

Unit 6 PBoM
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Medium
Eunji Kim
Used 1+ times
FREE Resource
24 Slides • 55 Questions
1

2

3

Know what quantity each unit represents!
Use Table D (cover page of the reference table) to check.
4
Multiple Choice
g or gram is representing...
mass
pressure
volume
heat
5
Multiple Choice
mL or milliliter is representing...
Note: milliliter is 1/1000 of Liter
mass
pressure
volume
heat
6
Multiple Choice
KJ or kilojoule is representing...
Note: a kilojoule is 1000 joules
mass
pressure
volume
heat
7
Multiple Choice
Which unit DOES NOT represent pressure?
mmHg
mL
kPa
atm
torr
8
Multiple Choice
Regents Practice: When a quantity of electricity is converted to heat, the heat energy produced is measured in...
amperes
volts
joules
degrees
9

10

11
Multiple Choice
Convert -16 °C to K.
-289
273
257
16
12
Multiple Choice
Convert 50 K to °C.
323
-223
273
-50
13
Multiple Choice
Regents Practice: What is the equivalent of 0 Kelvin on the Celsius scale?
-100°
100°
-273°
273°
14
Multiple Choice
Regents Practice: Which Kelvin temperature is equal to -73°C?
346K
200 K
173 K
100 K
15

16

17

18
Multiple Choice
Regents Practice: which substance has vibrating particles in regular fixed positions?
Ca(s)
Hg(l)
CaCl2(aq)
Cl2(g)
19
Multiple Choice
Regents Practice: Two basic properties of the gas phase are
a definite shape but no definite volume
no definite shape and no definite volume
no definite shape but a definite volume
a definite shape and a definite volume
20
Multiple Choice
Regents Practice: Which 5.0-milliliter sample of NH3 will take the shape of and completely fill a closed 100-milliliter container?
NH3(aq)
NH3(s)
NH3(l)
NH3(g)
21

22

23



24
Multiple Choice
When the temperature increases...
PE increases
KE increases
PE decreases
KE decreases
25
Multiple Choice
When the temperature decreases...
PE increases
KE increases
PE decreases
KE decreases
26
Multiple Choice
When the phases go from solid to liquid (endothermic)...
PE increases
KE increases
PE decreases
KE decreases
27
Multiple Choice
Regents Practice: As ice melts at standard pressure, its temperature remains at 0°C Until it has completely melted. Its potential energy...
decreases
increases
remains the same
28
Multiple Choice
Regents Practice: At which temperature would the molecules in a one gram sample of water have the lowest average kinetic energy?
(remember: average kinetic energy = temperature)
100 K
5°C
-100°C
5 K
29
Multiple Choice
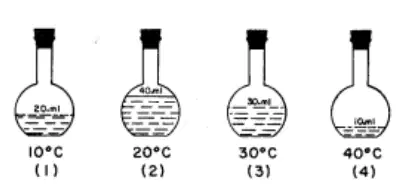
Regents Practice: In which flask do the molecules of H2O have the greatest kinetic energy?
1
2
3
4
30
Multiple Choice
Regents Practice: Which graph best shows the relationship between Kelvin temperature and average kinetic energy?
31
Multiple Choice
Regents Practice: The temperature of a substance is a measure of its particles
average potential energy
specific heat
average kinetic energy
total energy
32
Multiple Choice
Regents Practice: As ice at 0°C changes to water at 0°C, the average kinetic energy of the ice molecules
decreases
increases
remains the same
33
Multiple Choice
Regents Practice: Which change of phases is exothermic?
CO2 (s) -> CO2 (g)
H2O (l) -> H2O (s)
NaCl(s) -> NaCl (l)
H2O (s) -> H2O (g)
34
Multiple Choice
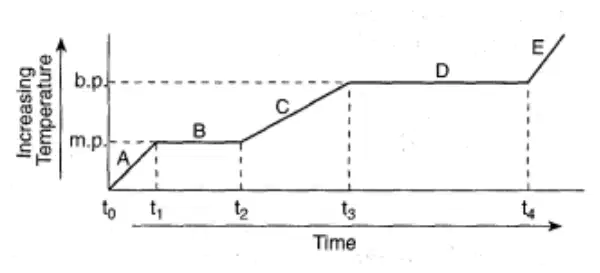
Regents Practice: The graph represents the relationship between temperature and time as heat is added uniformly to a substance, starting when the substance is solid below its melting point.
Which portions of the graph represent times when heat is absorbed and potential energy increases while kinetic energy remains constant?
B and D
A and C
C and D
A and B
35
Multiple Choice
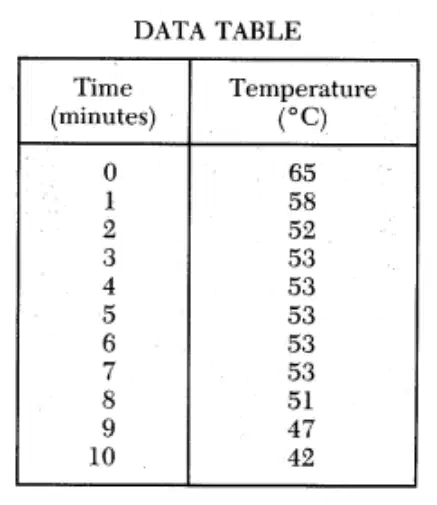
Regents Practice: A student observing the behavior of para dichlorobenzene first heats 10 grams of the substance in a hot water bath until it is completely liquefied. The following data are recorded as paradichlorobenzene cools.
53 °C
65°C
58°C
42°C
36

37
Multiple Choice
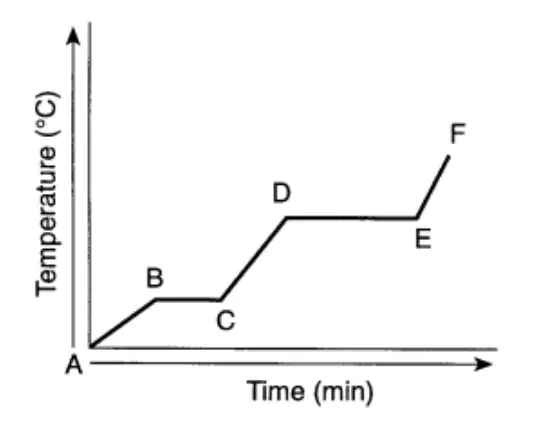
Regents Practice: The graph below represents the uniform heating of a substance, starting with the substance as a solid below its melting point.
Which segment of the graph represents a time when both the solid and liquid phases are present?
AB
BC
DE
EF
38

39
Multiple Choice
Regents Practice: Two samples of gold that have different temperatures are placed in contact with one another.
Heat will flow spontaneously from a sample of gold at 60°C to a sample of gold that has a temperature of...
80°C
70°C
50°C
60°C
40

41
Multiple Choice
Regents Practice: The strongest intermolecular forces of attraction exist in a liquid whose heat of vaporization is...
100 J/g
200 J/g
300 J/g
400 J/g
42
Multiple Choice
What is c (specific heat capacity) for H2O?
334
2260
4.18
43
Multiple Choice
What is the Heat of Fusion for H2O?
334
2260
4.18
44
Multiple Choice
If the phase of water changes from liquid to solid, which number are you going to use?
334
2260
4.18
45
Multiple Choice
If the phase of water changes from liquid to gas, which number are you going to use?
334
2260
4.18
46
Multiple Choice
Regents Practice: What is the total number of kilojoules of heat needed to change 150. grams of ice to water at 0°C?
50.1
2.22
184
484
47
Multiple Choice
Regents Practice: The temperature of 15 grams of water is increased by 3.0 Celsius degrees. How much heat was absorbed by the water?
For this question, which heat calculation formula will you use?
q = mCΔt because there's a temperature change!
q = mHf because the phase is changing from solid to liquid
q = mHv because the phase is changing from liquid to gas
48
Multiple Choice
Regents Practice: The temperature of 15 grams of water is increased by 3.0 Celsius degrees. How much heat was absorbed by the water?
189 J
21 J
50 J
76 J
49
Multiple Choice
Regents Practice: What is the total number of kilojoules of heat needed to change 150. grams of ice to water at 0°C?
For this question, which heat calculation formula will you use?
q = mCΔt because there's a temperature change!
q = mHf because the phase is changing from solid to liquid
q = mHv because the phase is changing from liquid to gas
50
Multiple Choice
Regents Practice: What is the total number of kilojoules of heat needed to change 150. grams of ice to water at 0°C?
50.1
2.22
184
484
51
Multiple Choice
Regents Practice: At 1 atmosphere of pressure, 25.0 grams of a compound at its normal boiling point is converted to a gas by the addition of 34,400 Joules. What is the heat of vaporization for this compound in Joules per gram?
25.0 J/g
34,400 J/g
2,260 J/g
1376 J/g
52
Multiple Choice
Regents Practice: At 1 atmosphere of pressure, 25.0 grams of a compound at its normal boiling point is converted to a gas by the addition of 34,400 Joules. What is the heat of vaporization for this compound in Joules per gram?
For this question, which heat calculation formula will you use?
q = mCΔt because there's a temperature change!
q = mHf because the phase is changing from solid to liquid
q = mHv because it mentioned "heat of vaporization"
53
Multiple Choice
Regents Practice: At 1 atmosphere of pressure, 25.0 grams of a compound at its normal boiling point is converted to a gas by the addition of 34,400 Joules. What is the heat of vaporization for this compound in Joules per gram?
For this question, which variable you are trying to find?
mass (m)
heat (q)
Heat of Vaporization (Hv)
54
Multiple Choice
Regents Practice: At 1 atmosphere of pressure, 25.0 grams of a compound at its normal boiling point is converted to a gas by the addition of 34,400 Joules. What is the heat of vaporization for this compound in Joules per gram?
25.0 J/g
34,400 J/g
2,260 J/g
1376 J/g
55

56

57
Multiple Choice

Regents Practice: The graph represents the vapor curves of four liquids.
Which liquid has the highest normal boiling point?
A
B
C
D
58

59

60

61
Multiple Choice
Regents Practice: which graph shows the pressure-temperature relationship expected for an ideal gas?
62
Multiple Choice
Regents Practice: Under which conditions does a real gas behave most like an ideal gas?
at high temperatures and high pressures
at high temperatures and low pressures
at low temperatures and low pressures
at low temperatures and high pressures
63
Multiple Choice
Regents Practice: One reason that a real gas deviates from a ideal gas is that the molecules of the real gas have
a straight-line motion
no net loss of energy on collision
forces of attraction for each other
a negligible volume
64
Multiple Choice
Regents Practice: Which of the following gases behave most like an ideal gas?
CO2 (g)
NH3 (g)
O2 (g)
H2 (g)
65

66

67
Multiple Choice
Regents Practice: Under which conditions will the volume of a given sample of a gas decrease?
increased pressure and decreased temperature
decreased pressure and decreased temperature
increased pressure and increased temperature
decreased pressure and increased temperature
68
Multiple Choice
Regents Practice: The volume of a 1.00-mole sample of an ideal gas will increase when,,,
increased pressure and decreased temperature
decreased pressure and decreased temperature
increased pressure and increased temperature
decreased pressure and increased temperature
69
Multiple Choice
Regents Practice: which graph represents the relationship between volume and Kelvin temperature for an ideal gas at constant pressure?
70

Same temperature, pressure, and volume
=> equal number of molecules!
71
Multiple Choice
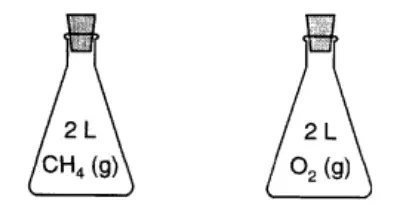
Regents Practice: Each stoppered flask on the left contains 2 liters of a gas at STP. Each gas sample has the same
number of molecules
mass
number of atoms
density
72

73

74
Multiple Choice
Regents Practice: When 7.00 moles of gas A and 3.00 moles of gas B are combined, the total pressure exerted by the gas mixture is 76.0 kPa. What is the partial pressure exerted by gas A in this mixture?
Which law will you use to answer this question?
Mole Fraction, because you are given with volumes and pressures, not only pressures.
Partial Pressure, because you are given ONLY with pressures.
75
Multiple Choice
Regents Practice: When 7.00 moles of gas A and 3.00 moles of gas B are combined, the total pressure exerted by the gas mixture is 76.0 kPa. What is the partial pressure exerted by gas A in this mixture?
22.8 kPa
53.2 kPa
7.60 kPa
76.0 kPa
76
Multiple Choice
Regents Practice: A sample of oxygen gas has a volume of 150. milliliters at 300 K. If the pressure of the sample is held constant and the temperature is raised to 600 K, the new volume of the sample will be....
Which law will you use to answer this question?
Mole Fraction, because you are given with volumes and pressures, not only pressures.
Partial Pressure, because you are given ONLY with pressures.
77
Multiple Choice
Regents Practice: A sample of oxygen gas has a volume of 150. milliliters at 300 K. If the pressure of the sample is held constant and the temperature is raised to 600 K, the new volume of the sample will be....
150. mL
300. mL
600 mL.
75.0 mL
78
Multiple Choice
Regents Practice: Gas samples A, B, and C are contained in a system at STP. The partial pressure of sample A is 38.0 kPa and the partial pressure of sample B is 19.0 kPa. What is the partial pressure of sample C?
Which law will you use to answer this question?
Mole Fraction, because you are given with volumes and pressures, not only pressures.
Partial Pressure, because you are given ONLY with pressures.
79
Multiple Choice
Regents Practice: Gas samples A, B, and C are contained in a system at STP. The partial pressure of sample A is 38.0 kPa and the partial pressure of sample B is 19.0 kPa. What is the partial pressure of sample C?
44.3 kPa
63.3 kPa
19.0 kPa
38.0 kPa

Show answer
Auto Play
Slide 1 / 79
SLIDE
Similar Resources on Wayground

72 questions
Larutan
Presentation
•
10th Grade

71 questions
M4 unit 4 Vocabulary 2
Presentation
•
10th Grade

73 questions
PREICFES 1 SESIÓN
Presentation
•
11th Grade

72 questions
Stoichiometry
Presentation
•
9th - 11th Grade

73 questions
Periodic Trends; Atomic Radius
Presentation
•
11th Grade

73 questions
Ceramics review with interactive questions.
Presentation
•
10th Grade

73 questions
Identify the States and UT
Presentation
•
10th Grade

73 questions
kelas tambahan
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade