

Chemistry 1-4: Electrons in Atoms
Presentation
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Easy
+9
Standards-aligned
Abby Fancsali
Used 41+ times
FREE Resource
33 Slides • 18 Questions
1
Electrons in Atoms

2
Multiple Choice
3
Multiple Choice
What is the atomic number?
the number of protons in the nucleus
the number of protons and neutrons in the nucleus
the number of neutrons in the nucleus
the number of protons in the energy levels
4
Fill in the Blanks
Type answer...
5
Multiple Choice
What is the mass number?
the number of protons in the nucleus
the number of protons and neutrons in the nucleus
the number of neutrons in the nucleus
the number of protons and electrons in the atom
6
Multiple Choice
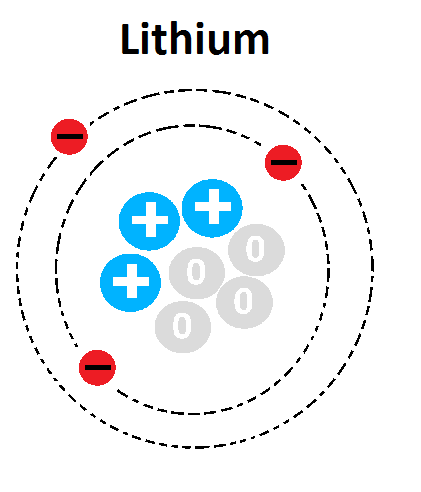
What is the atomic number of this atom?
1
3
4
7
7
Multiple Choice
8

Lesson Objectives
Identify the different blocks of the periodic table
Describe the electron configurations using different methods
Draw Valance electron structures for different atoms
9
Organizing the Periodic Table
The periodic table is organized into periods and groups
Periods: Horizontal Rows
Groups: Vertical Columns
We can divide the periodic table into different blocks based on the highest-level subshells with electrons
s
p
d
f

10
S-block

11
P-block

12
d-block

13
f-block

14
Electron Configurations
Electron Configurations: The way in which electrons are arranged in various orbitals around the nuclei of atoms
Electrons fill orbitals following certain rules
Energy plays a major role in how electrons arrange themselves in an atom
Three rules for Electron Configurations
15
The Aufbau Principle
The Aufbau Principle: Electrons occupy the lowest energy sub-shells first
This makes elements more stable
the 1s orbitals will always fill before the 2s orbitals
Order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p....
d sub-shells always a lower energy level than the rest of the period
16
Hunds Rule
Hunds Rule: In sub-shells with multiple orbitals (p, d, f), all orbitals must be filled before getting a second electron
Every orbital has to get an electron before getting seconds

17
Pauli's Exclusion Principle
Pauli's Exclusion principle: electrons in the same orbital can not be in the same spot at the same time
Spin: a property of electrons that describes their orientation in an orbital
We show this with two verticle arrows pointing in opposite directions

18
Writing Electron Configurations Sample Problem 1
Step 1: Use the Atomic number to determine the number of electrons
Example: Oxygen
19
Fill in the Blanks
Type answer...
20

Writing Electron Configurations Sample Problem 1
Step 1: Use the Atomic number to determine the number of electrons
Example: Oxygen has the atomic number of 8, so it has 8 electrons
Step 2: Use arrows to represent the number of electrons in each orbitals
Start in the 1s sub-shell
Each orbital in a shell has to get an electron before it can get a second one
Electrons in the same orbital spin in opposite directions
21
Writing Electron Configurations Sample Problem 1
Step 1: Use the Atomic number to determine the number of electrons
Example: Oxygen has the atomic number of 8, so it has 8 electrons
Step 2: Use arrows to represent the number of electrons in each orbitals
Start in the 1s sub-shell
Each orbital in a shell has to get an electron before it can get a second one
Electrons in the same orbital spin in opposite directions

22

Writing Electron Configurations Sample Problem 2
Write the Electron Configuration for Nickel
Step 1: Use the Atomic number to determine the number of electrons
23
Fill in the Blanks
Type answer...
24

Writing Electron Configurations Sample Problem 2
Write the Electron Configuration for Nickel
Step 1: Use the Atomic number to determine the number of electrons
Example: Nickel has the atomic number of 28, so it has 28 electrons
Step 2: Use arrows to represent the number of electrons in each orbitals
Start in the 1s sub-shell
Each orbital in a shell has to get an electron before it can get a second one
Electrons in the same orbital spin in opposite directions
25

Writing Electron Configurations Sample Problem 3
Write the Electron Configuration for Silicon
Step 1: Use the Atomic number to determine the number of electrons
26
Fill in the Blanks
Type answer...
27

Writing Electron Configurations
Step 1: Use the Atomic number to determine the number of electrons
Example: Silicon has the atomic number of 14, so it has 14 electrons
Step 2: Use arrows to represent the number of electrons in each orbitals
Start in the 1s sub-shell
Each orbital in a shell has to get an electron before it can get a second one
Electrons in the same orbital spin in opposite directions
28
Writing Electron Configurations
Step 1: Use the Atomic number to determine the number of electrons
Example: Silicon has the atomic number of 14, so it has 14 electrons
Step 2: Use arrows to represent the number of electrons in each orbitals
Start in the 1s sub-shell
Each orbital in a shell has to get an electron before it can get a second one
Electrons in the same orbital spin in opposite directions

29
Exceptions to Electron Configuration Rules
Some elements do not follow the normal electron configuration rules that others do
Chromium: 1s2 2s2 2p6 3s2 3p6 4s2 3d5 4s1
Copper: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4s1
30
Noble Gas Configurations
Electron Configurations can get long
Noble Gases: the last group of the periodic table
You can shorten the writing by starting with the Noble Gas before the element, and only writing what is in the period of the component in questions

31
Noble Gas Configuration Sample Problem 1
Write the Noble Gas Configuration for Sulfur
Step 1: Identify the Noble gas that comes before sulfur
32
Multiple Choice
Which Nobel Gas come before sulfur?
Helium
Neon
Argon
Krypton
Xenon
33
Noble Gas Configuration Sample Problem 1
Write the Noble Gas Configuration for Sulfur
Step 1: Identify the Noble gas that comes before sulfur
The Noble Gas before sulfur is Neon
Step 2: Write Neon's Chemical Symbol in Brackets
Step 3: Continue the configuration from where Neon is
Shortened Configuration: [Ne] 3s13p4
34
Noble Gas Configuration Sample Problem 2
Write the Noble Gas Configuration for Potassium
Step 1: Identify the Noble Gas that comes before Potassium.
35
Multiple Choice
Which Nobel Gas come before Potassium?
Helium
Neon
Argon
Krypton
Xenon
36
Noble Gas Configurations Sample 2
Write the Noble Gas Configuration for the element Potassium
Step 1: Identify the Noble Gas that comes before Potassium
The noble Gas before Sulfur is Argon (Ar)
Step 2: Write Argon's Chemical Symbol in Brackets
Step 3: Continue the configuration from where Argon is
Shortened Configuration: [Ar] 4s1

37
Valence Electrons
Electrons in an element are divided into two groups
Core electrons: exist in the inner and lower energy shells
Valence Electrons: electrons in the outermost p energy shell
are most involved in reactions
are unstable unless they have a full electron shell
Noble Gases have a full shell, which is why they don't react
The Octet Rule: Atoms are most stable when they have a total of eight electrons in their outer shell, and will respond to gain or lose the electrons needed

38
Identifying Valence Electrons
Counted by Looking at the Group numbers
Group 1=1, Group2=2
Groups 13-18: Valence electrons = Group number-10
Group 13=3, Group 14=4, Group 15=5, Group 16=6 Group 17=7, Group 18=8
Shown Using Electron Dot Structures
A visual representation of the valence electrons in an element
39
Valence Electrons Sample Problem 1
Sample Problem: Draw the electron dot structure for Sodium
Step 1: Identify which group Sodium is in
40
Multiple Choice

Which Group is Sodium in?
1
2
12
14
16
41
Valence Electrons Sample Problem 1
Sample Problem: Draw the electron dot structure for Sodium
Step 1: Identify which group Sodium is in
Sodium is in group 1 so it has 1 Valance Electron
Step 2: Write the Element symbol
Step 3: Draw dots around the Symbol, one for each valance electron
Start at the top and go clockwise
Don't Double up until all four sides have one

42
Valence Electrons Sample Problem 2
Sample Problem: Draw the electron dot structure for the Silicon
Step 1: Identify which group Silicon is in
43
Multiple Choice

Which Group is Silicon in?
1
2
13
14
16
44
Valence Electrons Sample Problem 2
Sample Problem: Draw the electron dot structure for the Silicon
Step 1: Identify which group Sodium is in
Silicon is in group 14 and has 4 Valance Electrons
Step 2: Write the Element symbol
Step 3: Draw dots around the Symbol, one for each valance electron
Start at the top and go clockwise
Don't Double up until all four sides have one

45
Valence Electrons Sample Problem 3
Sample Problem: Draw the electron dot structure for Chlorine
Step 1: Identify which group Chlorine is in
46
Multiple Choice

Which Group is Chlorine in?
2
13
13
14
17
47
Valence Electrons Sample Problem 3
Sample Problem: Draw the electron dot structure for Chlorine
Step 1: Identify which group Chlorine is in
Chlorine is in group 17 and has 7 Valance Electrons
Step 2: Write the Element symbol
Step 3: Draw dots around the Symbol, one for each valance electron
Start at the top and go clockwise
Don't Double up until all four sides have one

48
Draw
Draw the electron dot structure for sulfur
49
Draw
Draw the electron dot structure for calcium
50
Hotspot
Click all the Valence electrons in this atom.
51
Multiple Choice

How many valence electrons are in this element?
7
6
5
2
Electrons in Atoms

Show answer
Auto Play
Slide 1 / 51
SLIDE
Similar Resources on Wayground

45 questions
Ch 8 (Pt 1)
Presentation
•
10th - 12th Grade

45 questions
Subatomic and Isotopes Review
Presentation
•
9th - 12th Grade

51 questions
Lesson1: Motion
Presentation
•
9th - 12th Grade

47 questions
Valence electrons
Presentation
•
10th - 12th Grade

48 questions
Unit 4 Lesson 1 Bohr's Models and Lewis Dot Structures
Presentation
•
10th - 12th Grade

46 questions
Minerals & Mining
Presentation
•
9th Grade - University

47 questions
HAP1 Anatomical Terms and Body Cavities
Presentation
•
10th - 12th Grade

48 questions
Functional Physical Science FINAL REVIEW
Presentation
•
KG - 12th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade