

Grudge Ball Review
Presentation
•
Science
•
9th Grade
•
Medium
+15
Standards-aligned
Christina Slocum
Used 3+ times
FREE Resource
0 Slides • 51 Questions
1
Multiple Choice
Coca-Cola
A Homogeneous Mixture
A Heterogeneous Mixture
2
Multiple Choice
Element is a:
Pure Substance
Molecule
Mixture
Compound
3
Multiple Choice
Which one can be found on the periodic table?
Element
Compound
Molecule
4
Multiple Choice
Which of the following is not a compound?
Cl
HCl
NaCL
CO2
5
Multiple Choice

What is a mixture in which all the parts are EVENLY mixed?
Salad
Solution
Energy
Beaker
6
Multiple Choice
Matter is anything that...
Takes up space
Has mass
Takes up space AND has mass
None of these
7
Multiple Choice
What is the smallest unit of matter that has properties of that element?
Molecule
Compound
Mixture
Atom
8
Multiple Choice
electrons
positrons
neutrons
protons
9
Multiple Choice
energy
atoms
electrons
compounds
10
Multiple Choice
nucleus
electron cloud
core
center
11
Multiple Choice
6
12
3
cannot be determined
12
Multiple Choice
negatrons
electrons
neutrons
protons
13
Multiple Choice
2
1
3
6
14
Multiple Choice
protons plus the number of electrons
protons plus the number of neutrons
protons
neutrons
15
Multiple Choice
have an equal number of charged and non charged particles
have neutrons in their nuclei
have an equal number of electrons and protons
have an equal number of neutrons and protons
16
Multiple Choice
Smaller mass than a proton and a negative charge
Smaller mass than a proton and a positive charge
Greater mass than a proton and a negative charge
Greater mass than a proton and a positive charge
17
Multiple Choice
electrons
nucleus
protons
empty space
18
Multiple Choice

14
7
15
18
19
Multiple Choice
the number of protons
the number of protons and neutrons
the number of neutrons
the number of protons and electrons
20
Multiple Choice
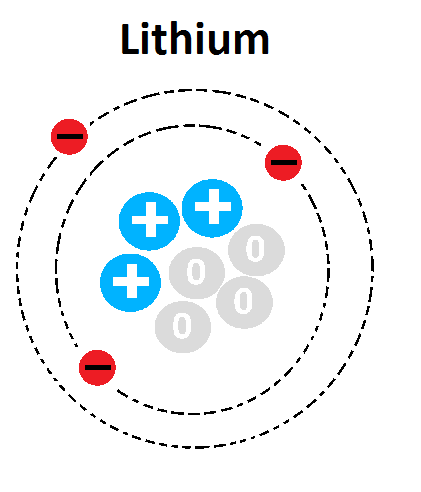
1
3
4
7
21
Multiple Choice
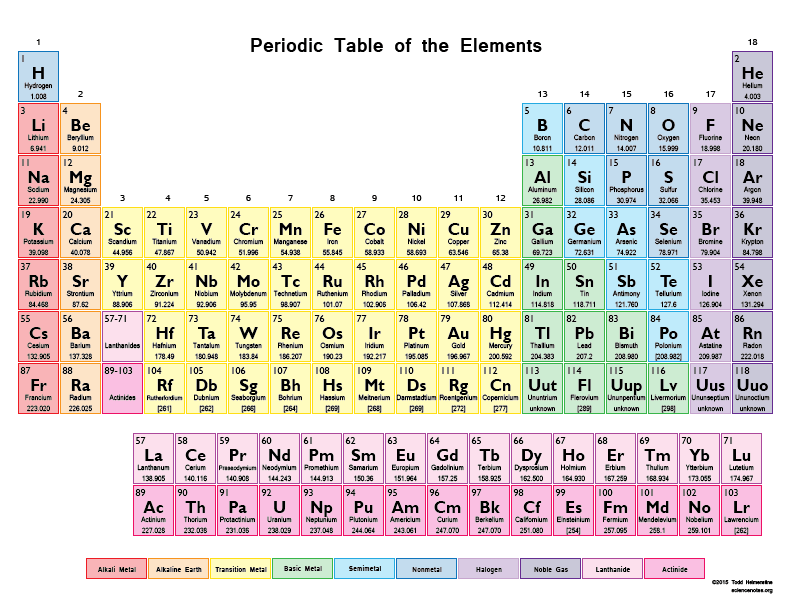
What is the atomic number of Barium, Ba? (enlarge the periodic table)
20
38
56
88
22
Multiple Choice
An element has the mass number 12 and atomic number 6. The number of neutrons in it is:
6
10
4
8
23
Multiple Choice
Atoms of the same element which have a different number of neutrons are called _________________.
ions
isotopes
quarks
molecules
24
Multiple Choice

Which one of Newton's laws is shown
3rd law
2nd law
1st law
25
Multiple Choice

What does this picture represent
Force,magnetism,acceleration
Force, money, Apples
Force, Mass, Acceleration
Funny, monkeys, Arrive
26
Multiple Choice

Which one of Newton's laws are displayed
2nd law
1st law
3rd law
27
Multiple Choice

Which of Newton's laws is shown
1st law
3rd law
2nd law
28
Multiple Choice
An object in motion will stay in motion and an object at rest will stay at rest unless acted upon by an outside force. Which law does this describe
2nd law
1st law
3rd law
29
Multiple Choice

If there is a big mass, it would need a stronger force to accelerate it. Which law does this describe?
3rd law
1st law
2nd law
30
Multiple Choice

When there is a change in motion?
When forces are balanced
When the forces are unbalanced
Whe forces are equal
Never
31
Multiple Choice
Every object in the universe exerts a force on every other object. This force is called___________.
Friction
Gravity
Push
Push
32
Multiple Choice
The measure of the gravitation force exerted by Earth on an object is the object's_________.
weight
mass
gravity
33
Multiple Choice
The amount of gravitational force between two objects depends on their____________.
color and density
mass and distance
color and shape
34
Multiple Choice

S = t/d
S = d/t
S = d x t
S = t x d
35
Multiple Choice
sec
mi
m/s
km
36
Multiple Choice
sec
mi
m/s
min
37
Multiple Choice
a. at the bottom of the first hill
b. at the top of the first hill
c. at the top of the second hill
d. at the bottom of the second hill
38
Multiple Choice
When you eat fruits and vegetables, chemical bonds are releasing stored energy, this is...
a. thermal energy
b. sound energy
c. electrical energy
d. chemical energy
39
Multiple Choice
a. turning on a stove to heat dinner
b. turning on a lamp
c. growing an apple tree
d. making toast in a toaster
40
Multiple Choice
a. law of energy changes
b. law of conservation of energy
c. law of power and energy
d. law of potential energy
41
Multiple Choice
A. Sound Energy
B. Potential Energy
C. Nuclear Energy
D. Thermal energy
42
Multiple Choice
A. Energy
B. Energy Conversion
C. Chemical Energy
D. Electrical Energy
43
Multiple Choice
Radiant Energy
Chemical Energy
Nuclear Energy
Radiation
44
Multiple Choice
_______ is the distance between waves, measured from one crest to the next crest.
Wavelength
Frequency
Amplitude
45
Multiple Choice
The _______ is the highest point on a transverse wave, and the ____ is the lowest point.
Trough, Crest
Crest, Trough
Compression, Rarefaction
Rarefaction, Compression
46
Multiple Choice
The _______ is the size or height of a wave, measured from the equilibrium to the crest or trough.
Amplitude
Wavelength
Frequency
47
Multiple Choice
Which planet is the closest planet to the sun?
Mercury
Venus
Mars
Pluto
48
Multiple Choice
Mercury
Earth
Neptune
Uranus
49
Multiple Choice
rocky
gasses
many moons
takes a long time to revolve around the sun
50
Multiple Choice
small
large
few moons
takes a short amount of time to go around the sun
51
Multiple Choice
clockwise
counter-clockwise
verticle
horizontal
Coca-Cola
A Homogeneous Mixture
A Heterogeneous Mixture
Show answer
Auto Play
Slide 1 / 51
MULTIPLE CHOICE
Similar Resources on Wayground

43 questions
Biogeochemical Cycles
Presentation
•
9th Grade

47 questions
LT 5 HR Diagram Synchronous
Presentation
•
8th Grade

44 questions
Interaction of Light Waves
Presentation
•
8th Grade

48 questions
Renewable and Nonrenewable Energy Sources
Presentation
•
8th Grade

47 questions
DNA and Cell Cycle Notes
Presentation
•
9th Grade

42 questions
De_Chem_1.6_Volcanoes
Presentation
•
9th Grade

45 questions
PMA 1 Review: 8th Grade
Presentation
•
8th Grade

48 questions
Matter and thermal Energy
Presentation
•
9th - 10th Grade
Popular Resources on Wayground

25 questions
The Ultimate College Knowledge Quiz
Quiz
•
8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Math Review
Quiz
•
6th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade
Discover more resources for Science

20 questions
MCAS Biology Review
Quiz
•
9th - 10th Grade

21 questions
Explore DNA Processes and Protein Formation
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

50 questions
living environment regents review
Quiz
•
9th Grade

17 questions
Unit 9 - Chemical and Nuclear Reactions
Quiz
•
9th - 12th Grade

50 questions
chem final review
Quiz
•
9th Grade

10 questions
Exploring Ecological Levels: Organism to Ecosystem
Interactive video
•
6th - 10th Grade

233 questions
BioPhysics B - Final Exam
Quiz
•
9th - 12th Grade