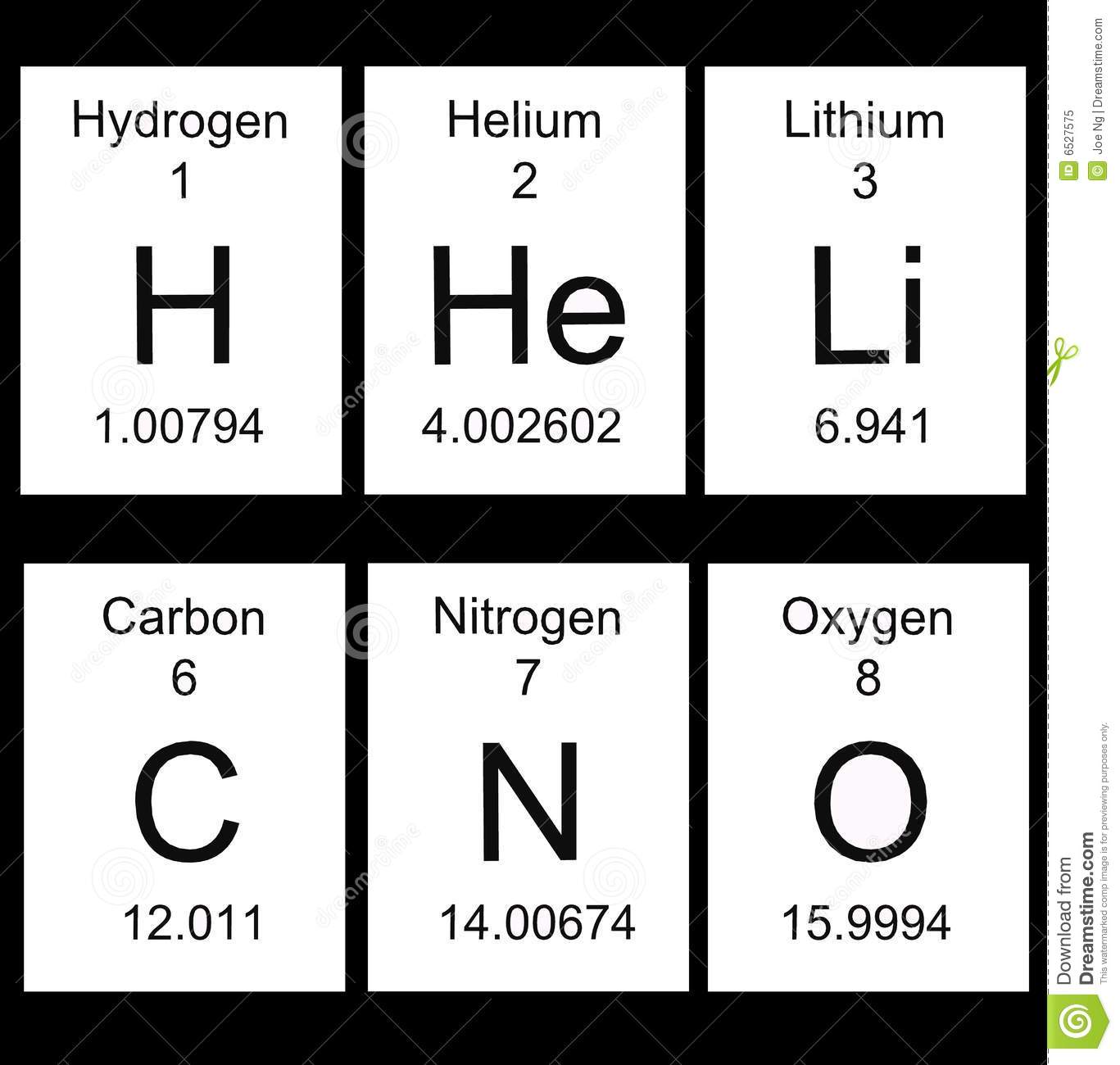
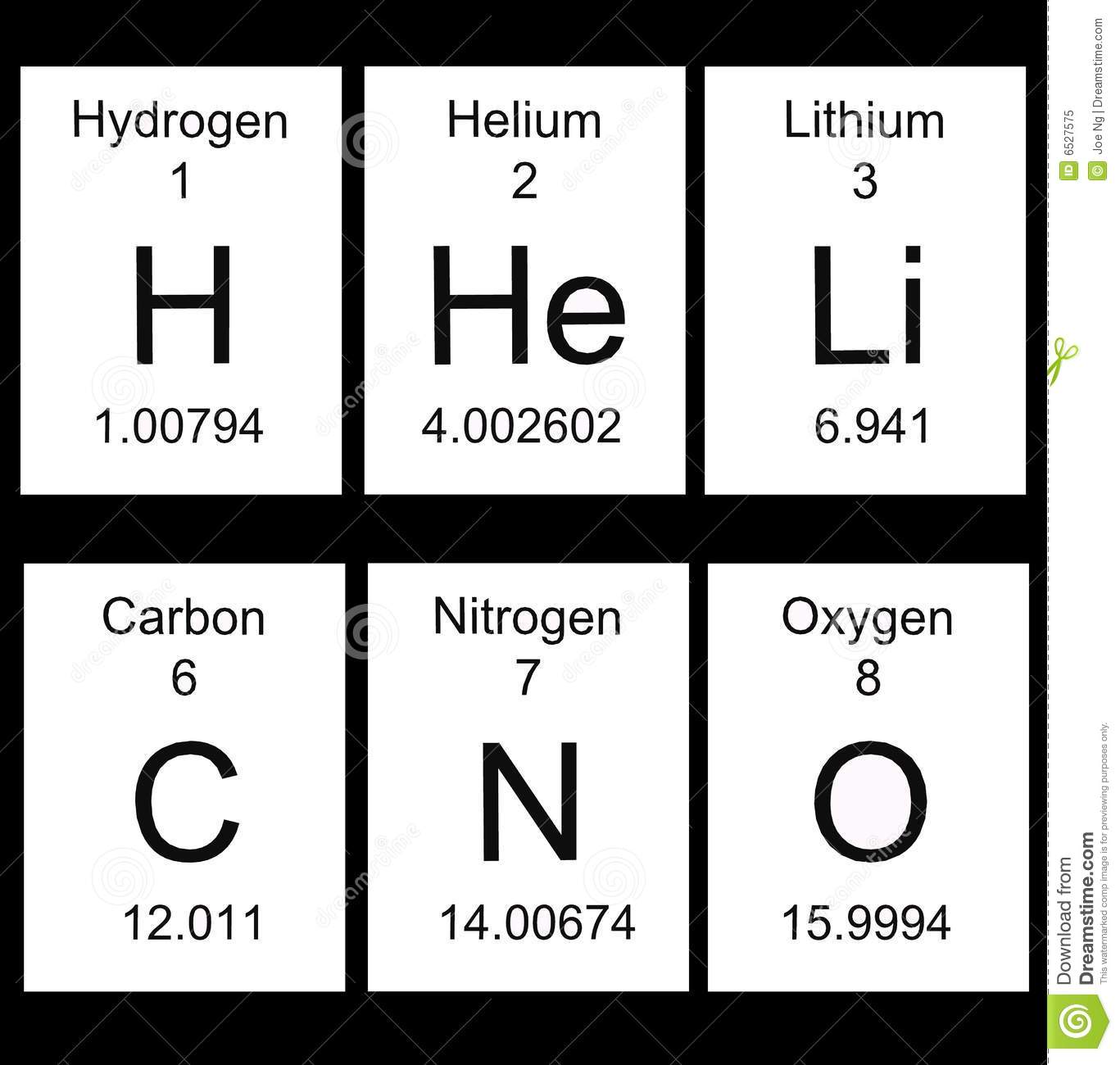
What does the 8 represent in the image attached?
Atomic mass
Element symbol
Atomic number
Element name
12
Multiple Choice

What does the decimal number at the bottom of the attached image represent?
Atomic mass
Element symbol
Atomic number
Element name
13
Multiple Choice
Which two subatomic particles determine an element’s mass number?
Electrons and neutrons
Protons and neutrons
Protons and electrons
None of the above
14
Multiple Choice
What determines an element’s atomic number?
The element's mass
The number of neutrons it has
The size of the element
The number of protons it has
15
Multiple Choice
What is the element name for the symbol H?
Helium
Hydrogen
Gold
Tungsten
16
Multiple Choice

Which of these elements has the greatest average atomic mass?
B
C
N
Al
17
Multiple Choice

Which of these elements has an atomic number of 14?
N
Al
Si
P
18

19

20

21

22

23
Multiple Choice
What are the vertical columns on the periodic called?
groups
periods
nonmetals
metals
metalloids
24
Multiple Choice
What do the vertical columns on the periodic table tell us?
The number of orbitals
The number of valence electrons (electrons on the outer most shell)
25
Multiple Choice
What are the horizontal rows on the periodic table called?
periods
groups
metals
nonmetals
metalloids
26

27

28

29

30

31
Multiple Choice
32
Multiple Choice
33
Multiple Choice
34
Multiple Choice
35
Multiple Choice

36
Multiple Choice
37
Multiple Choice

Show answer
Auto Play
Slide 1 / 37
SLIDE
Similar Resources on Wayground

30 questions
Eukaryotic Cell Structure
Presentation
•
9th - 12th Grade

30 questions
Endothermic/Exothermic & Calorimetry
Presentation
•
9th - 12th Grade

30 questions
Writing Ionic Formulas
Presentation
•
8th - 12th Grade

33 questions
Bonding Lesson Part 2
Presentation
•
9th - 12th Grade

36 questions
2nd 6wks
Presentation
•
9th - 12th Grade

33 questions
Energy & Energy Transformations
Presentation
•
9th - 12th Grade

33 questions
Population Ecology Review
Presentation
•
9th - 12th Grade

30 questions
Evidence of Evolution
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

120 questions
2nd Semester Final Review
Quiz
•
11th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade