

Aqueous Systems and Solutions
Presentation
•
Chemistry
•
10th Grade
•
Hard
Richard Leachman
Used 7+ times
FREE Resource
75 Slides • 37 Questions
1

2

3

4

5

6
Draw
Draw out a how the water molecules would interact with each other.
7

8

9

10

11

12

13

14

15

16

17

18

19

20

21

22

23

24

25

26

27

28

29

30
Multiple Choice
Thermal Energy
Specific Heat
Temperature
Kinetic Energy
31
Multiple Choice
Always from cold to warm
Always from warm to cold
Both warm to cold & cold to warm
It depends on the temperature
32
Multiple Choice

Large cup (Trenta)
Small cup (Tall)
Medium cup (Venti)
All three are the same temperature so they have the same thermal energy
33

34

35

36

37

38

39

40

41

42

43

44

45
Multiple Choice
_________________ substances can dissolve in a liquid.
Insoluble
Soluble
46
Multiple Choice
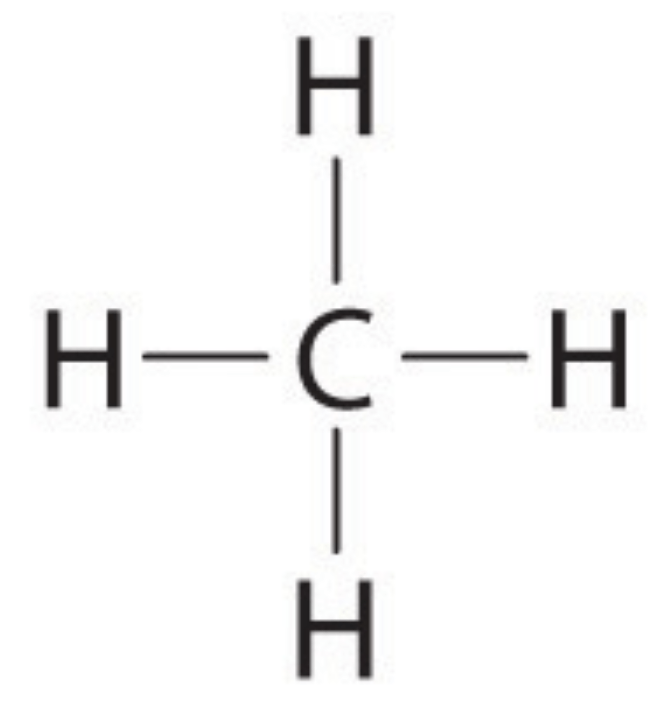
Classify the following molecule.
polar
nonpolar
47
Multiple Choice

Classify the following molecule.
polar
nonpolar
48
Multiple Choice

Why is the molecule nonpolar?
There are nonbonding pairs on the central atom.
There are different types of elements bonded to the central atom.
There are no nonbonding pairs on the central atom and all of the atoms bonded to the central atom are the same.
49
Multiple Choice

Why is the molecule polar?
There are nonbonding pairs on the central atom.
There are different types of elements bonded to the central atom.
There are no nonbonding pairs on the central atom and all of the atoms bonded to the central atom are the same.
50
Multiple Choice
Pepper is an example of a substance that is __________________ in water.
soluble
insoluble
dissolves
51
Multiple Select
Which substances are insoluble in water? (choose TWO answers)
sand
salt
sugar
flour
52
Multiple Choice

In this beaker there is some sand floating in water, but most of the sand has settled at the bottom. This would be best described as
A solution
A colloid
A suspension
A compound
53
Multiple Choice
In a ______________, one substance is dissolved in another substance.
solution
suspension
colloid
54

55

56

57

58

59

60

61
Multiple Choice

Which diagram best illustrates the ion-molecule attractions that occur when the ions of NaCl(s) are added to water?
1
2
3
4
62
Multiple Choice
A solid substance was tested in the laboratory. The test results are listed below.
• dissolves in water
• is an electrolyte
• melts at a high temperature
Based on these results, the solid substance could be
Cu
CuBr2
C
C6H12O6
63
Multiple Choice
A substance that conducts an electrical current when dissolved in water is called
catalyst
metalloid
electrolyte
nonelectrolyte
64
Multiple Choice
Which of the following is not an electrolyte?
KBr
LiOH
RbNO3
CH4
65
Multiple Choice
Which of the following best describes the components and properties of electrolytes? Electrolytes consist of –
ions and are conductive
metals and are non-polar
nonmetals and are anions.
covalent bonds and are polar
66
Multiple Choice
In order for a solution to be an electrolyte, the solute must
be a molecular compound.
restrict the flow of electricity.
create a saturated solution.
consist of positive and negative ions
67

68

69

70

71

72
Multiple Choice
Hydroxide
Hydrate
Decahydrate
None of the above
73
Multiple Choice
is composed of only hydrogen and oxygen
is a state of water
traps water inside of the compound
repels water from the compound
74
Multiple Select

ARE YOU HAPPY
yes
:)
don't click this one
no
75

76

77

78

79

80

81

82

83

84

85

86

87

88

89

90

91

92

93

94

95

96

97

98
Multiple Choice
Solution
Solvent
Solute
Compound
99
Multiple Choice
Capable of being a solid.
To have soul.
Capable of being dissolved.
To sink.
100
Multiple Choice
Colloid
Solution
Suspension
Both Orange and Blue are right
101
Multiple Choice
Colloid
Solution
Suspension
All Mixtures
102
Multiple Choice
mass/liters
moles
liters
moles/liter
103
Multiple Choice
saturated
solubility
concentration
miscible
104
Multiple Choice
contains the maximum amount of dissolved solute.
contains less solute than a saturated solution.
contains more solute than a saturated solution.
is the amount of a substance required to form a saturated solution.
105
Multiple Choice
contains the maximum amount of dissolved solute.
contains less solute than a saturated solution.
contains more solute than a saturated solution.
is the amount of a substance required to form a saturated solution.
106
Multiple Choice
contains the maximum amount of dissolved solute.
contains less solute than a saturated solution.
contains more solute than a saturated solution.
is the amount of a substance required to form a saturated solution.
107
Multiple Choice
The rapid, random movement of particles in colloidal dispersion.
A substance that dissolves in water and conducts electric current.
A substance that dissolves in water and does not conduct electric current.
The solution process when water is the solvent.
108
Multiple Choice
The rapid, random movement of particles in colloidal dispersion.
A substance that dissolves in water and conducts electric current.
A substance that dissolves in water and does not conduct electric current.
The solution process when water is the solvent.
109
Multiple Choice
colloid
solution
solute
solvent
110
Multiple Choice
colloid
solution
solute
solvent
111
Multiple Choice
2.5
0.4
2.5 moles/liter
10 moles/liter
112
Multiple Choice
9 moles NaCl
9000
9
4000 moles NaCl

Show answer
Auto Play
Slide 1 / 112
SLIDE
Similar Resources on Wayground

108 questions
Geometry Test 4 Review
Presentation
•
10th Grade

112 questions
Aqueous Solutions
Presentation
•
10th Grade

105 questions
Cells
Presentation
•
10th Grade

104 questions
AP MICRO W2021 - C3
Presentation
•
9th - 12th Grade

106 questions
Blood Type Genetic Problems
Presentation
•
9th - 12th Grade

100 questions
Review Units 1 - 5 Cutting Edge Elementary
Presentation
•
KG

99 questions
La Familia: Lesson
Presentation
•
9th Grade

107 questions
Federal Reserve
Presentation
•
12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade