

Unit 4 Section 3: Modeling Phase Changes
Presentation
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Medium
+12
Standards-aligned
Abby Fancsali
Used 26+ times
FREE Resource
13 Slides • 21 Questions
1
Unit 4 Section 3 Modeling Phase Change

2
Multiple Choice
Phase Energy
3
Multiple Choice
PSI, torr, mmHG, atm all are units for....
temp
vol
pressure
weather
4
Fill in the Blanks
Type answer...
5
Multiple Choice
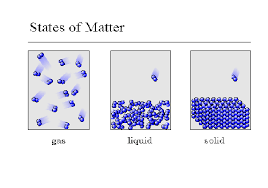
particles in an object have less energy
particles in an object have more energy
a gas contracts
a. particles in an object break apart.
6
Multiple Choice
Which of the following states of matter has the LOWEST level of energy?
Solid
Liquid
Gas
Plasma
7
Multiple Choice
As the particles increase in movement, they begin to _____ and move faster.
move closer
spread out
8
Multiple Choice
Which type of solid has low melting points and covalently bonded atoms?
9
Drag and Drop
10
Match
Match the description to the correct particle motion
The particle moves around an axis
The particle stays in one place while moving
The particle moves in space
Rotation
Vibration
Translation
Rotation
Vibration
Translation
11

Lesson Objectives
Summarize how variations in kinetic energy among particles result in changes in state
Relate Intermolecular forces to the energy needed to change states of matter.
Differentiate between the properties of various types of solids
12
Energy & Phase Changes
Phase Change: the transformation from one state of matter to another
Adding energy to a material increases the temperature of that material
At certain points, the temperature stops increasing while the substance changes state
Energy is used to either break or form intermolecular bonds

13
Types of Phase Changes
Some types of phase changes we experience on a daily basis
Solid → Liquid= Melting
Liquid → Solid= Freezing
Liquid → Gas= Vaporization
Gas → Liquid= Condensation
Other types are less common in our everyday lives
Solid →Gas= Sublimation
Gas → Solid= Deposition
The properties of a material can change at different states even if the particles are the same
14
Heating a Liquid
In the liquid state, particles have vibrational and rotational movement
still held together by intermolecular forces
Heating a liquid adds energy to the particles, allowing them to break those forces holding them together and become a gas.
Boiling Point: the temperature at which intermolecular forces break in a liquid
Liquids turn into a gas both at the surface and within the liquid
Maintaining requires constant energy
Can be predicted by looking at intermolecular forces
Compounds with Stronger forces have higher boiling points
15
Evaporation
A liquid doesn't have to boil for vaporization to occur
Every particle in a liquid has a different energy level
Below the boiling point, most particles do not have enough kinetic energy to vaporize into a gas, but a few do
Evaporation: The process by which particles at the surface of a liquid have enough energy to escape intermolecular forces and enter the gas phase
Increasing the temperature can increase the evaporation rate, even if it is not raised to the boiling point
16
Evaporation

17
Condensation
In the process of condensation, some particles have a lower energy level, which allows them to enter the liquid phase.
Cooling the temperature reduces the energy of particles, which allows particles to return to a liquid state

18
Vapor Pressure
When Liquid is in a container, some particles will evaporate
If those particles collide with the wall of the container, it produces pressure on the container that can cause them to condense back into a liquid
Vapor Pressure: the pressure of gas above a liquid that leads to an equilibrium, where some particles evaporate while others condense, so overall the state does not change

19
Vapor Pressure and Boiling
The boiling point is the temperature at which the vapor pressure is equal to the external pressure on a fluid
Changing the pressure on a liquid can change its boiling point
At sea level, warmer temperatures are needed to reach the boiling point
At higher altitudes with lower pressure, liquids boil at a lower temperature
At higher pressures, liquids boil at higher temperatures
Pressure cookers use vapor pressure to speed up cooking
Increasing the pressure increases the temperature to higher and higher point
20
Multiple Choice
TRUE OR FALSE: Matter can switch between the three phases of solid, liquid, and gas by gaining or losing energy as heat
21
Multiple Choice
Condensation is . . . .
22
Multiple Choice
As the temperature of an object increases, the energy will increase at the exact same rate
True
False
23
Multiple Select
Select all that apply.
In the process of evaporation.....
particles at the surface that have enough energy can escape the liquid and enter the gas phase.
some of the molecules in a gas will have lower energy than the rest and can transition to the liquid phase
it happens at the surface of liquids and its one type of vaporization
increasing the temperature of a liquid results in more particles having enough energy to evaporate
24
Multiple Choice
True or False: Increasing the temperature of a liquid results in more particles having enough energy to evaporate
False
True
25
Multiple Choice
The change from Liquid to Gas is...
Vaporization
26
Melting & Freezing
Particles in a solid have vibrational energy.
With enough energy, the intermolecular forces between particles can no longer contain them, and they break free from each other and change to a liquid phase
Melting Point: the temperature at which a solid becomes a liquid.
As you remove energy, the particles slow back down and return to the solid state.
Freezing Point: The point at which a liquid chages into a solid.

27
Sublimation
Solids generally melt into a liquid at a gradual temperature change
If the temperature and/or pressure are too low for the liquid state to exist, solids change to the vapor state without passing through the liquid phase
Sublimation: The change from a solid to a gas state
Occurs at STP with substances that have extremely weak intermolecular bonds
Deposition: The change of state directly from a gas to a solid
These changes give some solids' vapor pressure

28
Phase Diagrams
Phase Diagram: a graph that describes the conditions of temperature and pressure at which a substance exists as a solid, liquid, or gas
Triple point: a point on a phase diagram where all three states of matter can exist in equilibrium with each other.
Critical Point: a temperature or pressure at which a substance can not exist in the liquid state

29
Match
Match the phase change to it's name
Condensation
Vaporization
Melting
Freezing
Gas → liquid
Liquid → Gas
Solid → Liquid
Liquid → Solid
Gas → liquid
Liquid → Gas
Solid → Liquid
Liquid → Solid
30
Multiple Choice
Sublimation is the change of a
Solid to a liquid
Liquid to a solid
Gas to a solid
Solid to a Gas
Gas to a Liquid
31
Multiple Choice

This image is called a
Pressure map
phase diagram
transitional chart
Melting table
32
Multiple Choice
Change of a gas directly into a solid
Sublimation
Condensation
Evaporation
Deposition
33
Multiple Choice
A gas...
has a definite shape but no definite volume
has a definite volume but no definite shape
has fast-moving molecules
34
Multiple Choice
Melting
Condensation
Vaporization
Freezing
Unit 4 Section 3 Modeling Phase Change

Show answer
Auto Play
Slide 1 / 34
SLIDE
Similar Resources on Wayground

27 questions
Acids & Bases - Part 2
Presentation
•
9th - 12th Grade

28 questions
Acids & Bases - Introduction
Presentation
•
9th - 12th Grade

31 questions
Tissues (Epithelial & Connective)
Presentation
•
10th - 12th Grade

26 questions
Limiting Factors and Carrying Capacity EOC Review
Presentation
•
9th - 12th Grade

25 questions
Polyatomic Ions
Presentation
•
9th - 12th Grade

25 questions
Taxonomy and Classification
Presentation
•
9th - 12th Grade

25 questions
Separating a Mixture
Presentation
•
9th - 12th Grade

27 questions
Newton's Second law of Motion
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade