

The Periodic Table of the Elements
Presentation
•
Science
•
6th - 8th Grade
•
Medium
+7
Standards-aligned

Jawara Senghor
Used 94+ times
FREE Resource
9 Slides • 16 Questions
1
The Periodic Table of Elements

The building blocks of the universe
2

In the mid 1800s, a Russian chemist named Dmitri Mendeleev began to think of how elements could be organized. In the year 1869, he arranged the elements in order of increasing atomic mass and realized the properties of the elements occurred in a periodic, or regularly repeating pattern. This arrangement eventually became known as The Periodic Table of the Elements. He even used the pattern in his table to predict elements that had not yet been discovered.
The birth of the Period Table.
Organizing the Elements
3

In the year 1913, a British scientist named Henry Moseley rearranged Mendeleev's periodic table in the order of increasing atomic number instead of atomic mass. This new configuration corrected some of the flaws in Mendeleev's table. To this day, this is configuration of the periodic table that is used by scientists around the world.
A more accurate version of the Periodic Table
Re-arranging the Periodic Table
4
Multiple Choice

alphabetical
density
melting point
atomic mass
5
Multiple Choice
atomic mass
atomic number
valence electrons
number of isotopes
6

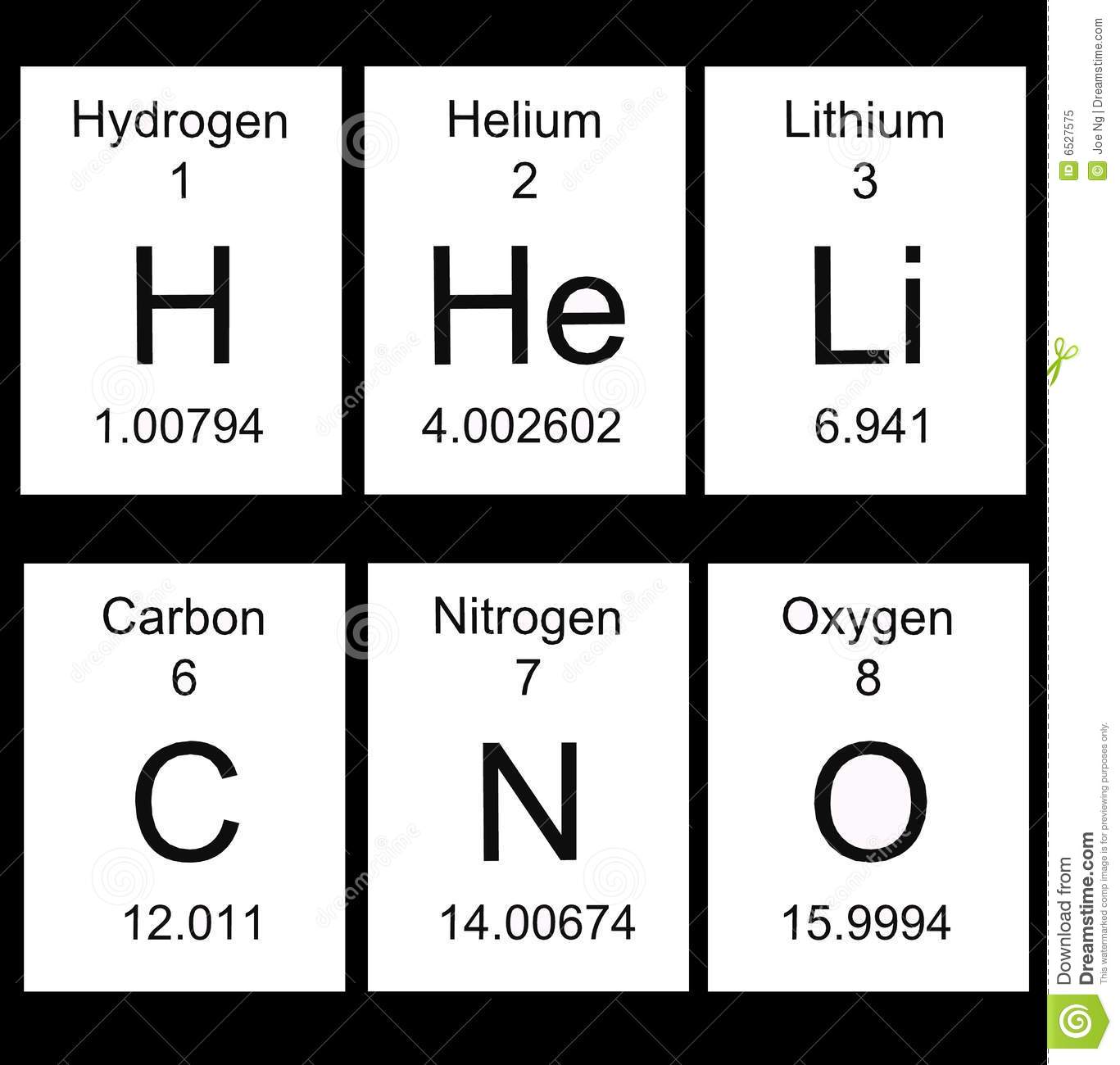
This is the information you can find in a Periodic Table square.
Atomic number - the number of protons in the nucleus of an atom of that element.
Chemical symbol - an abbreviation of the element's name
Element name - the common name of the element.
Average atomic mass - the average masses of all of the naturally occurring isotopes of that element.
How to read the Periodic Table
Reading a square
7
Multiple Choice
number of electrons
number of protons
number of neutrons
both electrons and protons in an atom.
8
Multiple Choice
atomic number
atomic mass
chemical symbol
element name
9

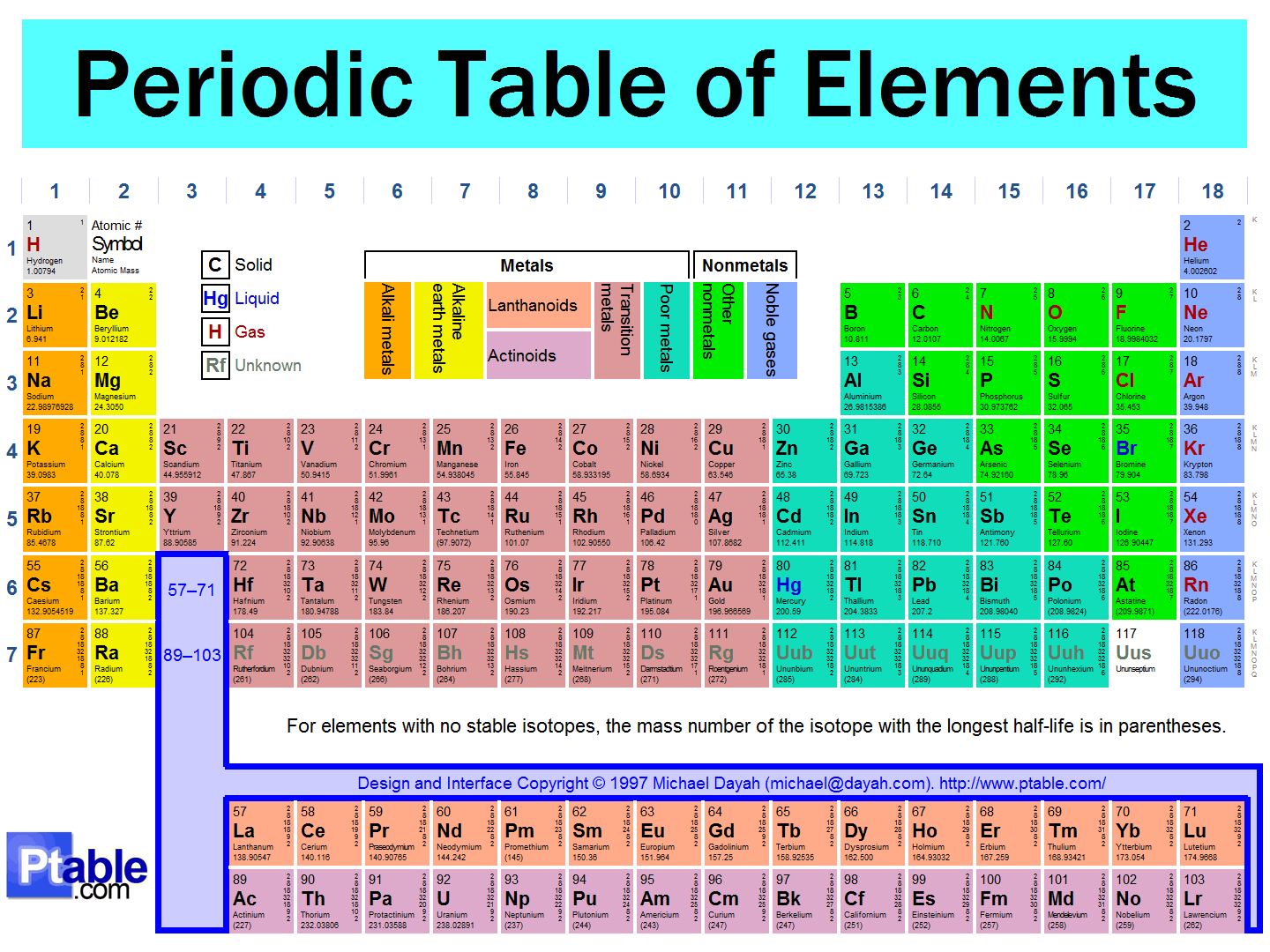
Metals:
The majority of the elements in the Periodic Table are classified as metals. Metals are usually solid at room temperature, malleable, ductile (means it can be turned into wires), shiny, and are good conductors of heat and electricity.
Classifying the Elements of the Periodic Table
Regions of the Table

10

Metalloids:
There is a zig-zag line that looks like a staircase and the elements that border this line are called metalloids. These elements have some properties of both metals and nonmetals. Some metalloids, like silicon, are used to make semiconductor chips in computers.
Classifying the elements of the Periodic Table
Regions of the Table

11

Nonmetals:
The elements to the right of the metalloid line are nonmetals which are usually dull, brittle and bad conductors of heat and electricity.
Classifying the elements of the Table
Regions of the Table

12
Multiple Select
What are the three main groups of the periodic table?
groups
metal
nonmetals
metalloids
periods
13
Multiple Choice

metals
nonmetals
metalloids
noble gases
14

Every vertical (up and down) column of elements is called a group. In total, there are 18 groups on the periodic table Elements in the same group often have similar physical and chemical properties and for this reason, a group is sometimes called a family. The properties of the elements in a group are similar because they have number of valence electrons.
Groups of the Periodic Table
Going up & down
15

Each horizontal (left to right) row of elements on the periodic table is called a period. The physical and chemical properties of elements change in predictable ways from one end of the period to another. For example, within any given period, atomic size decreases as you move from left to right. In addition to this, the density of the elements on the left and right side of a period are the least dense and the elements in the middle are the most dense.
Periods of the Table
Going left & right
16
Multiple Choice
What are the horizontal rows on the periodic table called?
periods
groups
metals
nonmetals
metalloids
17
Multiple Choice

Copper
29
Cu
63.546
18
Multiple Choice
What do the horizontal rows on the periodic table tell us?
The number of orbitals
The number of valence electrons (the electrons on the outer most shell)
19
Multiple Select
What does the number of protons tell us?
electrons
atomic number
atomic mass
neutrons
20
Multiple Choice

What is the atomic mass?
58.693
28
21
Multiple Choice

How many protons and electrons does nickel have?
28
58.69
22
Multiple Choice

Group 2
Group 3
Group 4
Group 7
23
Multiple Choice
3
2
4
1
24
Multiple Choice

The atoms on the right of the staircase are called
metalloids
non-metals
metals
noble gases
25
Multiple Choice
Metals
Nonmetals
Metalloids
Groups
The Periodic Table of Elements

The building blocks of the universe
Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

20 questions
Speed, Velocity & Acceleration
Presentation
•
6th - 8th Grade

20 questions
Newton's Laws and Collisions
Presentation
•
6th - 8th Grade

19 questions
Rock Cycle
Presentation
•
6th - 8th Grade

18 questions
Energy Transformations (Basic)
Presentation
•
6th - 8th Grade

18 questions
Reproduction
Presentation
•
6th - 8th Grade

21 questions
Breathing/gas exchange
Presentation
•
6th - 8th Grade

20 questions
ELECTRICITY AND CIRCUITS
Presentation
•
6th - 8th Grade

20 questions
Circulatory System Review
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

18 questions
Oceans, Atmosphere, Climate Vocabulary Amplify
Quiz
•
6th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade